Abstract
The translation method for predicting and constructing equilibrium phase diagrams of multicomponent systems is developed based on the compatibility of structural elements of partial constituent (n-component) systems and the general (n + 1)-component system with account for the requirements of the Gibbs phase rule. The invariant equilibrium states in the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°C are studied. This multicomponent system features the options of invariant equilibria where one and the same composition of equilibrium solid phases can be in equilibrium with saturated solutions of differing concentrations. Therefore, this invariant equilibrium is to be reflected in the diagram as a geometric (invariant) image that has a dimension (it may be conventionally referred to as a quasi-point) and not by a point. The dimension of this image is determined by the concentration bounds of components in the equilibrium saturated solution.
Similar content being viewed by others
INTRODUCTION
Kurnakov, the founder of the theory and practice of physicochemical analysis, recognized two independent parts in phase diagrams of chemical systems: a phase complex and coordinate axes [1, 2]. The phase complex as an independent part of the chemical diagram that comprises geometric images (fields, curves, and points), which are located in the diagram in accordance with the equilibrium concentrations of components (constituents) in the liquid phase of the system. In accordance with the Gibbs phase rule [3], the position of an invariant point in the diagram is to reflect the state of the system where a saturated solution is equilibrated with the greatest number of solid phases allowed by the complexity of the system.
SUBJECTS AND METHODS
The options of invariant point generation are intimately related to the complexity of the system. The more components are in the system, the greater the number of possible routes (options) for the formation of invariant points. According to theory of physicochemical analysis [3], whatever invariant point formation options, the equilibrium concentration of the liquid phase for this point is to be constant. According to [4], however, there are numerous cases where equilibrium saturated solutions with different contents of the constituents (components) are found for one and the same phase composition of the precipitates that correspond to invariant points. In the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°С, for example, two different compositions of an equilibrium liquid phase were found for one invariant point (Table 1, borrowed from [4]).
From Table 1 one can see that liquid phases with differing salt contents are characteristic of one and the same composition of equilibrium solid phases. Therefore, the four equilibrium solid phases that correspond to this invariant point can jointly crystallize not at a definite (constant) concentration of the liquid phase equilibrated with them, but over a definite range of concentrations of its salts. This means that the invariant point has a certain “dimension” in terms of salt concentration in the equilibrium liquid phase. If we agree with this statement, then, in our opinion, this may be related to the routes (options) to acquire this equilibrium state.
The above speculations are validated by the results of predictions of phase equilibria (phase complexes) for multicomponent systems by the translation method [5]. The underlying idea of the translation method is the compatibility of the structural elements of particular (e.g., n-component) systems with the structural elements of a general (e.g., an (n + 1)-component) system in one diagram [6, 7], and this is one of the most universal investigation tools for multicomponent systems [8].
The practicability of this method for predicting the structure of multicomponent systems flows from Kurnakov’s statement that “…any diagram of a multicomponent system can be considered to arise from the diagram of a system with fewer components, complicated by the introduction of new components or other equilibrium conditions, and the characteristic elements of the simpler diagram do not disappear, but only take another geometric image” [1, 2].
According to the translation method, the geometric images are transformed upon the transition from an n-component to the (n + 1)-component state in accordance with their topological properties and obeying the Gibbs phase rule, intersecting with each other to form geometric images (fields, curves, and points) of the system at the (n + 1)-component level. Experience of using the translation method for predicting and plotting equilibrium phase diagrams of multicomponent (five- and six-component) systems points to various options to form their geometric images [9–16]. Attempts have been made [17–24] at predicting possible combinations of solid phases at invariant points of the system Na,K,Mg,Ca||SO4,Cl–H2O at 25°C via Gibbs energy minimization. However, the results did not make it possible to construct equilibrium phase diagrams. In addition, the authors themselves recognized that the diagrams became progressively less reliable as the complexity of the system increased. In this paper, we focus on the options of the generation of invariant points in the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°C elucidated by the translation method.
The solubility method [4] for the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°С elucidated three invariant points with the following equilibrium solid phases: (1) Ha + Sy + Cn + Са · 6, (2) Ha + Cn + Th + Bi, and (3) Ha + Cn + Th + Са · 6 (the notations of equilibrium solid phases appear under Table 1). For invariant point 1, two saturated solution compositions were found experimentally; for each of the other two points, one composition was found. The data gained by the translation method prove the occurrence of three invariant points in the system and, at the same time, explain the different compositions of the saturated solution for one and the same invariant point. This fact can be validated by analysis of formation routes of invariant points of the system NaCl–KCl–MgCl2–CaCl2–H2O at 25°C.
RESULTS AND DISCUSSION
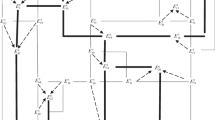
The five-component system NaCl–KCl–MgCl2–CaCl2–H2O comprises four four-component systems; the invariant points featured by these systems at 25°C, with their relevant equilibrium solid phases, are listed in Table 2. Figure 1 shows a schematic diagram [25] of the phase complex of the system NaCl–KCl–MgCl2–CaCl2–H2O at 25°C at the four-component level, constructed using the data of Table 2.
The translation of quaternary invariant points to the five-component level in the form of monovariant curves and their intersections in compliance with the Gibbs phase rule is accompanied with the formation of the following quinary invariant points:
\(E_{1}^{4}\) + \(E_{4}^{4}\) |
| \(E_{1}^{5}\)= Bi + Ha + Cn + Th; |
\(E_{1}^{4}\) + \(E_{8}^{4}\) |
| \(E_{2}^{5}\)= Bi + Ha + Cn + Th; |
\(E_{4}^{4}\) + \(E_{8}^{4}\) |
| \(E_{3}^{5}\) = Bi + Ha + Cn + Th; |
\(E_{2}^{4}\) + \(E_{3}^{4}\) |
| \(E_{4}^{5}\)= Ha + Cn + Sy + Са · 6; |
\(E_{2}^{4}\) + \(E_{6}^{4}\) |
| \(E_{5}^{5}\)= Ha + Cn + Sy + Са · 6; |
\(E_{3}^{4}\) + \(E_{6}^{4}\) |
| \(E_{6}^{5}\)= Ha + Cn + Sy + Са · 6; |
\(E_{5}^{4}\) + \(E_{7}^{4}\) |
| \(E_{7}^{5}\)= Ha + Cn + Th + Са · 6. |
Of the seven appearing quinary invariant points, \(E_{1}^{5},\)\(E_{2}^{5}\), and \(E_{3}^{5},\) as well as \(E_{4}^{5},\)\(E_{5}^{5}\), and \(E_{6}^{5}\), have identical equilibrium solid phases. This means that they are generated as a result of the trilateral intersection of the monovariant curves that appeared upon the translation of quaternary invariant points. Therefore, the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°C features three, and not seven, quinary invariant points, which are generated in the following way:
\(E_{1}^{4}\)+\(E_{4}^{4}\) + \(E_{8}^{4}\) |
| \(E_{1}^{5}\)= Bi + Ha + Cn + Th; |
\(E_{2}^{4}\)+\(E_{3}^{4}\) + \(E_{6}^{4}\) |
| \(E_{2}^{5}\) = Ha + Cn + Sy + Са · 6; |
\(E_{5}^{4}\) + \(E_{7}^{4}\) |
| \(E_{3}^{5}\) = Ha + Cn + Th + Са · 6. |
Figure 2 shows the schematic diagram [25] of the phase complex of the system NaCl–KCl–MgCl2–CaCl2–H2O at 25°С constructed by the translation method using these data.
At the invariant points found by the translation method, this equilibrium can be experimentally implemented by more than one route. For example, equilibrium at invariant point \(E_{3}^{5}\) can be acquired via addition of halite (NaCl) to a solution saturated with the solid phases Cn + Th + Са ⋅ 6 (\(E_{7}^{4}\)), or via addition of carnallite (KCl ⋅ MgCl2 ⋅ 6H2O) to a solution saturated with the solid phases Ha + Th + Са · 6 (\(E_{5}^{4}\)), i.e., by the additional saturation method [26]. The translation method also points to a third option to acquire equilibrium, namely, via mixing a saturated solution with the solid phases that correspond to point \(E_{5}^{4},\) with the respective solution for point \(E_{7}^{4}\), followed by bringing the thus-prepared mixture to equilibrium at 25°С. This option requires less time and material costs.
At the other quinary invariant points generated by the triple intersection of monovariant curves, there are far more equilibrium acquisition options: they each have three options for an additional saturation of the initial mixtures of quaternary invariant points by the following solid phase and one option associated with mixing the initial saturated solutions of all the three quaternary invariant points with their intrinsic equilibrium solid phases and the equilibration of the thus-prepared mixture.
Based on the above, we can assume that the different compositions of the saturated solution at the invariant point of a multicomponent system with the same composition of equilibrium solid phases (Table 1), are due to different equilibrium acquisition routes. Figure 3 shows the formation of quinary invariant points \(E_{1}^{5}\) and \(E_{2}^{5}\) of the system NaCl–KCl–MgCl2–CaCl2–H2O at 25°С, where the arrowed solid lines denote bilateral intersections and the arrowed dashed lines denote trilateral intersections of monovariant curves to generate invariant points \(E_{1}^{5}\) and \(E_{2}^{5}.\) The equilibrium solid phases intrinsic to the invariant points indicated in Fig. 3 are found in the above text.
The foregoing suggests that the diagram of a multicomponent system can image invariant equilibrium not only as a dimensionless point, but also as a geometric (invariant) image that has a certain dimension, the dimension being determined by different compositions of the equilibrium liquid phase intrinsic to one and the same composition of equilibrium solid phases. The variation in the composition of the equilibrium liquid phase can arise from different formation routes (options) of this geometric (invariant) image. Support to this suggestion comes not only from the above-considered example of solubility in the five-component system NaCl–KCl–MgCl2–CaCl2–H2O at 25°С, but also from studies of other multicomponent systems. For the five-component system Na,K,Mg||SO4,Cl–H2O at 0 and 50°С, for example, it was found experimentally [4] that the equilibrium liquid phase can have different compositions for the same composition of equilibrium solid phases (Table 3). The different chemical compositions of the saturated solution in the system Na,K,Mg||SO4,Cl–H2O at 0 and 50°C (Table 3) for the same composition of equilibrium solid phases that correspond to invariant points, can evidently be explained by the order of consecutive addition of certain salts to the initial mixture, and this is consistent with the different options for the translation of quaternary invariant points to the five-component level.
CONSLUSIONS
We have shown that it is in principle possible for the invariant points of equilibrium liquid phases having different chemical compositions to correspond to one and the same composition of equilibrium solid phases, given that the system is multicomponent. This may be substantiated by the variety of routes to acquire invariant equilibrium. This conclusion is consistent with the major principles of physicochemical analysis and the Gibbs phase rule as shown by the translation method for predicting and constructing phase diagrams of five- and six-component systems. Therefore, invariant equilibrium can be reflected in the diagram of a multicomponent system not only as a point (where there is only one equilibrium acquisition option), but also as a geometric (invariant) image (it may be conventionally referred to as a quasi-point), this image having a certain dimension (where there is more than one equilibrium acquisition version).
REFERENCES
N. S. Kurnakov, Dokl. Akad. Nauk SSSR 25, 384 (1939).
N. S. Kurnakov, Introduction to Physicochemical Analysis (Izd–vo Akad. Nauk SSSR, Moscow, 1940) [in Russian].
V. Ya. Anosov, M. I. Ozerova, and Yu. Ya. Fialkov, Major Methods of Physicochemical Analysis (Nauka, Moscow, 1976) [in Russian].
Experimental Solubility Data for Multinary Water–Salt Systems. Handbook (Khimizdat, St. Petersburg, 2003), Vol. 2, Books 1 and 2) [in Russian].
L. Soliev, Available from VINITI no. 8990 (1987).
Ya. G. Goroshchenko, Physicochemical Analysis of Homogeneous and Heterogeneous Systems (Naukova Dumka, Kiev, 1978) [in Russian].
A. G. Goroshchenko, The Center-of-Mass Method for Imaging Multicomponent Systems (Naukova Dumka, Kiev, 1982) [in Russian].
Ya. G. Goroshchenko and L. Soliev, Zh. Neorg. Khim. 32, 1676 (1987).
L. Soliev and M. Usmonov, Russ. J. Inorg. Chem. 57, 452 (2012).
L. Soliev, Russ. J. Inorg. Chem. 58, 585 (2013).
L. Soliev, Russ. J. Phys. Chem. 87, 1442 (2013).
L. Soliev, Russ. J. Inorg. Chem. 59, 1030 (2014).
L. Soliev, Russ. J. Inorg. Chem. 60, 1008 (2015).
L. Soliev, Dokl. Akad. Nauk Resp. Tajikistan 58 (1), 57 (2015).
L. Soliev, I. M. Nizomov, M. T. Dzhumaev, and I. Gulom, Vestn. Tadzh. Nats. Univ., Ser. Est. Nauk, No. 1/1 (156), 131 (2015).
Sh. Tursunbadalov and L. Soliev, J. Solution Chem. 44, 1626 (2015).
K. S. Pitser, J. Phys. Chem. 77, 268 (1973).
K. S. Pitser and G. Mayarga, J. Phys. Chem. 77, 2300 (1973).
K. S. Pitser and G. Mayarga, J. Solution Chem. 3, 359 (1974).
K. S. Pitser and J. Kim, J. Am. Chem. Soc. 96, 5701 (1974).
C. F. Harviec and J. H. Weare, Geochem. Cosmochim. Acta 44, 981 (1980).
J. R. Wood, Geochem. Cosmochim. Acta 39, 1147 (1975).
H. P. Eugster, C. F. Harvie, and J. H. Weare, Geochem. Cosmochim. Acta 44, 1335 (1980).
C. F. Harvie, H. P. Eugster, and J. H. Weare, Geochem. Cosmochim. Acta 46, 1603 (1982).
L. Soliev, Zh. Neorg. Khim. 33, 1305 (1988).
Ya. G. Goroshchenko, L. Soliev, and Yu. I. Gornikov, Ukr. Khim. Zh. 43, 1277 (1977).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Soliev, L. Invariant Equilibria in Multicomponent Systems. Russ. J. Inorg. Chem. 64, 894–898 (2019). https://doi.org/10.1134/S0036023619070167
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619070167