Abstract
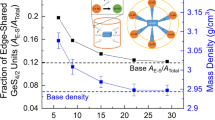
The formation of a bubble structure in the glass of composition 26Li2O · 74SiO2 is investigated. The stable reproducible characteristics of the bubble structure are obtained, namely, dependences of the number of bubbles on the conditions of glass synthesis, i.e., the synthesis temperature, the synthesis time, and the type of initial reagents. It is shown that the number of bubbles decreases with an increase in the height of the sample (glass melt in the crucible), as well as with an increase in the synthesis temperature at a fixed synthesis time or with an increase in the synthesis time for each specific temperature. The bubble size distribution curves are obtained for all synthesis conditions. The reproducible bubble structure is determined for each layer of the glass sample, which is subsequently used in studying the kinetics of heterogeneous nucleation.
Similar content being viewed by others
References
Slavyanskii, V.T., Gazy v stekle (Gases in Glass), Moscow: Oborongiz, 1957 [in Russian].
Shvetsov, B.S., On the Stratification of Glass during Its Cooling, Izv. Akad. Nauk SSSR, Otd. Mat. Estestv. Nauk, 1931, no. 8, pp. 1141–1150.
Kramer, F.W. Analysis and Interpretation of Gas Bubbles, in Proceedings of the NVG/NCNG Colloquium “Melting Processes in Glass Furnaces,” Aachen, Germany, March 4–5, 1998, Schaeffer, H.A. and Beerkens, R.G.C., Eds., Frankfurt (Main): Deutschen Glastechnischen Gesellschaft, 1998, pp. 90–95.
Roggendorf, H., Waldecker, G.G., and Scholze, H., Composition of Waste Gases from Glass Tank Furnaces, Glastech. Ber., 1988, vol. 56, no. 8, pp. 218–227.
Balkanli, B. and Ungan, A., Numerical Simulation of Bubble Behavior in Glass Melting Tanks: Part 2. Dissolved Gas Concentration, Glass Technol., 1996, vol. 37, no. 3, pp. 101–105.
Swarts, E.L., Bubble Generation at Glass/Refractory Interfaces: A Review of Fundamental Mechanisms and Practical Considerations, Glastech. Ber., 1992, vol. 65, no. 4, pp. 87–92.
Roi, T., Saidel, O., Nolle, G., and Hohne, D., Modeling of the Bubble Population in Glass Melts, Glastech. Ber., 1994, vol. 67, no. 10, pp. 263–271.
Izumitani, T. and Terai, R., Formation and Growth of Bubbles in the Process of Glass Melting, Yogyo Kyokaishi (J. Ceram. Assoc. Jpn.), 1962, vol. 70, no. 799, pp. 205–215.
Boganov, A.G., Rudenko, V.S., and Cheremisin, I.I., Mechanism of Formation and Growth of Bubbles in Silica Glass, Fiz. Khim. Stekla, 1984, vol. 10, no. 2, pp. 28–216.
Cheremisin, I.I., Rudenko, V.S., and Boganov, A.G., Kinetics of Dissolution of Gas Bubbles under High-Temperature Compression of Silica Glass, Fiz. Khim. Stekla, 1984, vol. 10, no. 3, pp. 365–373.
Nemec, L. and Tonatova, V., Behavior of Bubbles in Glass Melts under Effect of the Gravitational and Centrifugal Fields, Ceram.—Silik., 2005, vol. 49, no. 3, pp. 162–169.
Nemec, L., Jebava, M., and Cincibusova, P., The Removal of Bubbles from Glass Melts in Horizontal or Vertical Channels with Different Glass Flow Patterns, Ceram.—Silik., 2006, vol. 50, no. 3, pp. 140–152.
Vasnetsov, V.B., Shardakov, N.T., Kudyakov, V.Ya., and Deryabin, V.A., Formation of Bubble Structure in Films of Glass-Forming Melts, Fiz. Khim. Stekla, 1999, vol. 25, no. 3, pp. 373–377 [Glass Phys. Chem. (Engl. transl.), 1999, vol. 25, no. 3, pp. 281–284].
Sycheva, G.A., Influence of the Bubble Structure on the Nucleation Kinetics of Crystals in the 26Li2O · 74SiO2 Glass, Fiz. Khim. Stekla (in press) [Glass Phys. Chem. (Engl. transl.) (in press)].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.A. Sycheva, 2009, published in Fizika i Khimiya Stekla.
Rights and permissions
About this article
Cite this article
Sycheva, G.A. Formation of the bubble structure in the 26Li2O · 74SiO2 glass. Glass Phys Chem 35, 267–273 (2009). https://doi.org/10.1134/S1087659609030055
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659609030055