Abstract
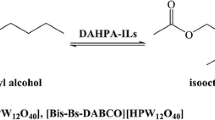
Inorganic ionic liquids are first shown to catalyze the alcoholysis of heptacyclo-[8.4.0.02,12.03,8.04,6.05,9.011,13]tetradecane (heptacyclic dimer of norbornadiene, binor-S) with primary and secondary alcohols. The reaction occurs at 65−100°C for 6−14 h via regioselective cleavage of the cyclopropane C4−C5 bond in binor-S to form 10-exo-alkoxyhexacyclo[9.2.1.02,7.03,5.04,8.09,13] tetradecanes in 85−90% yields.
Similar content being viewed by others
References
Koch, V.R., Miller, L.L., and Osteryoung, R.A., J. Am. Chem. Soc., 1976, vol. 98, p. 5277. doi https://doi.org/10.1021/ja00433a037
Qiao, K. and Deng, Y., J. Mol. Catal. A: Chem., 2001, vol. 17, p. 81. doi 10.1016/s13811169(01)00107-8
Wilkes, J.S., Frye, J.S., and Reynolds, G.F., Inorg. Chem., 1983, vol. 22, p. 3870. doi https://doi.org/10.1021/ic00168a011
Xin, H., Wu, Q., Han, M., Wang, D., and Jin, Y., Appl. Catal., A, 2005, vol. 292, p. 354. doi https://doi.org/10.1016/j.apcata.2005.06.012
Sun, X.W. and Thao, S.Q., Pet. Sci., 2011, vol. 8, p. 495. doi https://doi.org/10.1007/s12182-011-0167-4
Kim, D.S. and Ahn, W.S., Korean J. Chem. Eng., 2003, vol. 20, p. 39. doi https://doi.org/10.1007/bf02697182
Boon, J.A., Levisky, J.A., Pflug, J.L., and Wilkes, J.S., J. Org. Chem., 1986, vol. 51, p. 480. doi https://doi.org/10.1021/jo00354a013
Adams, C.J., Earle, M.J., and Seddon, K.R., Chem. Commun., 1998, p. 2097. doi https://doi.org/10.1039/a805599h
Xin-hua, Y., Min, C., Qi-xun, D., and Xiao-nong, C., Chem. Eng. J., 2009, vol. 146, p. 266. doi https://doi.org/10.1016/j.cej.2008.09.022
Adams, C.J., Earle, M.J., and Seddon, K.R., Chem. Commun., 1999, p. 1043. doi https://doi.org/10.1039/a901302d
Yoo, K., Namboodiri, V.V., Varma, R.S., and Smimiotis, P.G., J. Catal., 2004, vol. 222, p. 511. doi https://doi.org/10.1016/j.jcat.2003.11.018
Zhang, J., Huang, C., Chen, B., Ren, P., and Pu, M., J. Catal., 2007, vol. 249, p. 261. doi https://doi.org/10.1016/j.jcat.2007.04.019
Rongan, X., Rui, Z., Xianghai, M., Zhichang, L., Jiaying, M., and Chunming, X., Pet. Sci., 2011, vol. 8, p. 219. doi https://doi.org/10.1007/s12182-011-0138-9
Huang, M.Y., Wu, J.C., Shieu, F.S., and Lin, J.J., Fuel, 2011, vol. 90, p. 1012. doi https://doi.org/10.1016/j.fuel.2010.11.041
Huang, M.Y., Wu, J.C., Shieu, F.S., and Lin J.J., Catal. Commun., 2009, vol. 10, p. 1747. doi https://doi.org/10.1016/j.catcom.2009.05.030
Khusnutdinov, R.I., Mukminov, R.R., Aminov, R.I., Khalilov, L.M., Mescheryakova, E.S., and Dzhemi-lev, U.M., Tetrahedron Lett., 2015, vol. 56, p. 536. doi https://doi.org/10.1016/j.tetlet.2014.12.006
Dzhemilev, U.M., Khusnutdinov, R.I., Muslimov, Z.S, Tolstikov, G.A., and Nefedov, O.M., Russ. Chem. Bull., 1991, vol. 40, p. 236. doi https://doi.org/10.1007/BF00959680
Khusnutdinov, R.I., Muslimov, Z.S, Dzhemilev, U.M., and Nefedov, O.M., Russ. Chem. Bull., 1993, vol. 42, p. 692. doi https://doi.org/10.1007/BF00704004
Burdakova, E.S., Candidate Sci. (Techn.) Dissertation, Ufa, 2017.
Weissberg, A., Proskauer E., Riddick, J., and Toops, E., Organic Solvents, New York: Interscience, 1955.
Acknowledgments
Structural studies were performed using the equipment of the Agidel Regional Center for Collective Use.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Organicheskoi Khimii, 2019, Vol. 55, No. 5, pp. 677–682.
Rights and permissions
About this article
Cite this article
Aminov, R.I., Khusnutdinov, R.I. Alcoholysis of Binor-S with Alcohols under the Action of Ionic Liquid. Russ J Org Chem 55, 587–591 (2019). https://doi.org/10.1134/S1070428019050014
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428019050014