Abstract
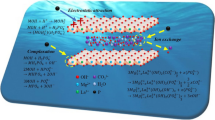
Crown ether–SiO2–TiO2 composites were obtained by adding crown ethers (dibenzo-18-crown-6 or dibenzo-21-crown-7) during the sol–gel synthesis of SiO2–TiO2. The behavior of the composites in the sorption of several alkali, alkaline-earth, and rare-earth metal cations from acidic solutions was investigated. The crown ether–SiO2–TiO2 composites of the crown ethers showed higher efficiency and selectivity in the sorption of barium cations, as well as ytterbium cations in the presence of Sr(II), Ce(III), La(III), and Nd(III) compared to the parent crown ethers. It was established by FTIR spectroscopy that the crown ether molecule immobilized on the oxide surface has a distorted structure, which can explain the change in the selectivity of complex formation in going from crown ethers to their composites.
Similar content being viewed by others
References
Crown Ethers and Analogs, Patai, S. and Rappoport, Z., Eds., New York: Wiley, 1989, p. 234.
Hiraoka, M., Crown Compounds. Their Characteristics and Applications, Amsterdam: Elsevier, 1982, p. 363.
King, R. B. and Heckley, P.R., J. Am. Chem. Soc., 1974, vol. 96, no. 10, p. 3118.
Nesterov, S.V., Russ. Chem. Rev., 2000, vol. 69, no. 9, p. 769. doi 10.1070/RC2000v069n09ABEH000586
Murashkevich, A.N., Alisienok, O.A., Zharskii, I.M., and Yukhno, E.K., J. Sol–Gel Sci. Technol., 2013, vol. 68, no. 3, p. 509. doi 10.1007/s10971-012-2947-8
Yakshin, V.V., Vilkova, O.M., Pluzhnik-Gladyr, S.M., and Kotlyar, S.A., Macroheterocycles, 2010, vol. 3, nos. 2–3, p. 114. doi 10.6060/mhc2010.2-3.114
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.V. Fedorova, A.I. Maksimovskikh, O.V. Koryakova, G.L. Rusinov, A.N. Murashkevich, V. N. Rychkov, V.N. Charushin, 2015, published in Rossiiskii Khimicheskii Zhurnal, 2015, Vol. 59, No. 5, pp. 92–97.
Rights and permissions
About this article
Cite this article
Fedorova, O.V., Maksimovskikh, A.I., Koryakova, O.V. et al. Crown Ether–SiO2–TiO2 Composites in the Sorption of Metal Ions from Acidic Aqueous Solutions. Russ J Gen Chem 88, 362–367 (2018). https://doi.org/10.1134/S1070363218020299
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363218020299