Abstract
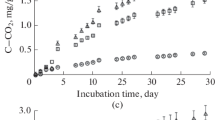
The mineralization and humification of leaf litter collected in a mixed forest of the Prioksko-Terrasny Reserve depending on temperature (2, 12, and 22°C) and moisture (15, 30, 70, 100, and 150% of water holding capacity ( WHC)) has been studied in long-term incubation experiments. Mineralization is the most sensitive to temperature changes at the early stage of decomposition; the Q 10 value at the beginning of the experiment (1.5–2.7) is higher than at the later decomposition stages (0.3–1.3). Carbon losses usually exceed nitrogen losses during decomposition. Intensive nitrogen losses are observed only at the high temperature and moisture of litter (22°C and 100% WHC). Humification determined from the accumulation of humic substances in the end of incubation decreases from 34 to 9% with increasing moisture and temperature. The degree of humification CHA/CFA is maximum (1.14) at 12°C and 15% WHC; therefore, these temperature and moisture conditions are considered optimal for humification. Humification calculated from the limit value of litter mineralization is almost independent of temperature, but it significantly decreases from 70 to 3% with increasing moisture. A possible reason for the difference between the humification values measured by two methods is the conservation of a significant part of hemicelluloses, cellulose, and lignin during the transformation of litter and the formation of a complex of humic substances with plant residues, where HSs fulfill a protectoral role and decrease the decomposition rate of plant biopolymers.
Similar content being viewed by others
References
L. N. Aleksandrova, Soil Organic Matter and the Processes of Its Transformation (Nauka, Leningrad, 1980) [in Russian].
K. S. Bobkova, A. V. Mashika, and A. V. Smagin, Dynamics of the Content of Organic Carbon in Automorphic Soils of Middle-Taiga Spruce Forests (Nauka, St. Petersburg, 2014) [in Russian].
M. M. Kononova, Problem of Soil Humus and Modern Challenges of Its Study (Academy of Sciences of the Soviet Union, Moscow, 1951) [in Russian].
E. Yu. Milanovskii, Soil Humus Substances as the Natural Hydrophobic-Hydrophilic Compounds (GEOS, Moscow, 2009) [in Russian].
M. A. Nadporozhskaya, L. B. L’vova, N. V. Kovsh, E. I. Fedoros, and O. G. Chertov, “Influence of lithogenic factor on the transformation of nitrogen compounds in forest soils,” Proceedings of the V All-Russian Conf. with International Participation on Forest Soil Science “Diversity of Forest Soils and Biological Diversity of Forests (Pushchino, 2013), pp. 84–86
O. G. Chertov, M. A. Nadporozhskaya, and A. S. Komarov, “Analysis of the dynamics of plant residue mineralization and humification in soil,” Eurasian Soil Sci. 40, 140–148 (2007).
D. S. Orlov, O. N. Biryukova, and N. I. Sukhanova, Organic Matter in the Soils Russia (Nauka, Moscow, 1996) [in Russian].
V. K. Pestryakov, N. V. Kovsh, A. I. Popov, and S. N. Chukov, “Modeling of the organic matter transformation under laboratory conditions,” Pochvovedenie, No. 3, 30–41 (1990).
A. V. Smagin, Gas Phase of Soils (Moscow State Univ., Moscow, 1999) [in Russian].
L. S. Travnikova, Organomineral Interactions: Role in Soil Formation, Fertility, and Tolerance Toward Degradation (Dokuchaev Soil Science Inst., Moscow, 2001) [in Russian].
S. Ya. Trofimov, P. Bottner, and M. M. Coateaux, “Decomposition of organic matter in organic horizons of forest soils in laboratory conditions,” Eurasian Soil Sci. 31, 1349–1357 (1998).
O. G. Chertov and A. S. Komarov, “Theoretical approaches to modeling the dynamics of soil organic matter,” Eurasian Soil Sci. 46, 845–853 (2013).
O. G. Chertov and M. A. Nadporozhskaya, “The effect of intensive nitrogen release during decomposition of the plant remains in laboratory conditions,” Izv. S.-Peterb. Lesotekh. Akad., No. 197, 272–283 (2011).
O. G. Chertov, S. N. Chukov, M. A. Nadporozhskaya, A. I. Popov, N. V. Kovsh, A. V. Belenets, I. N. Lapshina, V. D. Talashkina, and V. D. Davydov, “Analysis of the functional-dynamic characteristics of transformation of soil organic matter,” Vestn. S.-Peterb. Gos. Univ., Ser. 3: Biol., No. 3, 106–110 (1994).
J. Bauer, M. U. F. Kirschbaum, L. Weihermuller, J. A. Huisman, M. Herbst, and H. Vereecken, “Temperature response of wheat decomposition is more complex than the common approaches of most multipool models,” Soil Biol. Biochem. 40, 2780–2786 (2008).
B. Berg, “Decomposition patterns for foliar litter: a theory for influencing factors,” Soil Biol. Biochem. 78, 222–232 (2014).
F. J. Cook and V. A. Orchard, “Relationships between soil respiration and soil moisture,” Soil Biol. Biochem. 40, 1013–1018 (2008).
E. A. Davidson and I. A. Janssens, “Temperature sensitivity of soil carbon decomposition and feedbacks to climate change,” Nature 440, 165–173 (2006). doi 10.1038/nature04514
N. Fierer, J. M. Craine, K. McLauchlan, and J. P. Schimel, “Litter quality and the temperature sensitivity of decomposition,” Ecology 86, 320–326 (2005).
C. Prescott, “Litter decomposition: what controls it and how can we alter it to sequester more carbon in forest soils,” Biogeochemistry 101, 133–149 (2010).
C. M. Preston, J. R. Nault, J. A. Trofymov, and C. Smyth, “Chemical changes during 6 years decomposition of 11 litters in some Canadian forest sites,” Ecosystems 12, 1053–1077 (2009).
M. W. I. Schmidt, M. S. Torn, S. Abiven, T. Dittmar, G. Guggenbereger, I. A. Janssens, M. Kleber, I. Koegel-Knabner, J. Lehmann, D. A. C. Manning, P. Nannipieri, D. Rasse, S. Weiner, and S. Trumbore, “Persistence of soil organic matter as an ecosystem property,” Nature 478, 49–56 (2012).
S. A. Waksman and F. C. Gerretsen, “Influence of temperature and moisture upon the nature and extent of decomposition of plant residues by microorganisms,” Ecology 12 (1), 33–60 (1931).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Larionova, A.N. Maltseva, V.O. Lopes de Gerenyu, A.K. Kvitkina, S.S. Bykhovets, B.N. Zolotareva, V.N. Kudeyarov, 2017, published in Pochvovedenie, 2017, No. 4, pp. 438–448.
Rights and permissions
About this article
Cite this article
Larionova, A.A., Maltseva, A.N., Lopes de Gerenyu, V.O. et al. Effect of temperature and moisture on the mineralization and humification of leaf litter in a model incubation experiment. Eurasian Soil Sc. 50, 422–431 (2017). https://doi.org/10.1134/S1064229317020089
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1064229317020089