Abstract
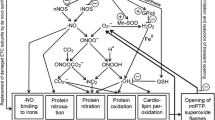
Melatonin (N-acetyl-5-methoxytryptamine), a neuroendocrine hormone of the pineal gland, participates in the modulation of the mitochondrial nonspecific pore (mitochondrial permeability transition pore, mPTP). According to the results we obtained, protoporphyrin IX (PPIX, a ligand of the TSPO translocator protein) induces the opening of the mPTP in brain mitochondria; melatonin slows the induction of the mPTP in rat brain mitochondria incubated with PPIX. Induction of the mPTP activates protein kinases/protein phosphatases that are involved in the regulation of protein phosphorylation. The TSPO modulates protein phosphorylation; in the presence of PPIX, the phosphorylation of 2′,3′-cyclic nucleotide-3′-phosphodiesterase (CNPase) increases. Here, we showed an increase in the degree of phosphorylation of CNPase in PPIX-treated rat brain mitochondria, a melatonin-induced decrease in level of CNPase phosphorylation in mitochondria incubated with PPIX; hence, we propose that melatonin participates in TSPOmodulated protein phosphorylation.
Similar content being viewed by others
References
Falcon, J., Besseau, L., Fuentes, M., Sauzet, S., Magnanou, E., and Boeuf, G., Ann. NY: Acad. Sci., 2009, vol. 1163, pp. 101–111.
Tan, D.-X., Manchester, L.C., Hardeland, R., Lopez-Burillo, S., Mayo, J.C., Sainz, R.M., and Reiter, R.J., J. Pineal Res., 2003, vol. 34, pp. 75–78.
Tan, D.X., Poeggeler, B., Reiter, R.J., Chen, L.D., Chen, S., Manchester, L.C., and Barlowwalden, L.R., Cancer Lett., 1993, vol. 70, pp. 65–71.
Menendez-Pelaez, A. and Reiter, R.J., J. Pineal Res., 1993, vol. 15, pp. 59–69.
Lopez, A., Garcia, J.A., Escames, G., Venegas, C., Ortiz, F., Lopez, L.C., and Acuna-Castroviejo, D., J. Pineal Res., 2009, vol. 46, pp. 188–198.
Acuna-Castroviejo, D., Escames, G., Leon, J., Carazo, A., and Khaldy, H., Adv. Exp. Med. Biol., 2003, vol. 527, pp. 549–557.
Castroviejo, D.A., Lopez, L.C., Escames, G., Lopez, A., Garcia, J.A., and Reiter, R.J., Curr. Top. Med. Chem., 2011, vol. 11, pp. 221–240.
Acuna-Castroviejo, D., Escames, G., Rodriguez, M.I., and Lopez, L.C., Front. Biosci.-Landmrk, 2007, vol. 12, pp. 947–963.
Martin, M., Macias, M., Escames, G., Reiter, R.J., Agapito, M.T., Ortiz, G.G., and Acuna-Castroviejo, D., J. Pineal Res., 2000, vol. 28, pp. 242–248.
Petrosillo, G., Colantuono, G., Moro, N., Ruggiero, F.M., Tiravanti, E., Di Venosa, N., Fiore, T., and Paradies, G., Am. J. Physiol. Heart Circ. Physiol., 2009, vol. 297, pp. H1487–1493.
Jou, M.J., J. Pineal Res., 2011, vol. 50, pp. 427–435.
Di Lisa, F. and Bernardi, P., Cardiovasc. Res., 2005, vol. 66, pp. 222–232.
Krestinina, O.V., Baburina, Y.L., and Azarashvili, T.S., Biol. Membrany, 2014, vol. 31, pp. 95–103.
Lim, S., Smith, K.R., Lim, S.T., Tian, R., Lu, J., and Tan, M., Cell Biosci., 2016, vol. 6, p.25.
Azarashvili, T., Krestinina, O., Odinokova, I., Evtodienko, Y., and Reiser, G., Cell Calcium, 2003, vol. 34, pp. 253–259.
Azarashvili, T., Krestinina, O., Galvita, A., Grachev, D., Baburina, Y., Stricker, R., Evtodienko, Y., and Reiser, G., Am. J. Physiol. Cell Physiol., 2009, vol. 296, pp. C1428–C1439.
Azarashvili, T., Krestinina, O., Galvita, A., Grachev, D., Baburina, Y., Stricker, R., and Reiser, G., J. Bioenerg. Biomembr., 2014, vol. 46, pp. 135–145.
Azarashvili, T., Grachev, D., Krestinina, O., Evtodienko, Y., Yurkov, I., Papadopoulos, V., and Reiser, G., Cell Calcium, 2007, vol. 42, pp. 27–39.
McEnery, M.W., Snowman, A.M., Trifiletti, R.R., and Snyder, S.H., Proc. Natl. Acad. Sci. U.S.A., 1992, vol. 89, pp. 3170–3174.
Pastorino, J.G., Simbula, G., Gilfor, E., Hoek, J.B., and Farber, J.L., J. Biol Chem., 1994, vol. 269, pp. 31041–31046.
Azarashvili, T., Krestinina, O., Yurkov, I., Evtodienko, Y., and Reiser, G., J. Neurochem., 2005, vol. 94, pp. 1054–1062.
Krestinina, O., Azarashvili, T., Baburina, Y., Galvita, A., Grachev, D., Stricker, R., and Reiser, G., Neurochem. Int., 2015, vol. 80, pp. 41–50.
Baburina, Y., Odinokova, I., Azarashvili, T., Akatov, V., Lemasters, J.J., and Krestinina, O., BBA-Biomembr., 2017, vol. 1859, pp. 94–103.
Brydon, L., Roka, F., Petit, L., de Coppet, P., Tissot, M., Barrett, P., Morgan, P.J., Nanoff, C., Strosberg, A.D., and Jockers, R., Mol. Endocrinol., 1999, vol. 13, pp. 2025–2038.
Niles, L.P. and Hashemi, F., Cell Mol. Neurobiol., 1990, vol. 10, pp. 553–558.
Morgan, P.J., Barrett, P., Howell, H.E., and Helliwell, R., Neurochem. Int., 1994, vol. 24, pp. 101–146.
Chan, A.S., Lai, F.P., Lo, R.K., Voyno-Yasenetskaya, T.A., Stanbridge, E.J., and Wong, Y.H., Cell. Signal., 2002, vol. 14, pp. 249–257.
Schuster, C., Williams, L.M., Morris, A., Morgan, P.J., and Barrett, P., J. Neuroendocrinol., 2005, vol. 17, pp. 170–178.
Petrosillo, G., Moro, N., Paradies, V., Ruggiero, F.M., and Paradies, G., J. Pineal Res., 2010, vol. 48, pp. 340–346.
Krestinina, O.V., Kruglov, A.G., Grachev, D.E., Baburina, Y.L., Evtodienko, Y.V., Moshkov, D.A., Santalova, I.M., and Azarashvili, T.S., Biol. Membrany, 2010, vol. 27, pp. 177–183.
Reiser, G., Kunzelmann, U., Steinhilber, G., and Binmoller, F.J., Neurochem. Res., 1994, vol. 19, pp. 1479–1485.
Afonso, S., Vanore, G., and Batlle, A., Free Radic. Res., 1999, vol. 31, pp. 161–170.
Krestinina, O.V., Grachev, D.E., Odinokova, I.V., Reiser, G., Evtodienko, Y.V., and Azarashvili, T.S., Biochemistry (Moscow), 2009, vol. 74, pp. 421–429.
Krestinina, O.V., Cand. Sc. (Biol.) Dissertation, Pushchino: ITEB RAN, 2005.
McEnery, M.W., Dawson, T.M., Verma, A., Gurley, D., Colombini, M., and Snyder, S.H., J. Biol. Chem., 1993, vol. 268, pp. 23289–23296.
Baburina, Y., Azarashvili, T., Grachev, D., Krestinina, O., Galvita, A., Stricker, R., and Reiser, G., Neurochem. Int., 2015, vol. 90, pp. 46–55.
Krestinina, O.V, Odinokova, I.V., Baburina, Yu.L., and Azarashvili, T.S., Biol. Membr., 2017, vol. 34, pp. 42–47.
Myllykoski, M., Itoh, K., Kangas, S.M., Heape, A.M., Kang, S.U., Lubec, G., Kursula, I., and Kursula, P., J. Neurochem., 2012, vol. 123, pp. 515–524.
Myllykoski, M., Seidel, L., Muruganandam, G., Raasakka, A., Torda, A.E., and Kursula, P., Brain Res., 2016, vol. 1641, pt. A, pp. 64–78.
Baburina, Y.L., Gordeeva, A.E., Moshkov, D.A., Krestinina, O.V., Azarashvili, A.A., Odinokova, I.V., and Azarashvili, T.S., Biochemistry (Moscow), 2014, vol. 79, pp. 555–565.
Baburina, Y., Odinokova, I., Azarashvili, T., Akatov, V., Lemasters, J.J., and Krestinina, O., Biochim. Biophys. Acta, 2017, vol. 1859, pp. 94–103.
de Faria, Poloni J., Feltes, B.C., and Bonatto, D., Funct. Integr. Genomics, 2011, vol. 11, pp. 383–388.
Williams, L.M., Drew, J.E., Bunnett, N.W., Grady, E., Barrett, P., Abramovich, D.R., Morris, A., and Slater, D., J. Neuroendocrinol., 2001, vol. 13, pp. 94–101.
Sprinkle, T.J., Crit. Rev. Neurobiol., 1989, vol. 4, pp. 235–301.
Leung, A.W. and Halestrap, A.P., Biochim. Biophys. Acta, 2008, vol. 1777, pp. 946–952.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.V. Krestinina, Yu.L. Baburina, I.V. Odinokova, T.S. Azarashvili, V.S. Akatov, 2018, published in Neirokhimiya, 2018, Vol. 35, No. 1, pp. 35–41.
Rights and permissions
About this article
Cite this article
Krestinina, O.V., Baburina, Y.L., Odinokova, I.V. et al. Melatonin Modulates Phosphorylation of 2′,3′-Cyclic Nucleotide-3′-Phosphodiesterase in the Presence of Protoporphyrin IX in the Brain Mitochondria of Rats during the Functioning of the Non-Specific Mitochondrial Pore. Neurochem. J. 12, 33–40 (2018). https://doi.org/10.1134/S1819712418010051
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1819712418010051