Abstract
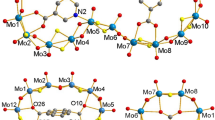
The bis(oxalate) complex of niobium (NH4)[NbO(C2O4)2(H2O)2]·3H2O can be used as a source of niobium to incorporate the {NbO}3+ group into a cavity of the monolacunary Dawson-type [α2-P2W17O61]10− anion. The product is isolated from the reaction mixture as dimethylammonium salt (Me2NH2)7[α2-P2W17NbO62]·3H2O (1) and characterized by single crystal XRD, IR and Raman spectroscopy, and elemental analysis. (NBu4)6(Me2NH2)[α2-P2W17NbO62] (2) is obtained by adding Bu4NBr to an aqueous solution of 1. The comparison of the electrochemical properties of complex 2 and (NBu4)6[P2W18O62] shows that the incorporation of niobium atom hinders the anion reduction.
Similar content being viewed by others
References
N. V. Maksimchuk, K. A. Kovalenko, S.S. Arzumanov et al. Inorg. Chem., 2010, 49, 2920–2930.
O. A. Kholdeeva, N. V. Maksimchuk, and G. M. Maksimov. Catal. Today, 2010, 157, 107–113.
N. Mizuno, K. Kamata, and K. Yamaguchi. Top. Catal, 2010, 53, 876–893.
N. Mizuno, H. Weiner, and R. G. Finke. J. Mol. Catal A Chem., 1996, 114, 15–28.
P. Zhang, M. Wang, Y. Na et al. Dalton Trans., 2010, 39, 1204–1206.
J.-J. Chen, M. D. Symes, S.-C. Fan et al. Adv. Mater., 2015, 27, 4649–4654.
C. Busche, L. Vilà-Nadal, J. Yan et al. Nature, 2014, 515, 545–549.
C. Jasmin, N. Raybaud, J.-C. Chermann et al. Biomedicine, 1973, 18, 319–27.
C. Jasmin, J.-C. Chermann, G. Herve et al. JNCI J. Natl. Cancer Inst., 1974, 53, 469–474.
J. T. Rhule, C. L. Hill, D.A. Judd et al. Chem. Rev., 1998, 98, 327–358.
Biomedical Inorganic Polymers: Bioactivity, Applications of Natural, Synthetic Polymeric Inorganic Molecules. / Eds: W. E. G. Müller, H. C. Schröder, and C. Heinz, 2013, 65, 116.
C. L. Hill, M. S. Weeks, and R. F. Schinazi. J. Med. Chem., 1990, 33, 2767–2772.
D. A. Judd, J. H. Nettles, N. Nevins et al. J. Am. Chem. Soc., 2001, 123, 886–897.
P. A. Abramov, A. A. Shmakova, M. Haouas et al. New J. Chem., 2017, 41, 256–262.
A. A. Shmakova, M. M. Akhmetova, V.V. Volchek et al. New J. Chem., 2018, 42, 7940–7948.
Inorganic syntheses. / Ed. A. P. Ginsberg., 1990, 27, 464.
C. R. Graham, L. S. Ott, and R. G. Finke. Langmuir, 2009, 25, 1327–1336.
G. M. Sheldrick. Acta Cryst. A, Found. Adv., 2015, 71, 3–8.
G. M. Sheldrick. Acta Cryst. C, Struct. Chem., 2015, 71, 3–8.
C. B. Hübschle, G. M. Sheldrick, and B. Dittrich. J. Appl Crystallogr., 2011, 44, 1281–1284.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Strukturnoi Khimii, 2019, Vol. 60, No. 6, pp. 1002–1007.
Funding
The work was supported by Russian Science Foundation grant No. 14-13-00645.
Conflict of Interests
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Shmakova, A.A., Gushchin, A.L., Abramov, P.A. et al. Synthesis and Electrochemical Properties of ((CH3)2NH2)7[P2W17NbO62]. J Struct Chem 60, 961–966 (2019). https://doi.org/10.1134/S0022476619060106
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476619060106