Abstract
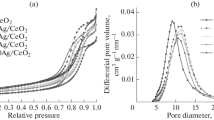
The interaction of silver with the surface of CeO2 in the Ag/CeO2 catalysts prepared by coprecipitation and impregnation techniques was studied by temperature-programmed reduction, X-ray diffraction, and high-resolution transmission electron microscopy. It was shown that coprecipitation technique led to formation of strong silver–support interaction and the epitaxy of silver particles (d 111 = 2.35 Å) on the surface of CeO2 (d 111 = 3.1 Å). This provided incresed catalytic activity in the oxidative dehydrogenation of ethanol at relatively low temperatures (a 15% conversion of ethanol with 100% selectivity for the formation of acetaldehyde was reached at 85°C). Above 130°C, the deep oxidation of ethanol to CO2 becomes the predominant direction of a catalytic reaction, and the Ag/CеО2 catalyst obtained by impregnation technique was most active in this region as a consequence of the weaker metal–support interaction.
Similar content being viewed by others
References
Qu, Z., Yu, F., Zhang, X., Wang, Y., and Gao, J., Chem. Eng. J., 2013, vol. 229, p. 522.
Fu, Q. and Wagner, T., Surf. Sci. Rep., 2007, vol. 62, p. 431.
Ma, L., Wang, D., Li, J., Bai, B., Fu, L., and Li, Y., Appl. Catal., B, 2014, vols. 148–149, p. 36.
Chang, S., Li, M., Hua, Q., Zhang, L., Ma, Y., Ye, B., and Huang, W., J. Catal., 2012, vol. 293, p. 195.
Bera, P., Patil, K.C., and Hegde, M.S., Phys. Chem., 2000, vol. 2, p. 3715.
Skaf, M., Aouad, S., Hany, S., Cousin, R., Abi-Aad, E., and Aboukais, A., J. Catal., 2014, vol. 320, p. 137.
Kayama, T., Yamazaki, K., and Shinjoh, H., J. Am. Chem. Soc., 2010, vol. 132, p. 13154.
Shimizu, K., Kawachi, H., and Satsuma, A., Appl. Catal., B, 2010, vol. 96, p. 169.
Aneggi, E., Llorca, J., Leitenburg, C., Dolcetti, G., and Trovarelli, A., Appl. Catal., B, 2009, vol. 91, p. 489.
Lee, C., Park, J., Shul, Y.-G., Einaga, H., and Teraoka, Y., Appl. Catal., B, 2015, vol. 174, p. 185.
Machida, M., Murata, Y., Kishikawa, K., Zhang, D., and Ikeue, K., Chem. Mater., 2008, vol. 20, p. 4489.
Yamazaki, K., Kayama, T., Dong, F., and Shinjoh, H., J. Catal., 2011, vol. 282, p. 289.
Leng, Q., Yang, D., Yang, Q., Hu, C., Kang, Y., Wang, M., and Hashim, M., Mater. Res. Bull., 2015, vol. 266.
Wang, L., He, H., Yu, Y., Sun, L., Liu, S., Zhang, C., and He, L., J. Inorg. Biochem., 2014, vol. 135, p. 45.
Mamontov, G.V., Grabchenko, M.V., Sobolev, V.I., Zaikovskii, V.I., and Vodyankina, O.V., Appl. Catal., A, 2016, vol. 528, p. 161.
Mitsudome, T., Mikami, Y., Matoba, M., Mizugaki, T., Jitsukawa, K., and Kaneda, K., Angew. Chem., Int. Ed. Engl., 2012, vol. 51, p. 136.
Zhang, J., Li, L., Huang, X., and Li, G., J. Mater. Chem., 2012, vol. 22, p. 10480.
Bokii, G.K., Kristallokhimiya (Crystal Chemistry), Moscow: Nauka, 1971.
Yao, H.C. and Yu-Yao, Y.F., J. Catal., 1984, vol. 86, p. 254.
Zhu, H., Qin, Z., Shan, W., Shen, W., and Wang, J., J. Catal., 2004, vol. 225, p. 267.
Mamontov, G.V., Dutov, V.V., Sobolev, V.I., and Vodyankina, O.V., Kinet. Catal., 2013, vol. 54, no. 4, p. 487.
Qu, Z., Huang, W., Cheng, M., and Bao, X., J. Phys. Chem. B, 2005, vol. 109, p. 15842.
Mamontov, G.V., Izaak, T.I., Magaev, O.V., Knyazev, A.S., and Vodyankina, O.V., J. Phys. Chem. A, 2011, vol. 85, no. 9, p. 1536.
Scire, S., Riccobene, P.M., and Crisafulli, C., Appl. Catal., B, 2010, vol. 101, p. 109.
Feio, L.S.F., Hori, C.E., Damyanova, S., Noronha, F.B., Cassinelli, W.H., Marques, C.M.P., and Bueno, J.M.C., Appl. Catal., A, 2007, vol. 316, p. 107.
Acerbi, N., Edman, TsangS.C., Jones, G., Golunski, S., and Collier, P., Angew. Chem., Int. Ed. Engl., 2013, vol. 52, p. 7737.
Liotta, L.F., Longo, A., Macaluso, A., Martorana, A., Pantaleo, G., Venezia, A.M., and Deganello, G., Appl. Catal., B, 2004, vol. 48, p. 133.
Mullins, D.R., Surf. Sci. Rep., 2015, vol. 70, p. 42.
Mitsudome, T., Noujima, A., Mikami, Y., Mizugaki, T., Jitsukawa, K., and Kaneda, K., Angew Chem., 2010, vol. 122, p. 5677.
Mikami, Y., Noujima, A., Mitsudome, T., Mizugaki, T., Jitsukawa, K., and Kaneda, K., Chem. Lett., 2010, vol. 39, p. 223.
Shimizu, K., Sugino, K., Sawabe, K., and Satsuma, A., Chem. Eur. J., 2009, vol. 15, p. 2341.
Vayssilov, G.N., Lykhach, Y., Migani, A., Staudt, T., Petrova, G.P., Tsud, N., Skala, T., Bruix, A., Illas, F., Prince, K.C., Matolin, V., Neyman, K.M., and Libuda, J., Nat. Mater., 2011, vol. 10, p. 310.
Dutov, V.V., Mamontov, G.V., Zaikovskii, V.I., and Vodyankina, O.V., Catal. Today, 2016, vol. 278, p. 150.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.V. Grabchenko, G.V. Mamontov, V.I. Zaikovskii, O.V. Vodyankina, 2017, published in Kinetika i Kataliz, 2017, Vol. 58, No. 5, pp. 654–661.
Rights and permissions
About this article
Cite this article
Grabchenko, M.V., Mamontov, G.V., Zaikovskii, V.I. et al. Effect of the metal−support interaction in Ag/CeO2 catalysts on their activity in ethanol oxidation. Kinet Catal 58, 642–648 (2017). https://doi.org/10.1134/S0023158417050056
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158417050056