Abstract
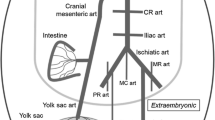
The aim of the study was to determine changes in the blood flow in arterial trunks (coming out of the heart of chickens) by changes of the lumen of these arteries during embryogenesis (on the 10th, 15th, and 19th days) and 6 days after the hatching. For this purpose, posthumous morphometry of aorta, pulmonary arteries, and arterial (Botallo’s) ducts (AD) from their exit from the heart until final extraorgan branching was conducted. It was demonstrated that, in this period, (1) initial lumens of aorta and pulmonary arteries are equal to each other and are equally increased (with temporary stop in last quarter of embryogenesis) with an increase of the body weight (BW); (2) the portion of the right ventricle in a total blood circulation minute volume (BCMV) is somewhat smaller than the portion of the left ventricle, but it approaches equality to it by the end of embryogenesis; (3) with the growth of embryos, the portion of total BCMV flowing through the anterior (before the inflow of AD into the aorta) part of the body decreases (from 41 to 33%); that in the average part increases (from 17 to 31%); that in the posterior part (after bifurcation of aorta), where chorioallantoic membrane (CAM) is located, remains almost unchanged; (4) after the hatching (and disappearance of CAM), BCMV of the left ventricle multiply increases due to the junction of two blood flows from the heart (through the ascending aorta and AD) into a single flow, which flows sequentially by lesser and greater circulations, resulting in multiple increase in the organ blood flow.
Similar content being viewed by others
References
Agren, P., Cogolludo, A.L., Kessels, C.G., Perez-Vizcaιno, F., De Mey, J.G., Blanco, C.E., and Villamor, E., Ontogeny of chicken ductus arteriosus response to oxygen and vasoconstrictors, Am. J. Physiol., 2007, vol. 292, pp. R485–R495.
Angelini, A., Allan, L.D., Anderson, R.H., Crawford, D.C., Chitta, S.K., and Ho, S.Y., Measurements of the dimensions of the aortic and pulmonary pathways in the human fetus: a correlative echocardiographic and morphometric study, Br. Heart J., 1988, vol. 60, pp. 221–226.
Attinger, E.O., Sugavara, H., Navarro, A., Riccetto, A., and Martin, R., Pressure-flow relations in dog arteries, Circ. Res., 1966, vol. 19, no. 2, pp. 230–246.
Belanger, C., Copeland, J., Muirhead, D., Heinz, D., and Dzialowski, E.M., Morphological changes in the chicken ductus arteriosi during closure at hatching, Anat. Rec., 2008, vol. 291, pp. 1007–1015.
Belichenko, V.M. and Shoshenko, K.A., Circulatory system in chicken skeletal muscle in the second half of embryogenesis: shape, blood flow, and vascular reactivity, Russ. J. Dev. Biol., 2009, vol. 40, no. 2, pp. 95–103.
Belichenko, V.M., Aizman, R.I., Khodyrev, E.V., Turganbaeva, A.S., and Shoshenko, K.A., Blood flow in skeletal muscles of chicken in the embryonic and early postembryonic periods, Ross. Fiziol. Zh. im. I.M. Sechenova, 2011, vol. 97, no. 7, pp. 733–743.
Broekhuizen, M.L., Hogers, B., De Ruiter, M.C., Poelmann, R.E., Gittenberger de Groot, A.C., and Wladimiroff, J.W., Altered hemodynamics in chick embryos after extraembryonic venous obstruction, Ultrasound. Obstet. Gynecol., 1999, vol. 13, no. 6, pp. 437–445.
Burggren, W., Khorrami, Sh., Pinder, A., and Sun, T., Body, eye, and chorioallantoic vessel growth are not dependent on cardiac output level in day 3–4 chicken embryos, Am. J. Physiol., 2004, vol. 287, pp. R1399–R1406.
Carlson, B., Patten’s Foundations of Embryology, New York: McGraw-Hill, 1981.
Caro, C.G., Pedley, T.G., Schroter, R.G., and Seed, W.A., The Mechanics of the Circulation, New York: Oxford University Press, 1978.
Chaoui, R., Heling, K.S., Taddei, F., and Bollmann, R., Doppler echocardiographic analysis of blood flow through the fetal aorta and pulmonary valve in the second half of pregnancy, Geburtshilfe Frauenheilkd, 1995, vol. 55, no. 4, pp. 207–217.
Cogolludo, A.L., Moral-Sanz, J., van der Sterren, S., Frazziano, G., van Cleef, A.N., Menendez, C., Zoer, B., Moreno, E., Roman, A., Perez-Vizcaino, F., and Villamor, E., Maturation of O2 sensing and signaling in the chicken ductus arteriosus, Am. J. Physiol., 2009, vol. 297, pp. L619–L630.
Felmeden, D.C., Blann, A.D., and Lip, G.Y.H., Angiogenesis: basic pathophysiology and implications for disease, Eur. Heart J., 2003, vol. 24, pp. 586–603.
Fisinin, V.I., Zhuravlev, I.V., and Aidinyan, T.G., Embrional’noe razvitie ptitsy (Bird Embryonic Development), Moscow: Agropromizdat, 1990.
Gardiner, H.M., Pasquini, L., Wolfenden, J., Barlow, A., Li, W., Kulinskaya, E., and Henein, M., Myocardial tissue Doppler and long axis function in the fetal heart, J. Cardiol., 2006, vol. 113, no. 1, pp. 39–47.
Hu, N. and Clark, E.B., Hemodynamics of the stage 12 to stage 29 chick embryo, Circ. Res., 1989, vol. 65, pp. 1665–1670.
Itskovitz, J., La Gamma, E.F., and Rudolph, A.M., Effects of cord compression on fetal blood flow distribution and O2 delivery, Am. J. Physiol., 1987, vol. 252, no. 1, Pt 2, pp. H100–H109.
Lee, S.H., Hungerford, J.E., Little, C.D., and Iruela-Arispe, M.L., Proliferation and differentiation of smooth muscle cell precursors occurs simultaneously during the development of the vessel wall, Dev. Dyn., 1997, vol. 209, pp. 342–352.
Lingman, G. and Marsál, K., Fetal central blood circulation in the third trimester of normal pregnancy—a longitudinal study. II. Aortic blood velocity waveform, Early Hum. Dev., 1986, vol. 13, no. 2, pp. 151–159.
Lucitti, J.L., Tobita, K., and Keller, B.B., Arterial hemodynamics and mechanical properties after circulatory intervention in the chick embryo, J. Exp. Biol., 2005, vol. 208, pp. 1877–1885.
Mielke, G. and Benda, N., Cardiac output and central distribution of blood flow in the human fetus, Circulation, 2001, vol. 103, pp. 1662–1668.
Mulder, A.L.M., Van Golde, J.C., Prinzen, F.W., and Blanco, C.E., Cardiac output distribution in response to hypoxia in the chick embryo in the second half of the incubation time, J. Physiol., 1998, vol. 508, pp. 281–287.
Mulder, A.L.M., Miedema, A., De Mey, J.G.R., Giussani, D.A., and Blanco, C.E., Sympathetic control of the cardiovascular response to acute hypoxemia in the chick embryo, Am. J. Physiol., 2002, vol. 282, pp. R1156–R1163.
Nathanielsz, P.W. and Hanson, M.A., The fetal dilemma: spare the brain and spoil the liver, J. Physiol., 2003, vol. 548, no. 2, p. 333.
Poutanen, T., Tikanoja, T., Sairanen, H., and Jokinen, E., Normal aortic dimensions and flow in 168 children and young adults, Clin. Physiol. Funct. Imaging, 2003, vol. 23, no. 4, pp. 224–229.
Rudolph, A.M. and Heymann, M.A., Circulatory changes during growth in the fetal lamb, Circ. Res., 1970, vol. 26, pp. 289–299.
Rudolph, A.M. and Heymann, M.A., Fetal and neonatal circulation and respiration, Annu. Rev. Physiol., 1974, vol. 36, pp. 187–207.
Rudolph, A.M., Distribution and regulation of blood flow in the fetal and neonatal lamb, Circ. Res., 1985, vol. 57, pp. 811–821.
Savolainen, S.M., Foley, J.F., and Elmore, A., Histology atlas of the developing mouse heart with emphasis on E11.5 to E18.5, Toxicol. Pathol., 2009, vol. 37, pp. 395–414.
Sedmera, D., Pexieder, T., Vuillemin, M., Thompson, R.P., and Anderson, R.H., Developmental patterning of the myocardium, Anat. Rec., 2000, vol. 258, pp. 319–337.
Shoshenko, K.A., Golub’, A.S., Brod, V.I., Barbashina, N.E., Ivanova, S.F., Krivoshapkin, A.L., and Osipov, V.V., Arkhitektonika krovenosnogo rusla (Vasculature Architectonics), Novosibirsk: Nauka, 1982.
Stevanov, M., Baruthio, J., Musse, O., Gounot, D., and Armspach, J.P., Determination of vessel cross section for flow rate quantification, Magn. Reson. Imaging, 2001, vol. 19, no. 6, pp. 891–897.
Turganbaeva, A.S., Belichenko, V.M., and Shoshenko, K.A., Blood flow in the brain and liver of chicken in embryonal and early postembryonal periods, Ross. Fiziol. Zh. im. I.M. Sechenova, 2011, vol. 97, no. 12, pp. 1361–1373.
Vimpeli, T., Huhtala, H., Wilsgaard, T., and Acharya, G., Fetal aortic isthmus blood flow and the fraction of cardiac output distributed to the upper body and brain at 11–20 weeks of gestation, Ultrasound. Obstet. Gynecol., 2009, vol. 33, no. 5, pp. 538–544.
Vince, M.A., Behaviour and aeration of the respiratory system in the domestic fowl embryo, J. Physiol., 1976, vol. 263, pp. 465–474.
Vroomene, de M., Takahashi, Y., Roman, C., and Heymann, M.A., Calcitonin gene-related peptide increases pulmonary blood flow in fetal sheep, Am. J. Physiol., 1998, vol. 274, no. 2, pp. H277–H282.
Wang, Y., Dur, O., Patrick, M.J., Tinney, J.P., Tobita, K., Keller, B.B., and Pekkan, K., Aortic arch morphogenesis and flow modeling in the chick embryo, Ann. Biomed. Eng., 2009, vol. 37, no. 6, pp. 1069–1081.
White, P.T., Experimental studies on the circulatory system of the late chick embryo, Exp. Biol., 1974, vol. 61, pp. 571–592.
Yang, A. and Siegel, P.B., Late embryonic and early postnatal growth of heart and lung white leghorn chickens, Growth Dev. Aging, 1997, vol. 61, nos. 3–4, pp. 119–126.
Zahka, K.G., Hu, N., Brin, K.P., Yin, F.C.P., and Clark, E.B., Aortic impedance and hydraulic power in the chick embryo from stages 18 to 29, Circ. Res., 1989, vol. 64, pp. 1091–1095.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.M. Belichenko, E.V. Khodyrev, C.A. Shoshenko, 2014, published in Ontogenez, 2014, Vol. 45, No. 2, pp. 89–101.
Rights and permissions
About this article
Cite this article
Belichenko, V.M., Khodyrev, E.V. & Shoshenko, C.A. Aorta, pulmonary artery, and blood flows on them in chickens in the second half of embryogenesis and after hatching. Russ J Dev Biol 45, 66–77 (2014). https://doi.org/10.1134/S1062360414020027
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062360414020027