Abstract
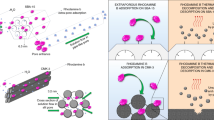
A new mesoporous carbon containing nitrogen and copper was prepared by carbonizing a polymer obtained by blending p-phenylenediamine, copper(II) sulfate, and glyoxal. XRD and XPS showed the existence of Cu metal and Cu2S, and XPS indicated the presence of nitrogen in the mesoporous carbon. The FT-IR spectra suggested that the material contains different functional groups, such as C=C and C=N. N2 adsorption–desorption indicated the presence of mesopores in the material. The mesoporous carbon was used as an adsorbent to investigate the adsorption for methyl orange. Static equilibrium test results showed that the adsorption isotherm for methyl orange is in agreement with the langmuir isotherm, and the Qm is 434 mg g–1. Adsorption kinetics indicated that the pseudo-second order adsorption mechanism is predominant in the adsorption process.
Similar content being viewed by others
References
M. T. Yagub, T. K. Sen, S. Afroze, et al., Adv. Colloid. Interface 209, 172 (2014).
V. K. Gupta, Suhas, J. Environ. Manage. 90, 2313 (2009).
R. Malarvizhi and H. Yuhshan, Desalination 264, 97 (2010).
L. Lin, S. R. Zhai, Z. Y. Xiao, et al., Bioresour. Technol. 136, 437 (2013).
G. C. Jou, Carbon 36, 1643 (1998).
M. H. Do, N. H. Phan, T. D. Nguyen, et al., Chemosphere 85, 1269 (2011).
X. J. Wang, Y. F. Liu, Z. H. Hu, et al., J. Hazard. Mater. 169, 1061 (2009).
Y. Z. Zhang, X. Y. Xiong, Y. Han, et al., Chem. Eng. J. 162, 1045 (2010).
Y. Li, X. Li, J. Li, et al., Water Res. 40, 1119 (2006).
M. Ghaedi, A. M. Ghaedi, A. Ansari, et al., Spectrochim. Acta A 132, 639 (2014).
Y. Yang, W. Guo, Y. Guo, et al., J. Hazard. Mater. 271, 150 (2014).
M. A. Gondal, C. Li, X. Chang, et al., J. Environ. Sci. Heal. A 47, 570 (2012).
C. Zhang, L. Zhou, J. Yang, et al., Environ. Sci. Pollut. R 21, 8398 (2014).
P. Zhang, Q. An, J. Guo, et al., J. Colloid. Interf. Sci. 389, 10 (2013).
Z. Wang, X. Liu, M. Lv, et al., Carbon 48, 3182 (2010).
Y. Tian, X. Wang, and Y. Pan, J. Hazard. Mater. 213–214, 361 (2012).
A. Nezamzadeh-Ejhieh and N. Moazzeni, J. Ind. Eng. Chem. 19, 1433 (2013).
W. U. De-Zhi, X. M. Fan, K. Tian, et al., T. Nonferr. Metal. Soc. 22, 1620 (2012).
J. J. Wang, J. Jiang, B. Hu, et al., Adv. Funct. Mater. 18, 1105 (2008).
P. Bober, J. Stejskal, M. Trchova, et al., J. Polym. Sci. Polym. Chem. 49, 3387 (2011).
J. M. L. Martínez, E. Rodríguez-Castellon, R. M. T. Sánchez, et al., J. Mol. Catal. A: Chem. 339, 43 (2011).
B. Wang, F. Gao, and H. Ma, J. Hazard. Mater. 144, 363 (2007).
L. Hou, L. Lian, D. Li, et al., Carbon 64, 141 (2013).
B. Wang, T. P. Ang, and A. Borgna, Microporous Mesoporous Mater. 158, 99 (2012).
M. Kawaguchi, S. Yagi, and H. Enomoto, Carbon 42, 345 (2004).
A. Vinu, Adv. Funct. Mater. 18, 816 (2008).
N. Mohammadi, H. Khani, V. K. Gupta, et al., J. Colloid. Interf. Sci. 362, 457 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Zhong, L., Li, B., Zhang, L. et al. A Mesoporous Сarbon Prepared by Сarbonizing Cu(II)–p-Phenylenediamine Polymer for the Adsorption of Methyl Orange from Water. Russ. J. Phys. Chem. 92, 785–792 (2018). https://doi.org/10.1134/S0036024418040180
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024418040180