Abstract
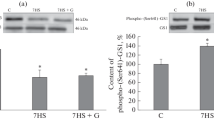
Using fluorescent microscopy, we found decreased expression of the β1 subunit of Na+/K+-ATPase and subunits of Ca2+-ATPase, increased expression of the α1S subunit of the L-type Ca2+-channel, and no changes in the expression of the α2 subunit of Na+/K+-ATPase in rat postural muscle under the conditions of modeled hypogravity. In the phasic muscle, we observed decreased expression of the β1 subunit, which was similar to that found in the postural muscle, whereas the other studied parameters remained without alterations. However, a decrease in the fluorescence intensity of the β1 subunit was insignificant due to a high variability of data. Thus, hypogravity negatively influenced primarily those skeletal muscles that are responsible for static load.
Similar content being viewed by others
Abbreviations
- m. EDL:
-
extensor digitorum longus muscle
References
Altaeva, E.G., Ogneva, I.V., and Shenkman, B.S., Dynamics of calcium levels and changes SERCA content in muscle fibers of rats and Mongolian gerbils during hind limb unloadings of various duration, Cell Tissue Biol., 2010, vol 4, no. 6, pp. 52, no. 9, pp. 594–599.
Blanco, G. and Mercer, R.W., Isozymes of the Na-KATPase: heterogeneity in structure, diversity in function, Am. J. Physiol., 1998, vol. 275, pp. F633–F650.
Chow, D.C. and Forte, J.G., Functional significance of the beta-subunit for heterodimeric P-type ATPases, J. Exp. Biol., 1995, vol. 198, pp. 1–17.
Eakle, K.A., Kabalm, M.A., Wang, S.G., and Fariey, R.A., The influence of beta subunit structure on the stability of Na+/K(+)-ATPase complexes and interaction with K(+), J. Biol. Chem., 1994, vol. 269, pp. 6550–6557.
Genin, A.M., Il’in, A.E., Kaplanskiĭ, A.S., Kasatkina, T.B., Kuznetsova, K.A., Pestov, I.D., and Smirnova, T.A., Bioethics of research on humans and animals in aviation, space and marine medicine, Aviakosm. Ekol. Med., 2001, vol. 35, no. 4, pp. 14–20.
Grigor’ev, A.I., Kozlovskaia, I.B., and Shenkman, B.S., The role of support afferents in organisation of the tonic muscle system, Ross. Fiziol. Zh. Im. I. M. Sechenova, 2004, vol. 90, no. 5, pp. 508, no. 21.
Kravtsova, V.V., Ogneva, I.V., Altaeva, E.G., Razgovorova, I.A., Tiapkina, O.V., Nikol’skiĭ, E.E., Shenkman, B.S., and Krivoĭ, I.I., Electrogenic activity of Na-KATPase and calcium ions in m. soleus fibers of rats and Mongolian gerbil during simulation of gravitational unloading, Aviakosm. Ekol. Med., 2010, vol. 44, no. 2, pp. 35–44.
Krivoi, I.I., Kravtsova, V.V., Altaeva, E.G., Kubasov, I.V., Prokof'ev, A.V., Drabkina, T.M., Nikol’sky, E.E., and Shenkman, B.S., Decrease in the electrogenic contribution of Na, K-ATPase and resting membrane potential as a possible mechanism of Ca2+ accumulation in rat soleus muscle in a short-term gravity unloading, Biophysics (Moscow), 2008, vol. 53, no. 6, pp. 586–592.
Lemas, M.V. and Fanbrough, D.M., Sequence Analysis of DNA Encoding an avian Na+, K(+)-ATPase beta 2-subunit, Biochim. Biophys. Acta, 1993, vol. 1149, pp. 339–342.
McDonough, A.A., Geering, K., and Farley, R.A., The sodium pump needs its beta subunit, FASEB J., 2002, vol. 4, pp. 1598–1605.
Morey-Holton, E.R. and Globus, R.K., Hindlimb unloading rodent model: technical aspects, J. Appl. Physiol., 2002, vol. 92, pp. 1367–1377.
Mukhina, A.M., Altaeva, E.G., Nemirovskaia, T.L., and Shenkman, B.S., The role of L-type calcium channels in the accumulation of Ca2+ in soleus muscle fibers in the rat and changes in the ratio of myosin and SERCA isoforms in conditions of gravitational unloading, Ross. Fiziol. Zh. Im. I.M. Sechenova, 2006, vol. 92, no. 11, pp. 1285, no. 1295.
Renganathan, M., Wang, Z.M., Messi, M.L., and Delbono, O., Calcium regulates L-type Ca2+ channel expression in rat skeletal muscle cells, Pflugers. Arch., 1999, vol. 438, pp. 649–655.
Seok, J.H., Hong, J.H., Jeon, J.R., Hur, G.M., Sung, J.Y., and Lee, J.H., Aldosterone directly induces Na, K-ATPase alpha 1-subunit mRNA in the renal cortex of rat, Biochem. Mol. Biol. Int., 1999, vol. 47, pp. 251–254.
Shamraj, O.I. and Lingrel, J.B., A putative fourth Na+, K(+)-ATPase alpha-subunit gene is expressed in testis, Proc. Natl. Acad. Sci. USA., 1994, vol. 91, pp. 12952–12956.
Shao, Y., Ojamaa, K., Klein, I., and Ismail-Beigi, F., Thyroid hormone stimulates Na, K-ATPase gene expression in the hemodynamically unloaded heterotopically transplanted rat heart, Thyroid, 2000, vol. 10, pp. 753–759.
Shull, G.E., Greeb, J., and Lingrel, J.B., Molecular cloning of three distinct forms of the Na+, K+-ATPase alpha-subunit from rat brain, Biochemistry, 1986, vol. 25, pp. 8125–8132.
Shull, G.E., Schwartz, A., and Lingrel, J.B., Amino-acid sequence of the catalytic subunit of the (Na+ + K+)ATPase deduced from a complementary DNA, Nature, 1985, vol. 316, pp. 691–695.
Skou, J.C. and Esmann, M., The Na, K-ATPase, J. Bioenerg Biomembr., 1992, vol. 24, pp. 249–261.
Soukup, T., Zacharová, G., and Smerdu, V., Fibre type composition of soleus and extensor digitorum longus muscles in normal female inbred Lewis rats, Acta Histochem., 2002, vol. 104, pp. 399–405.
Tyapkina, O., Volkov, E., Nurullin, L., Shenkman, B., Kozlovskaya, I., Nikolsky, E., and Vyskočil, F., Resting membrane potential and Na(+), K(+)-ATPase of rat fast and slow muscles during modeling of hypogravity, Physiol. Res., 2009, vol. 58, pp. 599–603.
Tyapkina, O.V., Malomuzh, A.I., Nurullin, L.F., and Nikol’sky, E.E., Quantal and non-quantal acetylcholine release at neuromuscular junctions of muscles of different types in a model of hypogravity, Dokl. Biol. Sci., 2013, vol. 448, pp. 4–6.
Tyapkina, O.V., Nurullin, L.F., Petrov, K.A., and Volkov, E.M., The effect of modelling of hypogravity on postsynaptic acetylcholine receptors and activity of acetylcholinesterase in neuromuscular synapses of fast and slow muscles of rat, Cell Tissue Biol., 2014a, vol 8, no. 6, pp. 499–503.
Tyapkina, O.V., Volkov, E.M., and Nurullin, L.F., The role of chloride ions in the maintenance of resting membrane potential in rat fast and slow muscles during hypogravity modeling. Bull. Exp. Biol. Med., 2014, vol. 157, no. 5, pp. 577–579.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.F. Nurullin, O.V. Tyapkina, E.M. Volkov, 2016, published in Tsitologiya, 2016, Vol. 58, No. 6, pp. 444–450.
Rights and permissions
About this article
Cite this article
Nurullin, L.F., Tyapkina, O.V. & Volkov, E.M. Studies of the expression of subunits α2 and β1 of Na+/K+-ATPase, α1S (L-type) Ca2+-channel, and SERCA 1/2/3 of Ca2+-ATPase of phasic and postural rat muscles in a model of hypogravity using the method of fluorescent microscopy. Cell Tiss. Biol. 10, 402–409 (2016). https://doi.org/10.1134/S1990519X16050096
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X16050096