Abstract
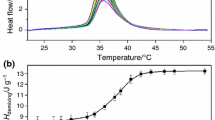
The effect of salts on the behavior of aqueous solutions of poly(N-vinylpyrrolidone), which is known to show no thermal sensitivity in the absence of additives, has been studied. The introduction of some salts, for example, KF, KH2PO4, K2CO3, and Na2SO4, leads to the occurrence of phase transitions. The corresponding phase transition temperatures depend on the nature and concentration of salts. Even moderate concentrations (0.3–0.6 mol/l) of the most active salts (K2CO3 and Na2SO4) are shown to reduce the phase transition temperature to 10–20°C.
Similar content being viewed by others
References
Yu. E. Kirsh, Prog. Polym. Sci. 18, 519 (1991).
Yu. E. Kirsh, Poly(N-vinylpyrrolidone) and Other Poly(N-vinylamides) (Nauka, Moscow, 1998) [in Russian].
Y. Akin, E. Celik, W. Sigmund, and Y. S. Hascicek, IEEE Trans. Appl. Supercond. 13, 2563 (2003).
K. Park and H. Park, in The Polymeric Materials Encyclopedia, Ed. by J. C. Salomone (CRC, Boca Raton, 1996), Vol. 10, p. 7785.
V. O. Aseyev, H. Tenhu, and F. M. Winnik, Adv. Polym. Sci. 196, 1 (2006).
Y. Maeda, T. Nakamura, and I. Ikeda, Macromolecules 35, 217 (2002).
A. A. A. DeQueiroz, A. Gallardo, and J. S. Roman, Biomaterials 21, 1631 (2000).
Yu. E. Kirsh, N. A. Yanul, K. K. Kalainsh, and V. G. Maslov, J. Mol. Liq. 82, 117 (1999).
Yu. E. Kirsh, N. A. Yanul’, Yu. M. Popkov, and S. F. Timashev, Zh. Fiz. Khim. 73, 313 (1999).
Yu. E. Kirsh and K. K. Kalnin’sh, Zh. Prikl. Khim. (S.-Peterburg) 72, 1233 (1999).
M. Ataman, Colloid Polym. Sci. 265, 19 (1987).
B. I. Nakhmanovich, Ya. G. Urman, E. V. Kristal’nyi, and A. A. Arest-Yakubovich, Polymer Science, Ser. B 45, 135 (2003) [Vysokomol. Soedin., Ser. B 45, 978 (2003)].
Author information
Authors and Affiliations
Additional information
Original Russian Text © B.I. Nakhmanovich, N.I. Pakuro, E.I. Akhmet’eva, G.I. Litvinenko, A.A. Arest-Yakubovich, 2007, published in Vysokomolekulyarnye Soedineniya, Ser. B, 2007, Vol. 49, No. 5, pp. 941–944.
This work was supported by the Russian Foundation for Basic Research, project no. 06-03-32505.
Rights and permissions
About this article
Cite this article
Nakhmanovich, B.I., Pakuro, N.I., Akhmet’eva, E.I. et al. Thermal sensitivity of poly(N-vinylpyrrolidone) solutions in water-saline media. Polym. Sci. Ser. B 49, 136–138 (2007). https://doi.org/10.1134/S1560090407050077
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1134/S1560090407050077