Abstract
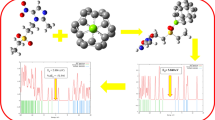
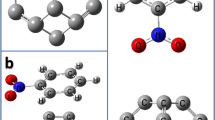
The absorption feasibility of benzene molecule in the C24, Si@C24, Si-doped C24, and C20 fullerenes has been studied based on calculated electronic properties of these fullerenes using Density functional Theory (DFT). It is found that energy of benzene adsorption on C24, Si@C24, and Si-doped C24 fullerenes were in range of –2.93 and –51.19 kJ/mol with little changes in their electronic structure. The results demonstrated that the C24, Si@C24, and Si-doped C24 fullerenes cannot be employed as a chemical adsorbent or sensor for benzene. Silicon doping cannot significantly modify both the electronic properties and benzene adsorption energy of C24 fullerene. On the other hand, C20 fullerene exhibits a high sensitivity, so that the energy gap of the fullerene is changed almost 89.19% after the adsorption process. We concluded that the C20 fullerene can be employed as a reliable material for benzene detection.
Similar content being viewed by others
References
Agency for Toxic Substances and Disease Registry (ATSDR), Toxicological Profile for Benzene (U. S. Public Health Service, U. S. Department of Health and Human Services, Atlanta, GA, 2007).
M. Sittig, Handbook of Toxic and Hazardous Chemicals and Carcinogens, 2nd ed. (Noyes, Park Ridge, NJ, 1985).
U. S. Environmental Protection Agency, Integrated Risk Information System (IRIS) on Benzene (Natl. Center for Environmental Assessment, Office of Research and Development, Washington, DC, 2009).
P. Bechthold, S. Ardenghi, V. Cardoso Schwindt, E. A. González, P. V. Jasen, V. Orazi, M. E. Pronsato, and A. Juan, Appl. Surf. Sci. 282, 17 (2013).
P. V. Jasen, E. A. González, G. Brizuela, and A. Juan, J. Mol. Catal. A: Chem. 323, 23 (2010).
R. Koide, E. J. M. Hensen, J. F. Paul, S. Cristol, E. Payen, H. Nakamura, and R. A. van Santen, Catal. Today 130, 178 (2008).
N. Fernandez, Y. Ferro, Y. Carissan, J. Marchois, and A. Allouche, Phys. Chem. Chem. Phys. 16, 1957 (2014).
H. W. Kroto, J. R. Heath, S. C. O’Brien, R. F. Curl, and R. E. Smalley, Nature (London) 318, 162 (1985).
T. Akasaka, S. Nagase, and A. New, Family of Carbon Clusters (Kluwer Academic, Dordrecht, 2002).
K. Muthukumar and J. A. Larsson, J. Mater. Chem. 18, 3347 (2008).
M. Yoon, S. Yang, and Z. Zhang, J. Chem. Phys. 131, 64707 (2009).
T. W. Chamberlain, N. R. Champness, M. Schröder, and A. N. Khlobystov, Chem.–Eur. J. 17, 668 (2011).
L. Senapati, J. Schrier, and K. B. Whaley, Nano Lett. 4, 2073 (2004).
Xu Liang, Li Chao, Li Feng, Li Xiaojun, and Tao Shuqing, Spectrochim. Acta A 98, 183 (2012).
Wen-Kai Zhao, Chuan-Lu Yang, Jing-Fen Zhao, Mei-Shan Wang, and Xiao-Guang Ma, Physica B 407, 2247 (2012).
H. Prinzbach, A. Weiler, P. Landenberger, F. Wahl, J. Worth, L. T. Scott, M. Gelmont, D. Olevano, and B. Issendorff, Nature 407, 60 (2000).
Y. P. An, C. L. Yang, M. S. Wang, X. G. Ma, and D. H. Wang, J. Clust. Sci. 22, 31 (2011).
Y. P. An, C. L. Yang, M. S. Wang, X. G. Ma, and D. H. Wang, Curr. Appl. Phys. 10, 260 (2010).
C. Tian et al., Chem. Phys. Lett. 511, 393 (2011).
M. T. Baei, Heteroatom. Chem. 24, 516 (2013).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996).
M. Schmidt, K. Baldridge, J. Boatz, S. Elbert, M. Gordon, J. Jensen, S. Koseki, N. Matsunaga, K. A. Nguyen, S. Su, T. L. Windus, M. Dupuis, and J. A. Montgomery, Jr., J. Comput. Chem. 14, 1347 (1993).
E. D. Glendening, A. E. Reed, J. E. Carpenter, and F. Weinhold, NBO Version 3.1 TCI (Univ. of Wisconsin, Madison, 1998).
R. Kumar and A. Rani, Physica B 406, 1173 (2011).
C. Kim, B. Kim, S. M. Lee, C. Jo, and Y. H. Lee, Phys. Rev. B 65, 165418 (2002).
X. Zhou, W. Q. Tian, and X.-L. Wang, Sens. Actuators B: Chem. 151, 56 (2010).
Y. Cui and C. M. Lieber, Science 291, 851 (2001).
S. Li, Semiconductor Physical Electronics, 2nd ed. (Springer, USA, 2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Baei, M.T. Benzene Adsorption on C24, Si@C24, Si-Doped C24, and C20 Fullerenes. Russ. J. Phys. Chem. 91, 2530–2538 (2017). https://doi.org/10.1134/S0036024417130143
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024417130143