Abstract
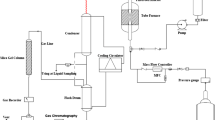
A catalyst based on zeolite was synthesized using the hydrothermal method, and alumina phosphate was embedded in its structure. The correspondent reactor tests were carried out to assess the synthesized catalyst where methanol with a purity of 99.9% was used. The results indicated that alumina phosphate is considerably effective on catalyst functionality. Given the results, the best performance was obtained when the ratio of alumina phosphate to alumina is 0.7 weighted in catalyst and fixed-bed reactor serves in a temperature of 300°C and WHSV of 0.8 h‒1. Structure and morphology of synthesized catalyst were characterized by BET, XRF, SEM, and XRD. The catalyst was evaluated in the process of converting methanol to dimethyl ether in a fixed-bed reactor under operating conditions of 300°C, 1 atm and 0.5 mL min‒1 of feed (pure methanol). The results of test indicated by increasing the amount of alumina phosphate to alumina, the conversion rate of methanol was increased to a constant value and does not change in ratios higher than 0.7. Moreover, the methanol conversion rate will reach temperature 300°C at 84%.
Similar content being viewed by others
References
Van Noyen, J., De Wilde, A., Schroeven, M., Mullens, S., and Luyten, J., Int. J. Appl. Ceramic Tech., 2012, vol. 9, no. 5, pp. 902–910.
Narula, C.K., Daw, C.S., Hoard, J.W., and Hammer., T., Int. J. Appl. Ceramic Tech., 2005, vol. 2, no. 6, pp. 452–466.
Yilmaz, B., Topics in Catalysis, 2009, vol. 52, no. 6, pp. 888–895.
Garcla, J., Li, K., and Davis, M.E., Mesoporous Zeolites: Preparation, Characterization and Applications, Wiley, 2015.
Rownaghi, A.A. and Hedlund, J., Ind. & Eng. Chem. Res., 2011, vol. 50, no. 21, pp. 11872–11878.
Bjorgen, M., Joensen, F., Holm, M.S., Olsbye, U., Lillerud, and Svelle, S., Appl. Catal. A-Gen., 2008, vol. 345, no. 1, pp. 43–50.
Zaidi, H.A. and Pant, K.K., Korean J. Chem. Eng., 2005, vol. 22, no. 3, pp. 353–357.
Conte, M., Lopez-Sanchez, J.A., He, Q., Morgan, D.J., Ryabenkova, Y., Bartley, J.K., et al., Catal. Sci. Technol., 2012, vol. 2, no. 1, pp. 105–112.
Hajimirzaee, S., Ainte, M., Soltani, B., Behbahani, R.M., Leeke, G.A., and Wood, J., Chem. Eng. Res. & Design, 2015, vol., no. 93, pp. 541–553.
Yaripour, F., Mollavali, M., Jam, S.M., and Atashi, H., Energy & Fuels, 2009, vol. 23, no. 4, pp. 1896–1900.
Zaidi, H.A. and Pant, K.K., Canadian J. Chem. Eng., 2005, vol. 83, no. 6, pp. 970–977.
Kianfar, E., Salimi, M., Pirouzfar, V., and Koohestani, B., Int. J. Appl. Ceram. Technol., 2018, vol. 15, no. 3, pp. 734–741.
Treacy, M.M. and Higgins, J.B., Collection of Simulated XRD Powder Patterns for Zeolites, 5th ed., Elsevier, vol. 2007.
Koekkoek, A.J.J., Kim, W., Degirmenci, V., Xin, H., Ryoo, R., Hensen, E.J.M., J. Catalysis, 2013, vol. 299, pp. 81–90.
Isernia, L.F., Mat. Res., 2013, no. 16, pp. 792–802.
Shukla, D.B. and Pandya, V.P., J. Chem. Tech. & Biotechn., 1989, vol. 44, no. 2, pp. 147–154.
Cañizares, P., de Lucas, A., Dorado, F., Durán, A., and Asencio, I., Appl. Catalysis A: General, 1998, vol. 169, no. 1, pp. 137–150.
Van Donk, S., Janssen, A.H., Bitter, J.H., and de Jong K.P., Catalysis Reviews, 2003, vol. 45, no. 2, pp. 297–319.
Ni, Y., Sun, A., Wu, X., Hai, G., Hu, J., Li, T., et al., Micropor. & Mesopor. Mat., 2011, vol. 143, no. 2, pp. 435–442.
Wu, W. and Weitz, E., Appl. Sur, Sci., 2014, vol. 316, pp. 405–415.
Cruz-Cabeza, A.J., Esquivel, D., Jiménez-Sanchidrián, C., and Romero-Salguero, F.J., Materials, 2012, vol. 5, no. 1, pp. 121–134
Biscardi, J.A., Meitzner, G.D., and Iglesia, E., J. Catalysis, 1998, vol. 179, no. 1, pp. 192–202.
Kianfar, E., Salimi, M., Pirouzfar, V., and Koohestani, B., Synthesis and Modification of Zeolite ZSM-5 Catalyst with Solutions of Calcium Carbonate (CaCO3) and Sodium Carbonate (Na2CO3) for Methanol to Gasoline Conversion, 2018, https://doi.org/10.1515/ijcre-2017-0229.
Bjørgen, M., Svelle, S., Joensen, F., Nerlov, J., Kolboe, S., Bonino, F., Palumbo, L., Bordiga, S., and Olsbye, U., J. Catal., 2007, vol. 249, pp. 195–207.
Shareh, F.B., Kazemeini, M., Asadi, M., and Fattahi, M., Pet. Sci. Technol., 2014, vol. 32, pp. 1349–1356.
Wan, Z., Wu, W., Chen, W., Yang, H., and Zhang, D., Ind. Eng. Chem. Res., 2014, vol. 53, no. 50, pp. 19471–19478.
Dagle, R.A., Lizarazo-Adarme, J.A., Lebarbier Dagle, V., Gray, M.J., White, J.F., King, D.L., and Palo, D.R., Fuel Process. Technol., 2014, vol. 123, pp. 65–74.
Bjørgen, M., Joensen, F., Lillerud, K.P., Olsbye, U., and Svelle, S., Catal. Today, 2009, vol. 142, pp. 90–97.
Wu, L., Degirmenci, V., Magusin, P.C.M.M., Lousberg, N.J.H.G.M., and Hensen, E.J.M., J. Catal., 2013, vol. 298, pp. 27–40.
Lee, J.H., Park, M.B., Lee, J.K., Min, H.-K., Song, M.K., and Hong, S.B., J. Am. Chem. Soc., 2010, vol. 132, pp. 12971–12982.
Di Zuo xing, Cheng, Y., Jiao, X., Li, J., Wu, J., and Zhang, D., Fuel, 2013, vol. 104, pp. 878–881.
Hosseini, S., Taghizadeh, M., and Eliassi, A., J. Natural Gas Chem., 2012, vol. 21, pp. 344–351.
Hassanpour, S., Yaripour, F., and Taghizadeh, M., Fuel Proc. Tech., 2010, vol. 91, pp. 1212–1221.
Sabour, B., Peyrovi, M.H., Hamoule, T., and Rashidzadeh, M., J. Ind. & Eng. Chem., 2014, vol. 20, pp. 222–227.
Rownaghi, A.A., Rezaei, F., Stante, M., and Hedlund, J., Appl. Catal., B: Env., 2012, vols. 119–120, pp. 56–61.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the author in English.
Rights and permissions
About this article
Cite this article
Kianfar, E. Synthesis and Characterization of AlPO4/ZSM-5 Catalyst for Methanol Conversion to Dimethyl Ether. Russ J Appl Chem 91, 1711–1720 (2018). https://doi.org/10.1134/S1070427218100208
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427218100208