Abstract
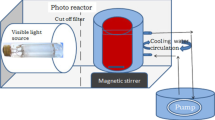
Zinc oxide (ZnO) nanostructured materials have received significant attention because of their unique physicochemical and electronic properties. In particular, the functional properties of ZnO are owed to its morphology and defect structure. ZnO particles were successfully synthesized by chemical precipitation. CTAB (cetyltrimethylammonium bromide), BS-12 (dodecyl dimethyl betaine) and graphene oxide (GO) were selected as templates to induce the formation of ZnO, respectively. By varying the amount of surfactant added during the synthesis process, the structural properties and the crystalline phase of the synthesized nanospheres were characterized by X-ray powder diffraction (XRD), scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FT-IR), ultraviolet and visible spectrophotometry (UV‒Vis). Simultaneously, photo catalytic degradation of Rhodamine B (RhB) was carried out under natural sunlight irradiation while ZnO or ZnO/GO particles were used as catalyst. GO is prone to induce formation of wurtzite hexagonal phase of ZnO. Compared with CTAB and BS-12, ZnO/GO composites had a remarkably photocatalytic degradation.
Similar content being viewed by others
References
R. Shi, P. Yang, J. Wang, A. Zhang, Y. Zhu, et al., CrystEngComm. 14, 5996 (2012).
Z. J. Yu, M. R. Kumar, D. L. Sun, L. T. Wang, et al., Mater. Lett. 166, 284 (2016).
Z. S. Wang, C. H. Huang, Y. Y. Huang, et al., Chem. Mater. 13, 678 (2001).
M. L. Curri, R. Comparelli, et al., Mater. Sci. Eng. C 23, 285 (2003).
R. Wu and C. Xie, Mater. Res. Bull. 39, 637 (2004)
Z. Hu, G. Oskam, and P. C. Searson, J. Colloid Interface Sci. 263, 454 (2003).
Z. W. Pan, Z. R. Dai, and Z. L. Wang, Science 291, 1947 (2001).
J. H. Kim, W. C. Choi, H. Y. Kim, Y. Kang, and Y. K. Park, Powder Technol. 153, 166 (2005).
S. Y. Chu, T. M. Yan, and S. L. Chen, J. Mater. Sci. Lett. 19, 349 (2000).
S. Komarneni, M. Bruno, and E. Mariani, Mater. Res. Bull. 35, 1843, (2000).
T. Tani, L. Madler, and S. E. Pratsinis, J. Nanopart. Res. 4, 337 (2002).
Z. R. Dai, Z. W. Pan, and Z. L. Wang, Adv. Function. Mater. 13, 9 (2003).
W. Ao, J. Li, H. Yang, X. Zeng, and X. Ma, Powder Technol. 168, 148 (2006).
K. Choi, T. Kang, and S. G. Oh, Mater. Lett. 75, 240 (2012).
T. Thilagavathi and D. Geetha, Appl. Nanosci. 4, 127 (2014).
Y. Tan, E. M. Steinmiller, and K. S. Choi, Langmuir 21, 9618 (2005).
D. Chandra, S. Mridha, D. Basak, and A. Bhaumik, Chem. Commun. 17, 2384 (2009).
S. H. Kim, T. Y. Olson, et al., Microporous Mesoporous Mater. 151, 64 (2012).
X. Pan, M. Q. Yang, and Y. J. Xu, Phys. Chem. Chem. Phys. 16, 5589 (2014).
Y. Zhang, Z. R. Tang, X. Fu, and Y. J. Xu, ACS Nano 5, 7426 (2011).
H. Moussa, E. Girot, K. Mozet, et al., Appl. Catal. B: Environ. 185, 11 (2016).
O. Akhavan, Carbon 49, 11 (2011).
T. Xu, L. Zhang, H. Cheng, and Y. Zhu, Appl. Catal. B: Environ. 101, 382 (2011).
J. Wang, T. Tsuzuki, B. Tang, et al., ACS Appl. Mater. Interfaces 4, 3084 (2012).
Y. Wang, F. Wang, and J. He, Nanoscale 5, 11291 (2013).
Y. Cheng, Y. Fan, Y. Pei, and M. Qiao, Catal. Sci. Technol. 5, 3903 (2015).
C. Yao, A. Xie, Y. Shen, et al., Cryst. Res. Technol. 49, 982 (2014).
H. Zhang, A. Xie, Y. Shen, et al., Phys. Chem. Chem. Phys. 14, 12757 (2012).
S. Deng, V. Tjoa, et al., J. Am. Chem. Soc. 134, 4905 (2012).
S. Chen, J. Zhu, X. Wu, Q. Han, and X. Wang, ACS Nano 4, 2822 (2010).
Q. Yu, W. Fu, C. Yu, H. Yang, et al., J. Phys. Chem. C 111, 17521 (2007).
X. Pan, M. Q. Yang, and Y. J. Xu, Phys. Chem. Chem. Phys. 16, 5589 (2014).
Y. Liang, D. Wu, X. Feng, and K. Müllen, Adv. Mater. 21, 1679 (2009).
L. Kashinath, K. Namratha, and K. Byrappa, Mater. Today: Proc. 3, 74 (2016).
K. Kaviyarasu and P. A. Devarajan, Adv. Mater. Lett. 4, 582 (2013).
Y. Gong, X. Meng, C. Zou, Y. Yao, et al., Mater. Lett. 106, 171 (2013).
T. Xu, L. Zhang, H. Cheng, and Y. Zhu, Appl. Catal. B: Environ. 101, 382 (2011)
Q. P. Luo, X. Y. Yu, B. X. Lei, et al., J. Phys. Chem. C 116, 8111 (2012).
A. R. Marlinda, N. M. Huang, M. R. Muhamad, et al., Mater. Lett. 80, 9 (2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Yao, C., Zhu, J., Li, H. et al. Synthesis of ZnO Photocatalysts Using Various Surfactants. Russ. J. Phys. Chem. 91, 2680–2685 (2017). https://doi.org/10.1134/S0036024417130088
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024417130088