Abstract
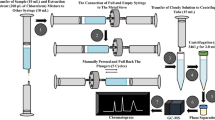
A method for the determination of a mustard gas biomarker (an S-hydroxyethylthioethyl adduct with albumin) in blood plasma was optimized with the use of HPLC with high-resolution tandem mass-spectrometric detection. This method is based on the hydrolysis of this adduct by the proteinase K enzyme with the formation of the following stable tripeptide with cysteine, proline, and phenylalanine: S-[HETE]-Cys- Pro-Phe. The conditions of the sample preparation of human plasma artificially contaminated by mustard gas (the selection of an aliquot portion volume and an enzyme for the hydrolysis), the mass-spectrometric detection (the selection of optimum pairs of ion reactions and high-resolution detection modes), and the gradient elution program in the HPLC separation of an analyzed mixture were optimized. The detection limit of mustard gas in human blood plasma was 1 ng/mL. The approach proposed was tested in the analysis of human blood plasma samples by the standard addition technique and also within the framework of the first official biomedical test carried out by the Organization for the Prohibition of Chemical Weapons (OPCW) in 2016, and it exhibited a good accuracy, reproducibility, and specificity of determination.
Similar content being viewed by others
References
The Convention on the Prohibition of the Development, Production, Stockpiling and Use of Chemical Weapons and on Their Destruction GE 92–61926, Paris, 1993, p. 52.
Capacio, B.R., Smith, J.R., Lawrence, R.J., Boyd, B.L., Witriol, A.M., Conti, M.L., Collins, J.L., and Sciuto, A.M., J. Anal. Toxicol., 2008, vol. 32, p. 37.
Noort, D., Benschop, H.P., and Black, R.M., Toxicol. Appl. Pharm., 2002, vol. 184, p. 116.
Barr, J.R., Pierce, C.L., Smith, J.R., Capacio, B.R., Woolfitt, A.R., Solano, M.I., Wooten, J.V., Lemire, S.W., Thomas, J.D., Ash, D.H., and Ashley, D.L., J. Anal. Toxicol., 2008, vol. 32, p. 106.
Newmark, J., Langer, J.M., Capacio, B., Barr, J., and McIntosh, R.G., Mil. Med., 2007, vol. 172, p. 196.
Benschop, H.P., Van der Schans, G.P., Noort, D., Fidder, A., Mars-Groenendijk, R.H., and de Jong, L.P., J. Anal. Toxicol., 1997, vol. 21, p. 249.
Xu, H., Nie, Z., Zhang, Y., Li, C., Yue, L., Yang, W., Chen, J., Dong, Y., Liu, Q., Lin, Y., Wu, B., Feng, J., Lia, H., Guo, L., and Xie, J., Toxicol. Rep., 2014, vol. 1, p. 533.
Van der Schans, G.P., Mars-Groenendijk, R., de Jong, L.P., Benschop, H.P., and Noort, D., J. Anal. Toxicol., 2004, vol. 28, p. 316.
Noort, D., Hulst, A.G., Trap, H.C., de Jong, L.P., and Benschop, H.P., Arch. Toxico.l, 1997, vol. 71, p. 171.
Black, M.R. and Read, W.R., J. Chromatogr. B: Biomed. Sci. Appl., 1995, vol. 665, p. 95.
Noort, D., Fidder, A., Hulst, A.G., Woolfitt, A.R., Ash, D., and Barr, J.R., J. Anal. Toxicol., 2004, vol. 28, p. 333.
Pantazides, B., Crow, B., Garton, J., Quinones-Gonzales, J., Blake, T., Thomas, J., and Johnson, R.A., Chem. Res. Toxicol, 2015, vol. 28, no. 2, p. 256.
Noort, D., Hulst, A.G., de Jong, L.P., and Benschop, H.P., Chem. Res. Toxicol., 1999, vol. 12, no. 8, p. 715.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Braun, I.V. Rybal’chenko, M.A. Ponsov, Ya.V. Stavitskaya, L.A. Tikhomirov, A.P. Grechukhin, 2017, published in Zhurnal Analiticheskoi Khimii, 2017, Vol. 72, No. 3, pp. 252–258.
Rights and permissions
About this article
Cite this article
Braun, A.V., Rybal’chenko, I.V., Ponsov, M.A. et al. Optimization of a method for the determination of a mustard gas biomarker in human blood plasma by liquid chromatography–high-resolution mass spectrometry. J Anal Chem 72, 303–308 (2017). https://doi.org/10.1134/S1061934817030030
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934817030030