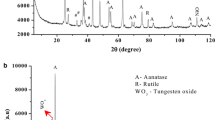
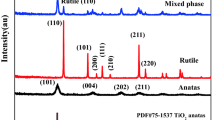
Abstract
Anatase titania based TiO2/С nanocomposites have been synthesized via the thermal decomposition of titanium glycerolate prepared by the reaction between glycerin and tetraethoxytitanium or tetrabutoxytitanium. The content of carbon in the composite material is from 22.3 to 23.5 wt %. The principal physicochemical characteristics of the synthesized compounds have been determined by X-ray diffraction and thermogravimetric analysis, IR and Raman spectroscopy, and scanning electron microscopy (SEM). It has been found that the textural and morphological features of the TiO2/C nanocomposites are governed by the type of titanium alkoxide used to synthesize the precursor, i.e., titanium glycerolate.






Similar content being viewed by others
REFERENCES
M. J. Gratzel, Photochem. Photobiol. 4 (2), 145 (2003). https://doi.org/10.1016/S1389-5567(03)00026-1
J. A. Byrne, A. Davidson, P. S. M. Dunlop, et al., J. Photochem. Photobiol. A 148, 365 (2002).
M. Abdullah and S. K. Kamarudin, Renew. Sust. En. Rev. 76, 212 (2017).
Y.-S. Hu, L. Kienle, Y. -G. Guo, et al., Adv. Mater. 18, 1421 (2006). https://doi.org/10.1002/adma.200502723
X. Chen and S. S. Mao, Chem. Rev. 107, 2891 (2007). https://doi.org/10.1021/cr0500535
H. G. Yang and H. C. Zeng, J. Phys. Chem. B 108, 3492 (2004). https://doi.org/10.1021/jp0377782
L. Chen, J. Tian, H. Qiu, et al., Ceram. Int. 35, 3275 (2009). https://doi.org/10.1016/j.ceramint.2009.05.021
P. Roy, S. Berger, and P. Schmuki, Angew. Chem., Int. Ed. Engl. 50, 2904 (2011). https://doi.org/10.1002/anie.201001374
D. P. Cozzoli, A. Kornowski, and H. Weller, J. Am. Chem. Soc. 125, 14 539 (2003). https://doi.org/10.1021/ja036505h
D. C. Valentin, E. Finazzi, and G. Pacchioni, Chem. Phys. 339, 44 (2007). https://doi.org/10.1016/j.chemphys.2007.07.020
T. Ohno, T. Mitsui, and M. Matsumura, Chem. Lett. 32, 364 (2003). https://doi.org/10.1246/cl.2003.364
J. C. Yu, J. Yu, W. Ho, et al., Chem. Mater. 14, 3808 (2002). https://doi.org/10.1021/cm020027c
X. Shi, Z. Zhang, K. Du, et al., J. Power Sources 330, 1 (2016). https://doi.org/10.1016/j.jpowsour.2016.08.132
Y. Guo, D. He, S. Xia, et al., J. Nanomater 2012, 1 (2012). https://doi.org/10.1155/2012/202794
M.-P. Zheng, Y.-P. Jin, G.-L. Jin, et al., J. Mater. Sci. Lett. 19, 433 (2000). https://doi.org/10.1023/a:1006703224379
C.-Y. Yen, Y.-F. Lin, C.-H. Hung, et al., Nanotechnol. 19, 045 604 (2008). https://doi.org/10.1088/0957-4484/19/04/045604
Q. Huang, S. Tian, D. Zeng, et al., ACS Catal. 3, 1477 (2013). https://doi.org/10.1021/cs400080w
M.-Q. Yang, N. Zhang, and Y.-J. Xu, ACS Appl. Mater. Int. 5, 1156 (2013). https://doi.org/10.1021/am3029798
L. Zeng, C. Zheng, L. Xia, et al., J. Mater. Chem. 1, 4293 (2013). https://doi.org/10.1039/c3ta10275k
F. J. Maldonado-Hodar, C. Moreno-Castilla, and J. Rivera-Utrilla, Appl. Catal. A 203, 151 (2000). https://doi.org/10.1016/s0926-860x(00)00480-4
Y. Gea, J. Zhu, Y. Lub, et al., Electrochim. Acta 176, 989 (2015).
L. Xiong, Y. Xu, P. Lei, et al., Solid State Ionics 268, 265 (2014). https://doi.org/10.1016/j.ssi.2014.08.009
J. Zhao, Y. Liu, M. Fan, et al., Inorg. Chem. Frontiers 2, 198 (2015). https://doi.org/10.1039/c4qi00191e
Y. Wei, J. Zhu, Y. Gan, et al., Adv. Powder Technol. 29, 2289 (2018). https://doi.org/10.1016/j.apt.2018.05.016
X. Jiang, Y. Wang, T. Herricks, et al., J. Mater. Chem. 14, 695 (2004). https://doi.org/10.1039/b313938g
J. Jiang, F. Gu, W. Shao, et al., Ind. Eng. Chem. Res. 51, 2838 (2012). https://doi.org/10.1021/ie202049j
G. S. Zakharova, E. I. Andreikov, V. A. Osipova, et al., Inorg. Mater. 49, 1216 (2013). https://doi.org/10.1134/S0020168513100154
H.-B. Kim and D.-J. Jang, Cryst. Eng. Commun. 17, 3325 (2015). https://doi.org/10.1039/c5ce00257e
G. S. Zakharova, A. Ottmann, L. Moller, et al., J. Mater. Sci. 53, 12 244 (2018). https://doi.org/10.1007/s10853-018-2488-9
J. Das, F. S. Freitas, I. R. Evans, et al., J. Mater. Chem. 20, 4425 (2010). https://doi.org/10.1039/b921373b
M. Velasco, F. Rubio, J. Rubio, and J. L. Oteo, Thermochim. Acta 326, 91 (1999). https://doi.org/10.1016/j.supflu.2018.04.017
S. Doeuff, M. Henry, C. Sanchez, et al., J. Non-Cryst. Solids 89, 206 (1987).
D. A. Kazakov, A. V. Portnova, S. A. Onorin, et al., Izv. Vyssh. Uchebn. Zaved., Khim. Tekhnol. 54 (6), 39 (2011).
G. M. Kuz’micheva, E. V. Savinkina, L. N. Obolenskaya, et al., Crystallogr. Repts. 55, 866 (2010). https://doi.org/10.1134/S1063774510050287
T. Ohsaka, F. Izumi, and Y. Fujiki, J. Raman Spectrosc. 7, 321 (1978). https://doi.org/10.1002/jrs.1250070606
W. F. Zhang, Y. L. He, M. S. Zhang, et al., J. Phys. D: Appl. Phys. 33, 912 (2000). https://doi.org/10.1088/0022-3727/33/8/305
A. C. Ferrari and J. Robertson, Phys. Rev. B 61, 14 095 (2000). https://doi.org/10.1103/physrevb.61.14095
R. Liu, W. Guo, B. Sun, et al., Electrochim. Acta 156, 274 (2015). https://doi.org/10.1016/j.electacta.2015.01.012
K. S. W. Sing, D. H. Everett, R. A. W. Haul, et al., Pure Appl. Chem. 57, 603 (1985). https://doi.org/10.1351/pac198557040603
ACKNOWLEDGMENTS
We are grateful to the TESCAN Company for help in SEM studies of the samples.
Funding
This work was performed in compliance with the state task to the Institute of Solid State Chemistry (Ural Branch, Russian Academy of Sciences) (project no. AAAA-16-116122810209-5).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Glushachenkova
Rights and permissions
About this article
Cite this article
Fattakhova, Z.A., Zakharova, G.S., Andreikov, E.I. et al. A New Method for the Synthesis of TiO2/C Composites with the Use of Titanium Organic Compounds. Russ. J. Inorg. Chem. 64, 857–863 (2019). https://doi.org/10.1134/S0036023619070076
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619070076