Abstract
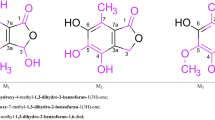
1,3-Benzodioxoles synthesized by condensation of 3,6-di-tert-butylbenzene-1,2-diol with carbonyl compounds showed antiradical activity due to their ability to undergo one-electron oxidation with formation of stable radical cations. On this basis, the antiknock effect of their structural analogs, 1,3-dioxolanes derived from vicinal diols, was interpreted in terms of oxidation of these compounds with active radicals generated from fuel hydrocarbons to produce more stable radical or radical ion species, depending on the fuel composition. The formation of radical species was detected in model oxidation reactions of 2,2-dimethyl-1,3-dioxolane and 2,2-dimethyl-1,3-dioxolan-4-ylmethanol with radicals generated by photolysis of iron(III) chloride and benzoyl peroxide.
Similar content being viewed by others
References
Varbanskaya, R.A., Pudov, V.S., Komissarova, N.L., Belostotskaya, I.S., Vol’eva, V.B., and Ershov, V.V., USSR Inventor’s Certificate no. 1 143 752, 1984; Byull. Izobret., 1985, no. 9.
Malysheva, N.N., Prokof’ev, A.I., Solodovnikov, S.P., Bubnov, N.N., Prokof’eva, T.I., Vol’eva, V.B., Ershov, V.V., and Kabachnik, M.I., Izv. Akad. Nauk SSSR, Ser. Khim., 1988, p. 1040.
Vol’eva, V.B., Belostotskaya, I.S., Novikova, I.A., Dzhuaryan, E.V., and Ershov, V.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1980, p. 2414.
Komissarova, N.L., Belostotskaya, I.S., Vol’eva, V.B., Dzhuaryan, E.V., Novikova, I.A., and Ershov, V.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1981, p. 2360.
Varfolomeev, S.D., Nikiforov, G.A., Vol’eva, V.B., Makarov, G.G., and Trusov, L.I., Russian Patent no. 2 365 617, 2009; Byull. Izobret., 2009, no. 24.
Pokholok, T.V., Zaitseva, N.I., Pariysky, G.B., and Toptygin, D.Ya., Polym. Photochem., 1982, vol. 2, p. 429.
Pariiskii, G.B., Toptygin, D.Ya., Davydov, E.Ya., Ledneva, O.A., Mikheev, Yu.A., and Karasev, V.M., Vysokomol. Soedin., Ser. B, 1972, vol. 14, p. 511.
Pshezhetskii, S.Ya., Kotov, A.G., Milinchuk, V.K., Roginskii, V.A., and Tupikov, V.I., EPR svobodnykh radikalov v radiatsionnoi khimii (ESR of Free Radicals in Radiation Chemistry), Moscow: Khimiya, 1972, p. 52.
Dyment, O.N., Kazanskii, K.S., and Miroshnikov, A.M., Glikoli i drugie proizvodnye okisi etilena i propilena (Glycols and Other Derivatives of Ethylene and Propylene Oxides), Dyment, O.N., Ed., Moscow: Khimiya, 1976, p. 121.
Kasaikina, O.T., Kartasheva, Z.S., and Pisarenko, L.M., Russ. J. Gen. Chem., 2008, vol. 78, p. 1533.
Vol’eva, V.B., Novikova, I.A., Ostapets-Sveshnikova, G.D., Belostotskaya, I.S., and Ershov, V.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1980, p. 2416.
Dzhuaryan, E.V., Belostotskaya, I.S., and Ershov, V.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1973, p. 658.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Full Member of the Russian Academy of Sciences I.P. Beletskaya on her jubilee
Original Russian Text © V.B. Vol’eva, I.S. Belostotskaya, N.L. Komissarova, A.V. Malkova, T.V. Pokholok, E.Ya. Davydov, 2013, published in Zhurnal Organicheskoi Khimii, 2013, Vol. 49, No. 3, pp. 458–461.
Rights and permissions
About this article
Cite this article
Vol’eva, V.B., Belostotskaya, I.S., Komissarova, N.L. et al. Antiradical activity of dioxolane derivatives. Russ J Org Chem 49, 446–449 (2013). https://doi.org/10.1134/S1070428013030226
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428013030226