Abstract
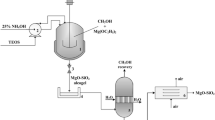
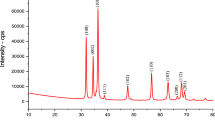
The present work deals with the adsorption of Cu2+ and Pb2+ on zeolites (ZSM-5, mordenite) and mesoporous materials (MCM-48, MCM-41). The characterization of the synthesized samples was performed by means of XRD, SEM, and thermogravimetric analysis. The batch method was employed to study the influence of adsorbent nature, contact time, initial metal ion concentration, and adsorbent load. The adsorption on MCM-48 follows a pseudo-second-order kinetic model. This material was found to be more effective for the removal of lead in a batch process as compared to the other adsorbents and the removal efficiency of the materials for Pb(II) followed the order MCM-48 > mordenite > ZSM-5 > MCM-41 and that for Cu(II) followed the order ZSM-5 > mordenite > MCM-41 > MCM-48.
Similar content being viewed by others
References
E. Alvarez-Ayuso, A. Garcia-Sanchez, and X. Querol, Water Res. 37, 4855 (2003).
V. J. Inglezakis, M. D. Loizidou, and H. P. Grigoropoulou, J. Colloid Interface Sci. 261, 49 (2003).
K. Kalia and S. J. S. Flora, J. Occup. Health 47, 1 (2005).
A. T. Paulino, L. B. Santos, and J. Nozaki, React. Funct. Polym. 68, 634 (2008).
M. Ajmal, A. H. Khan, S. Ahmad, and A. Ahmad, Water Res. 32, 3085 (1998).
R. Gündogan, B. Acemioglu, and M. H. J. Alma, Colloid Interface Sci. 269, 303 (2004).
D. Mohan, C. U. Pittman, and P. H. Steele, J. Colloid Interface Sci. 297, 489 (2006).
A. K. Sengupta and D. Clifford, Environ. Sci. Technol. 20, 149 (1986).
J. Geselbarcht, in Water Reuse Conference Proceedings (AWWA, 1996), p.187.
J. L. Schnoor, Phytoremediation, TE-97-01 (Ground-Water Remediation Technol. Anal.Center, Pittsburgh, 1997).
L. E. Applegate, Chem. Eng. 91, 64 (1984).
H. Yanagisawa, Y. Matsumoto, and M. Machida, Appl. Surf. Sci. 256, 1619 (2010).
S. K. Pitcher, R. C. T. Slade, and N. I. Ward, Sci. Total Environ. 334, 161 (2004).
E. Erdem, N. Karapinar, and R. Donat, J. Colloid Interface Sci. 280, 309 (2004).
C. T. Kresge, M. E. Leonowicz, W. J. Roth, J. C. Vartuli, and J. S. Beck, Nature 359, 710 (1992).
J. S. Beck, J. C. Vartuli, W. J. Roth, M. E. Leonowicz, C. T. Kresge, K. D. Schmitt, C. T. W. Chu, D. H. Olson, E. W. Sheppard, S. B. McCullen, J. B. Higgins, and J. L. Schlenkert, J. Am. Chem. Soc. 114, 10834 (1992).
W. Shengju, Z. Bingru, and L. Fengting, Pet. Process. Sect. 7, 134 (2009).
A. Corma, Chem. Rev. 97, 2373 (1997).
A. Tagushi and F. Schüth, Microporous Mesoporous Mater. 77, 1 (2005).
M. Teymouri, A. Samadi-Maybodi, and A. Vahid, Int. Nano Lett. 1, 34 (2011).
B. Kalita and A. K. Talukdar, Mater. Res. Bull. 44, 254 (2009).
B. O. Hincapie, L. J. Garces, Q. H. Zhang, A. Sacco, and S. L. Suib, Microporous Mesoporous Mater. 67, 19 (2004).
B. Boote, H. Subramanian, and K. T. Ranjit, Chem. Commun. 43, 4543 (2007).
M. W. Wan, C. C. Kan, B. D. Rogel, and M. L. P. Dalida, Carbohydr. Polym. 80, 891 (2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Dutta, D., Roy, S.K., Das, B. et al. Removal of Cu(II) and Pb(II) from Aqueous Solutions Using Nanoporous Materials. Russ. J. Phys. Chem. 92, 976–983 (2018). https://doi.org/10.1134/S0036024418050102
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024418050102