Abstract
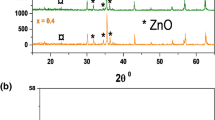
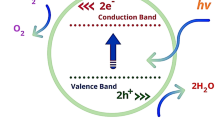
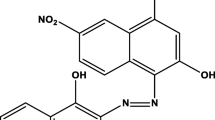
Composite ferrite nanoparticles Co1 – xZnxFe2O4/Ni1 – xZnxFe2O4, x = 0.1–0.5) have been synthesized by co-precipitation method and annealed at temperature of 800°C for 2 h in air. The synthesized samples have been characterized by X-ray powder diffraction, FE-SEM/EDS, and UV-Vis spectroscopy. The prepared nanoparticles exhibit a cubic crystal structure observed from X-ray powder diffraction experiment. It has been observed that the Co0.7Zn0.3Fe2O4/Ni0.7Zn0.3Fe2O4 nanoparticles exhibit higher optical absorbance spectrum at 400 to 800 nm wavelength due to its smaller crystal size (100.8 nm) as compared to the composite ferrite nanoparticles Co0.9Zn0.1Fe2O4/Ni0.9Zn0.1Fe2O4 (176.2 nm), Co0.8Zn0.2Fe2O4/Ni0.8Zn0.2Fe2O4 (134.3 nm), Co0.6Zn0.4Fe2O4/Ni0.6Zn0.4Fe2O4 (165.6 nm), and Co0.5Zn0.5Fe2O4/Ni0.5Zn0.5Fe2O4 (245.6 nm) nanoparticles. The photocatalytic activity of composite ferrite nanoparticles have been studied by performing the decomposition of methylene blue dye solution under UV light irradiation within 0 to 4 h. The methylene blue dye solution was considerably photodegraded by Co0.7Zn0.3Fe2O4/Ni0.7Zn0.3Fe2O4 photocatalyst under UV irradiation within 0–4 h to the efficiency of 96%. The pseudo first order rate constant of the degradation has been found to be 0.0144 S–1. The degradation mechanisms are discussed.

Similar content being viewed by others
REFERENCES
A. Masakazu, T. Masato, I. Keita, et al., in Semiconductor Photochemistry and Photophysics (Marcel Dekker, 2003).
A. Masakazu, T.Masato, I. Keita, et al., Curr. Opin. Solid State Mater. Sci. 6, 381 (2002). https://doi.org/10.1016/S1359-0286(02)00107-9
H. R. Pant, C.H. Park, B. Pant, et al., Ceram. Int. 38, 2943 (2012). https://doi.org/10.1016/j.ceramint.2011.11.071
S. B. Sun, X. T. Chang, X. J. Li, and Z. J. Li, Ceram. Int. 39, 5197 (2013). https://doi.org/10.1016/j.ceramint.2012.12.018
M. H. Habibi and M.H. Rahmati, Spectrochim. Acta A 133, 13 (2014). https://doi.org/10.1016/j.saa.2014.04.110
L. Zhong and F. Haghighat, Build. Environ. 91, 191 (2015). doi.org/https://doi.org/10.1016/j.buildenv.2015.01.033
Ni. Meng, K. H. L. Michael, Y. C. L. Dennis, and K. Sumath, Renew. Sust. Energy Rev. 11, 401 (2007) https://doi.org/10.1016/j.rser.2005.01.009
H. M. Coleman, K. Chiang, and R. Amal, Chem. Eng. J. 113, 65 (2005).https://doi.org/10.1016/j.cej.2005.07.014
C. H. Wu, Chemosphere 57, 601 (2004) https://doi.org/10.1016/j.chemosphere.2004.07.008
D. L. Liao, C. A. Badour, and B. Q. Liao, J. Photochem. Photobiol. 194, 11 (2008). https://doi.org/10.1016/j.jphotochem.2007.07.008
N. Dehghan-Niarostami, F. Taleshi, A. Pahlavan, et al., Int. Nano Lett. 4, 121 (2014). https://doi.org/10.1007/s40089-014-0121-8
C. Borgohain, K. K. Senapati, K. C. Sarma, and P. Phukan, J. Mol. Catal. A: Chem. 363–364, 495 (2012). https://doi.org/10.1016/j.molcata.2012.07.032
C. Singh, S. Jauhar, V. Kumar, et al., Mater. Chem. Phys. 156, 188 (2015). https://doi.org/10.1016/j.matchemphys.2015.02.046
G. Fan, J. Tong, and F. Li, Ind. Eng. Chem. Res. 51, 13639 (2012). https://doi.org/10.1021/ie201933g
R. Sharma, S. Bansal, and S. Singhal, RSC Adv. 8, 1 (2015). doi:
A. Afkhami, S. Sayari, R. Moosavi, and T. Madrakian, J. Indust. Engin. Chem. 21, 920 (2015). https://doi.org/10.1016/j.jiec.2014.04.033
P. Xiong, Y. Fua, L. Wang, and X. Wang, Chem. Engin. J. 196–196, 149 (2012). https://doi.org/10.1016/j.cej.2012.05.007
J. Chen, T. Chen, Li. W. Lai, et al., Materials 8, 4273 (2015). https://doi.org/10.3390/ma8074273
S. T. Assar, H. F. Abosheiasha, and M. K. El Nimr, J. Magn. Magn.Mater. 354, 136 (2014). https://doi.org/10.1016/j.jmmm.2013.10.022
S. F. Mansour and M. A. Elkestawy, Ceram. Int. 37, 1175 (2011). doi.org/https://doi.org/10.1016/j.ceramint.2010.11.038
H. Zhu, S. Zhang, Y. X. Huang, et al., Nano Lett. 13, 2947 (2013). https://doi.org/10.1021/nl4013248
J. Joseph, R. B. Tangsali, V. P. Mahadevan Pillai, et al., Mater. Res. Bull. 61, 475 (2014). https://doi.org/10.1016/j.materresbull.2014.10.061
S. A. Morrison, C. L. Cahill, S. Calvin, et al., J. Appl. Phys. 95, 6392 (2004). https://doi.org/10.1063/1.1715132
H. Malika, A. Mahmood, K. Mahmood, et al., Ceram. Int. 40, 9439 (2014). https://doi.org/10.1016/j.ceramint.2014.02.015
I. Sharifia, H. Shokrollahia, M. M. Doroodmand, and R. Safia, J. Magn. Magn. Mater. 324, 1854 (2012). https://doi.org/10.1016/j.jmmm.2012.01.015
D. Zou, D. Yan, L. Xiao, and Y. Dong, Surf. Coat. Technol. 202, 1928 (2008). https://doi.org/10.1016/j.surfcoat.2007.08.022
C. Singh, S. Jauhar, V. Kumar, et al., Mater. Chem. Phys. 156, 188 (2015). https://doi.org/10.1016/j.matchemphys.2015.02.046
M. S. Anwar, F. Ahmed, and B. H. Koo, Acta Mater. 71, 100(2014). https://doi.org/10.1016/j.actamat.2014.03.002
M. H. Habibi and J. Parhizkar, Spectrochim. Acta Part A: Mol. Biomol. Spect. 150, 879 (2015). https://doi.org/10.1016/j.saa.2015.06.040
H. Zhang, G. Chen, and D. W. Bahnemann, J. Mater. Chem. 19, 5089 (2009). https://doi.org/10.1039/B821991E
A. S. Ahmed, M. M. Shafeeq, M. L. Singla, et al., J. Lumin. 131, 1 (2011). https://doi.org/10.1016/j.jlumin.2010.07.017
J. Tauc, R. Grigorovici, and A. Vancu, Phys. Status Solidi 15, 627 (1966). https://doi.org/10.1002/pssb.19660150224
S. Valencia, J. M Marin, and G. Restrepo, Open Mater. Sci. J. 4, 9 (2009). https://doi.org/10.2174/1874088X01004010009
R. Elilarassi and G. Chandrasekaran, J Mater Sci. Mater. Electron. 21, 1168 (2010). https://doi.org/10.1007/s10854-009-0041-y
O. Yayapao, T. Thongtem, A. Phuruangrat, and S. Thongtem, J. Alloys Compd. 576, 72 (2013). https://doi.org/10.1016/j.jallcom.2013.04.133
Author information
Authors and Affiliations
Corresponding author
SUPPLEMENTARY MATERIALS
Rights and permissions
About this article
Cite this article
Rahman, A., Jayaganthan, R. Photocatalytic Studies of Composite Ferrite Nanoparticles. Russ. J. Inorg. Chem. 64, 946–954 (2019). https://doi.org/10.1134/S0036023619070131
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619070131