Abstract
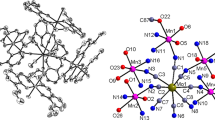
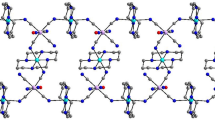
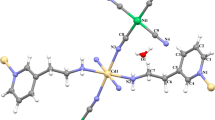
By using K2[Ni(CN)4] as a building block and two Mn(III) compounds containing bicompartimental Schiff-base ligands as assembling segments, two new cyanide-bridged Ni–Mn complexes of the formula {[Mn(L1)(H2O)]4[Ni(CN)4]}[ClO4]2·2CH3CN (1) and {[Mn(L2)(H2O)]2[Ni(CN)4]}·CH3CN H2O (2) (L1 = N,N’-1,2-propylene-bis(3-methoxysalicylideneiminate; L2 = N,N’-1,2-propylene-bis(3-ethoxysalicylideneiminate) have been synthesized and characterized by elemental analysis, IR spectroscopy and X-ray analysis. Single Xray diffraction analysis revealed the cationic pentanuclear and neutral trinuclear structures for complexes 1 and 2, respectively, and indicated that the structure of the Schiff-base ligand had obvious influence on the structural types of the target cyanide-bridged complexes. Both cyanide-bridged complexes are self-complementary via coordinated aqua ligand from one complex and the free O4 compartment from the neighboring complex, therefore giving supramolecular two-dimensional network and one-dimensional zig-zag chain structure. Study of magnetic properties revealed weak antiferromagnetic coupling within the Mn2 dimer formed by the intermolecular hydrogen bond.
Similar content being viewed by others
References
Rodríguez-Velamazán, J.A., González, M.A., Real, J.A., Castro, M., Muñoz, M.C., Gaspar, A.B., Ohtani, R., Ohba, M., Yoneda, K., Hijikata, Y., Yanai, N., Mizuno, M., Ando, H., and Kitagawa, S., J. Am. Chem. Soc., 2012, vol. 134, no. 11, p. 5083. doi 10.1021/ja206228n
Jeon, I.R., Calancea, S., Panja, A., Piñero Cruz., D.M., Koumousi, E.S., Dechambenoit, P., Coulon, C., Wattiaux, A., Rosa, P., Mathonière, C., and Clérac, R., Chem. Sci., 2013, vol. 4, no. 6, p. 2463. doi 10.1039/C3SC22069A
Liu, C.M., Xiong, R.G., Zhang, D.Q., and Zhu, D.B., J. Am. Chem. Soc., 2010, vol. 132, no. 12, p. 4044. doi 10.1021/ja910310p
Miyasaka, H., Saitoh, A., and Abe, S., Coord. Chem. Rev., 2007, vol. 251, nos. 21–24, p. 2622. doi 10.1016/j.ccr.2007.07.028, and references therein.
Zhang, H.Y., Xue, C.C., Shi, J.W., Liu, H., Dong, Y.H., Zhao, Z.D., Zhang, D.P., and Jiang, J.Z., Crystal Growth & Des., 2016, vol. 16, no. 10, p. 5753. doi 10.1021/acs.cgd.6b00820
Mousavi, M., Béreau, V., Desplanches, C., Duhayonab, C., and Sutter, J.P., Chem. Commun., 2010, vol. 46, no. 40, p. 7519. doi 10.1039/C0CC02498H
Venkatakrishnan, T.S., Sahoo, S., Bréfuel, N., Duhayon, C., Paulsen, C., Barra, A.L., Ramasesha, S., and Sutter, J.P., J. Am. Chem. Soc., 2010, vol. 132, no. 17, p. 6047. doi 10.1021/ja9089389
Miyasaka, H., Matsumoto, N., Okawa, H., Re, N., Gallo, E., and Floriani, C., J. Am. Chem. Soc., 1996, vol. 118, no. 5, p. 981. doi 10.1021/ja952706c
Zhang, D.P., Wang, H.L., Chen, Y.T., Ni, Z.H., Tian, L.J., and Jiang, J.Z., Inorg. Chem., 2009, vol. 48, no. 23, p. 11215. doi 10.1021/ic901530p
Zhang, D.P. and Wang, P., Inorg. Chem. Commun., 2012, vol. 26, no. 12, p. 77. doi 10.1016/j.inoche.2012.09.013
Andruh, M., Chem. Commun., 2011, vol. 47, no. 11, p. 3025. doi 10.1039/C0CC04506C
Hu, K.Q., Jiang, X., Wu, S.Q., Cui, A.L., Liu, C.M., and Kou, H.Z., Inorg. Chem., 2015, vol. 54, no. 4, p. 1206. doi 10.1021/ic502874n
Sheldrick, G.M., SHELX-97, Universität Göttingen, Germany, 1997.
Tang, J.K., Coster, J.S., Golobi, A., Kozlevar, B., Robertazzi, A., Vargiu, A.V., Gamez, P., and Reedijk, J., Inorg. Chem., 2009, vol. 48, no. 12, p. 5473. doi 10.1021/ic900507w
Kahn, O., Molecular Magnetism, Weinheim: VCH, 1993.
Kennedy, B.J. and Murray, K.S., Inorg. Chem., 1985, vol. 24, no. 10, p. 1552. doi 10.1021/ic00204a029
Nastase, S., Tuna, F., Maxim, C., Muryn, C.A., Avarvari, N., Winpenny, R.E.P., and Andruh, M., Crystal Growth & Des., 2007, vol. 7, no. 9, p. 1825. doi 10.1021/cg070332g
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Shi, J., Lan, W., Liu, Q. et al. Hydrogen-Bond Directed Cyanide-Bridged Supramolecular 2D and 1D Bimetallic Coordination Polymers: Synthesis, Crystal Structure, and Magnetic Properties. Russ J Gen Chem 88, 319–324 (2018). https://doi.org/10.1134/S1070363218020202
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363218020202