Abstract
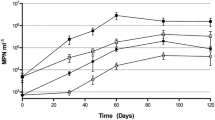
Diversity of the oil-degrading microbial strains isolated from the water and sediments of the Gulf of Finland (Baltic Sea) in winter and in summer was studied. Substrate specificity of the isolates for aliphatic and aromatic hydrocarbons was studied. The isolates belonged to 32 genera of the types Proteobacteria (alpha-, beta-, and gammaproteobacteria), Actinobacteria,Firmicutes, and Bacteroidetes. Seasonal variations of the oil-degrading microbial communities was revealed. The presence of the known genes responsible for the degradation of oil aliphatic and aromatic hydrocarbons was determined. The alkB sequence of the alkane hydroxylase gene was found in ~16% of the studied strains. The sequence of the phnAc phenanthrene 3,4- dioxygenase was found in Sphingobacterium sp. and Arthrobacter sp. isolates retrieved in winter and summer. In five Pseudomonas sp. strains from winter samples, the classical operons of naphthalene degradation (nah) were localized in catabolic plasmids, of which three belonged to IncР-9, one, to IncР-7, and two to an unidentified incompatibility group. Burkholderia and Delftia strains contained the operons for naphthalene degradation via salicylate and gentisate (nag). The presence of nag genes has not been previously reported for Delftia spp. strains. The sequences of the nagG salicylate 5-hydroxylase gene were also found in Achromobacter, Sphingobacterium, and Stenotrophomonas strains.
Similar content being viewed by others
References
Bakirov, E.A., Ermolkin, V.I., Larin, V.I., Mal’tsev, A.K., and Rozhkov, E.L., Geologiya nefti i gaza (Oil and Gas Geology), Moscow: Nedra, 1990.
De Bruyne, K., Slabbinck, B., Waegeman, W., Vauterin, P., De Baets, B., and Vandamme, P., Bacterial species identification from MALDI-TOF mass spectra through data analysis and machine learning, Syst. Appl. Microbiol., 2011, vol. 34, pp. 20–29.
Evans, W.C., Fernley, H.N., and Griffiths, E., Oxidative metabolism of phenantrene and anthracene by soil pseudomonads: the ring-fission mechanism, J. Biochem., 1965, vol. 95, pp. 819–831.
Ferrero, M., Llobet-Brossa, E., Lalucat, J., Garcia-Valdes, E., Rosselo-Mora, R., and Bosch, R., Coexistence of two distinct copies of naphthalene degradation genes in Pseudomonas strains isolated from the western mediterranean region, Appl. Environ. Microbiol., 2002, vol. 68, pp. 957–962.
Greated, A. and Thomas, C.M. A pair of PCR primers for IncP-9 plasmids, Microbiology (UK), 1999, vol. 145, pp. 3003–3004.
Griffin, R., Churchill, P., Churchill, S., and Jones, L., Biodegradation rate enhancement of hydrocarbons by an oleophilic fertilizer and a rhamnolipid biosurfactant, J. Environ. Qual., 1995, vol. 24, pp. 19–28.
HELCOM, HELCOM Annual Report on Discharges Observed during Aerial Surveillance in the Baltic Sea, 2015, 2016.
Izmalkova, T.Y., Sazonova, O.I., Nagornih, M.O., Sokolov, S.L., Kosheleva, I.A., and Boronin, A.M., The organization of naphthalene degradation genes in Pseudomonas putida strain AK5, Res. Microbiol., 2013, vol. 164, pp. 244–253.
Izmalkova, T.Yu., Mavrodi, D.V., Sokolov, S.L., Kosheleva, I.A., and Boronin, A.M., Molecular classification of IncP-9 naphthalene degradation plasmids, Plasmid, 2006, vol. 56, pp. 1–10.
Izmalkova, T.Yu., Sazonova, O.I., Kosheleva, I.A., and Boronin, A.M., Phylogenetic analysis of the genes for naphthalene and phenanthrene degradation in Burkholderia sp. strains, Russ. J. Genet., 2013, vol. 49, no. 6, pp. 609–616.
Izmalkova, T.Yu., Sazonova, O.I., Sokolov, S.L., Kosheleva, I.A., and Boronin, A.M., The P-7 incompatibility group plasmids responsible for biodegradation of naphthalene and salicylate in fluorescent pseudomonads, Microbiology (Moscow), 2005, vol. 74, no. 3, pp. 290–295.
Jutkina, J., Heinaru, E., Vedler, E., Juhanson, J., and Heinaru, A., Occurrence of plasmids in the aromatic degrading bacterioplankton of the Baltic Sea, Genes, 2011, vol. 2, pp. 853–868.
Kimura, M., The Neutral Theory of Molecular Evolution, Cambridge University Press, Cambridge, 1983.
Kostka, J.E., Prakash, O., Overholt, W.A., Green, S.J., Freyer, G., Canion, A., Delgardio, J., Norton, N., Hazen, T.C., and Huettel, M., Hydrocarbondegrading bacteria and the bacterial community response in gulf of Mexico beach sands impacted by the deepwater horizon oil spill, Appl. Environ. Microbiol., 2011, vol. 77, pp. 7962–7974.
Kreitsberg, R., Tuvikene, A., Barsiene, J., Fricke, N.F., Rybakovas, A., Andreikenaite, L., Rumvolt, K., and Vilbaste, S., Biomarkers of environmental contaminants in the coastal waters of Estonia (Baltic Sea): effects on eelpouts (Zoarces viviparus), J. Environ. Monit., 2012, vol. 14, pp. 2298–2308.
Laurie, A.D. and Lloyd-Jones, G., Quanification of phnAc and nahAc in contaminated New Zealand soils by competitive PCR, Appl. Environ. Microbiol., 2000, vol. 66, pp. 1814–1817.
Laurie, A.D. and Lloyd-Jones, G., The phn genes of Burkholderia sp. strain RP007 constitute a divergent gene cluster for polycyclic aromatic hydrocarbon catabolism, J. Bacteriol. 1999, vol. 181, pp. 531–540.
Peix, A., Ramirez-Bahena, M.-H., and Velazquez, E., Historical evolution and current status of the taxonomy of genus Pseudomonas, Infect. Genet. Evol., 2009, vol. 9, pp. 1132–1147.
Reunamo, A., Riemann, L., Leskinen, P., and Jørgensen, K.S., Dominant petroleum hydrocarbondegrading bacteria in the Archipelago Sea in South-West Finland (Baltic Sea) belong to different taxonomic groups than hydrocarbon degraders in the oceans, Mar. Pollut. Bull., 2013, vol. 72, pp. 174–180.
Riemann, L., Leitet, C., Pommier, T., Simu, K., Holmfeldt, K., Larsson, U., and Hagström, Å., The native bacterioplankton community in the central Baltic Sea is influenced by freshwater bacterial species, Appl. Environ. Microbiol., 2008, vol. 74, pp. 503–515.
Rojo, F., Degradation of alkanes by bacteria, Environ. Microbiol., 2009, vol. 11, pp. 2477–2490.
Rossello-Mora, R.A., Lalucat, J., and Garcia-Valdes, E., Comparative biochemical and genetic analysis of naphthalene degradation among Pseudomonas stutzeri strain, Appl. Environ. Microbiol., 1994, vol. 60, pp. 966–972.
Sambrook, J., Fritsch, E.F., and Maniatis, T., Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor: Cold Spring Harbor Laboratory Press, 1989.
Sokolov, S.L., Sazonova, O.I., Gafarov, A.B., Ivanova, A.A., Vetrova, A.A., Prisiazhnaya, N.V., Kosheleva, I.A., and Boronin, A.M., Assessment of degradative potential of oil-oxidizing microorganisms of the Baltic Sea in model microcosms, Biotechnology in Russia, 2017, vol. 33, no. 4, pp. 76–84.
Timmis, K.N., Lehrbach, P.R., Harayama, S., Don, R.H., Mermod, N., Bas, S., Leppick, R., Weightman, A.J., Reineke, W., and Knackmuss, H.-J., Analysis and manipulation of plasmid-encoded pathways for the catabolism of aromatic compounds by soil bacteria, in Plasmids in Bacteria, Helinski, D.R., Cohen, C.N., Clewell, D.B., Jackson, D.A., and Hollaender, A., Eds., N.-Y.: Plenum, 1985, pp. 719–739.
Van Beilen, J.B. and Funhoff, E.G., Alkane hydroxylases involved in microbial alkane degradation, Appl. Microbiol. Biotechnol., 2007, vol. 74, pp. 13–21.
Van de Peer, Y. and De Wachter, R., TREECON for Windows: a software package for the construction and drawing of evolutionary trees for the Microsoft Windows environment, Comput. Applic. Biosci., 1994, vol. 10, pp. 569–570.
Viggor, S., Juhanson, J., Jõesaar, M., Mitt, M., Truu, J., Vedler, E., and Heinaru, A., Dynamic changes in the structure of microbial communities in the Baltic Sea coastal seawater microcosms modified by crude oil, shale oil or diesel fuel, Microbiol. Res., 2013, vol. 168, pp. 415–427.
Wilkstrom, P., Wilklund, A., Anderson, A.C., and Forman, M., DNA recovery and PCR quantification of catechol-2,3-dioxygenase genes from different soil types, J. Biotechnol., 1996, vol. 52, pp. 107–120.
Yakimov, M.M., Timmis, K.N., and Golyshin, P.N., Obligate oil-degrading marine bacteria, Curr. Opin. Biotechnol., 2007, vol. 18, pp. 257–266.
Yang, Y.Y., Wang, J., Liao, J.Q., Xie, S.G., and Huang, Y., Abundance and diversity of soil petroleum hydrocarbondegrading microbial communities in oil exploring areas, Appl. Microbiol. Biotechnol., 2015, vol. 99, pp. 1935–1946.
Zhou, N.Y., Fuenmayor, S.L., and Williams, P.A., nag genes of Ralstonia (formerly Pseudomonas) sp. strain U2 encoding enzymes for gentisate catabolism, J. Bacteriol., 2001, vol. 183, pp. 700–708.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.Yu. Izmalkova, A.B. Gafarov, O.I. Sazonova, S.L. Sokolov, I.A. Kosheleva, A.M. Boronin, 2018, published in Mikrobiologiya, 2018, Vol. 87, No. 2, pp. 204–216.
Rights and permissions
About this article
Cite this article
Izmalkova, T.Y., Gafarov, A.B., Sazonova, O.I. et al. Diversity of Oil-Degrading Microorganisms in the Gulf of Finland (Baltic Sea) in Spring and in Summer. Microbiology 87, 261–271 (2018). https://doi.org/10.1134/S0026261718020054
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261718020054