Abstract
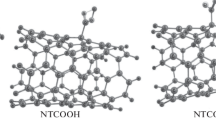
A doctor can find out how pancreas is working using bentiromide. This drug can exist as six possible tautomers. Herein, by density functional theory (DFT) and handling the solvent effects with the polarizable continuum model (PCM), the mechanism of its tautomerization, energies, and structural parameters of the tautomers are investigated. Also, the natural bond orbital analysis (NBO) is used for exploring the frontier molecular orbitals. The most stable tautomer of bentiromide has three carbonyl groups in its structure. The amount of other tautomers is negligible in the aqueous solution. The non-covalent interactions of the most stable tautomer of bentiromide with an armchair (5,5) single-wall carbon nanotube and a γ-Fe2O3 nanoparticle are explored. In each case, three possible forms are optimized. Their most stable form is determined. The intermolecular H bonds have a critical role in the energy behavior of the interaction between bentiromide and the γ-Fe2O3 nanoparticle.
Similar content being viewed by others
References
A. Imondi, R. Stradley, and R. Wolgemuth. Gut, 1972, 13, 726.
K. Gyr, R. Wolf, A. Imondi, and O. Felsenfeld. Gastroenterology, 1975, 68, 488.
C. Arvanitakis and N. Greenberger. The Lancet, 1976, 307, 663.
H. Harada, K. Mishima, T. Shundo, N. Yamamoto, T. Sasaki, T. Hinofuji, T. Hayashi, Y. Uchida, and I. Kimura. Am. J. Gastroenterology, 1979, 71.
C. Yamato and K. Kinoshita. J. Pharmacol. Exp. Ther., 1978, 206, 468.
C. Lang, K. Gyr, G. Stalder, and D. Gillessen. British J. Surgery, 1981, 68, 771.
R. Chowdhury and C. Forsmark. Aliment. Pharm. Therap., 2003, 17, 733.
F. Heshmatipour, S.A. Beyramabadi, A. Morsali, and M. M. Heravi. J. Struct. Chem., 2016, 57, 1096.
A. Khaleghi-Rad, S. A. Beyramabadi, A. Morsali, M. Ebrahimi, and M. Khorzandi-Chenarboo. J. Struct. Chem., 2017, 58, 244.
H. L. H. Mendoza, G. Salgado-Morán, W. Cardona-Villada, A. G. Pacheco, and D. Glossman-Mitnik. J. Serb. Chem. Soc., 2016, 77.
B. Eren and Y. Y. Gurkan. J. Serb. Chem. Soc., 2017, 82, 277.
M. Frisch, G. Trucks, H. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, J. Montgomery Jr, T. Vreven, K. Kudin, and J. Burant. Gaussian Inc. Pittsburgh, PA, 2003.
C. Lee, W. Yang, and R. G. Parr. Phys. Rev. B, 1988, 37, 785.
R. Cammi and J. Tomasi. J. Comput. Chem., 1995, 16, 1449.
G. Zhurko and D. Zhurko. URL: http://www.chemcraftprog.com, 2009.
N. Tezer and N. Karakus. J. Mol. Model., 2009, 15, 223.
N. Özdemir, M. Dinçer, A. Çukurovalı, and O. Büyükgüngör. J. Mol. Model., 2009, 15, 1435.
W. Ma and Y. Fang. J. Nanoparticle Res., 2006, 8, 761. 1018
S. Farhadi, F. Mahmoudi, and J. Simpson. J. Mol. Struct., 2016, 1108, 583.
M. Anuratha, A. Jawahar, M. Umadevi, V. Sathe, P. Vanelle, T. Terme, O. Khoumeri, V. Meenakumari, and A. M. F. Benial. Spectrochim. Acta A, 2015, 149, 558.
Y. Wang, S. Liu, Z. Liu, J. Yang, and X. Hu. Spectrochim. Acta A, 2013, 105, 612.
F. Gobal, R. Arab, and M. Nahali. J. Mol. Struct.: THEOCHEM, 2010, 959, 15.
A. Magham, A. Morsali, Z. Es′haghi, S. Beyramabadi, and H. Chegini. Prog. React. Kinet. Mec., 2015, 40, 119.
E. Mohammad-Hasani, S. A. Beyramabadi, and M. Pordel. Indian J. Chem. A, 2017, 56, 626.
Acknowledgments
We gratefully acknowledge Dr. Ali Morsali (Associate Professor in Islamic Azad University, Mashhad Branch) for his kind cooperation in this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Text © The Author(s), 2019, published in Zhurnal Strukturnoi Khimii, 2019, Vol. 60, No. 6, pp. 1049–1058.
Conflict of Interests
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Tanhaei, R., Beyramabadi, S.A. & Behmadi, H. Geometry, Tautomerism, and Noncovalent Interaction of Bentiromide with a Carbon-Nanotube and γ-Fe2O3 Nanoparticles: A Quantum Mechanical Study. J Struct Chem 60, 1008–1018 (2019). https://doi.org/10.1134/S0022476619060155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476619060155