Abstract
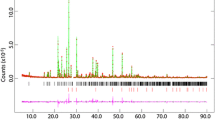
The crystal structural characteristics of the [NiEn3]MoO4 complex salt (En is ethylenediamine) at 90 K are as follows: space group \(P\overline 3 ,\;a = 15.9307\left( 9 \right)\;\acute{\mathring{\mathrm{A}}} ,\;c = 9.9238\left( 6 \right)\;\acute{\mathring{\mathrm{A}}} ,\;V = 2181.1\left( 3 \right)\;{\acute{\mathring{\mathrm{A}}} ^3},\;Z = 6,\;{d_{\rm{x}}} = 1.822\;{\rm{g/c}}{{\rm{m}}^{\rm{3}}}\), Ni-N is \(2.1182\left( {12} \right) - 2.1498\left( {11} \right)\;\acute{\mathring{\mathrm{A}}} ,\) ∠Ni-N-N is 80.76(4)-82.27(4)°. According to the differential scanning calorimetry data in a range from 295 K to 310 K, there is a thermal anomaly with peaks at T1 = 299.6 K and T2 = 304.7 K. The crystal structural characteristics at 320 K are as follows: space group \(P\overline 3 \,1c,\;a = 9.2491\left( 4 \right)\;\acute{\mathring{\mathrm{A}}} ,\;c = 9.9713\left( 4 \right)\;\acute{\mathring{\mathrm{A}}} ,\;V = 738.72\left( 7 \right)\;{\acute{\mathring{\mathrm{A}}} ^3},\;Z = 2,\;{d_x} = 1.794\;{\rm{g/c}}{{\rm{m}}^3}\), Ni-N is \(2.1302\left( {14} \right)\;\acute{\mathring{\mathrm{A}}} \), ∠N-Ni-N is 80.96(8)°. With increasing temperature from 90 K to 320 K a decrease in the average Mo-O distance from \(1.769\;\acute{\mathring{\mathrm{A}}} \) to \(1.725\;\acute{\mathring{\mathrm{A}}} \) is observed in the structure. The comparative analysis of the interionic N-H…O and C-H…O contacts is carried out. The ex situ powder X-ray diffraction study of the formation process of metal and carbide phases by the [NiEn3]MoO4 thermal decomposition in the presence of LiH in the He atmosphere is performed. At the temperature of 1323 K a Mo2C and MoNi4 phase mixture forms in the first minute. With increasing keeping time the Ni2Mo4Cx phase forms.
Similar content being viewed by others
References
B.-Z. Lin, G.-H. Han, F. Geng, and C. Ding. Acta Cryst., 2006, E62, 532–534.
F. H. Allen. Acta Crystallogr., 2002, B58(3–1), 380–388.
M. Lutz. Acta. Cryst. Sect. C: Cryst. Struct. Comm., 2010, 66, 330–335.
G. B. Jameson, R. Shneider, E. Dubler, and H. R. Oswald. Acta Cryst., 1982, B38, 3016–3020.
A. S. Sukhih, S. P. Khranenko, S. P. Pishchur, and S. A. Gromilov. J. Struct. Chem., 2018, 59(3), 657–663.
S. P. Khranenko, V. Y. Komarov, E. Y Gerasimov, A. V. Zadesenets, E. A. Maksimovsky, and S. A. Gromilov. J. Struct. Chem., 2017, 58(7), 1448–1452.
S. A. Gromilov, E. Y. Gerasimov, S. P. Khranenko, V. Y. Komarov, and A. V. Zadesenets. J. Struct. Chem., 2017, 58(7), 1443–1447.
S. P. Khranenko, A. S. Sukhih, S. P. Pishchur, P. S. Buneeva, V. Y. Komarov, and S. A. Gromilov. J. Struct. Chem., 2018, 59(8), 1960–1965.
W. Kraus and G. Nolze. J. Appl. Crystallogr., 1996, 29(3), 301–303.
Ya. Gibner and I. Vasilyeva. J. Therm. Anal., 1998, 53, 151–160.
S. A. Gromilov, R. E. Nikolaev, and S. V. Cherepanova. J. Struct. Chem., 2018, 59(2), 395–397.
A. V. Panchenko, N. D Tolstykh, and S. A Gromilov. J. Struct. Chem., 2014, 55(7), 1209–1214.
A. Yelisseyev, A. Khrenov, V. Afanasiev, V. Pustovarov, S. Gromilov, A. Panchenko, N. Pokhilenko, and K. Litasov. Diam. Rel. Mater., 2015, 58, 69–77.
A. Rodriguez-Navarro. J. Appl. Cryst., 2006, 39(6), 905–909.
C. Prescher and V. B. Prakapenka. High Pres. Res., 2015, 35(3).
Powder Diffraction File. PDF-2. International Centre for Diffraction Data, USA.
Inorganic Crystal Structure Database. D-1754. Eggenstein-Leopoldshafen, Germany.
Bruker AXS Inc. APEX2 V2013.6-2, SAINT V8.32B and SADABS-2012/1. Bruker Advanced X-ray Solutions, Madison, Wisconsin, USA.
O. V. Dolomanov, L. J. Bourhis, R. J Gildea, J. A. K. Howard, and H. Puschmann. J. Appl. Cryst., 2009, 42, 339–341.
G. M. Sheldrick. Acta Cryst., 2015, A71, 3–8.
G. M. Sheldrick. Acta Cryst., 2015, C71, 3–8.
A. L. Spek. Acta Cryst., 2009, D65, 148–155.
G. B. Jameson, R. Shneider, E. Dubler, and H. R. Oswald. Acta Cryst., 1982, B38, 3016–3020.
F. L. Hirshfeld. Theor. Chim. Acta, 1977, 44(2), 129–138.
M. A. Spackman and P. G. Byrom. Chem. Phys. Lett. 1997, 267(3–4), 215–220.
M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, P. R. Spackman, D. Jayatilaka, and M. A. Spackman. CrystalExplorer17. University of Western Australia, 2017. http://hirshfeldsurface.net.
D. Jayatilaka and D. J. Grimwood. Comput. Sci. — ICCS, 2003, 4, 142–151.
E. A. Bykova, S. P. Khranenko, and S. A. Gromilov. J. Struct. Chem., 2012, 53(1), 186–190.
J. S. O. Evans, T. A Mary, and A. W. Sleight. J. Solid State Chem., 1998, 137, 148–160.
D. A. Woodcock, P. Lightfoot, and C. Ritter. J. Solid State Chem., 2000, 149, 92–98.
M. Wu, X. Liu, D. Chen, Q. Huang, H. Wu, and Y. Liu. Inorg. Chem., 2014, 53, 9206–9212.
L. F. Kozin and N. V. Mashkova. Ukr. Himich. Zhurn, 2009, 75(11), 48–54.
L. F. Kozin and S. V. Volkov. Modern Energetics and Ecology: Problems and Prospects [in Russian]. Naukova Dumka: Kiev, 2006, 647.
H. Ago, N. Uehara, N. Yoshihara, M. Tsuji, M. Yumura, and N. Tomonaga. Carbon, 2006, 44, 2912–2918.
Yu. I. Bauman, I. V. Mishakov, Yu. V. Rudneva, P. E. Plyusnin, Yu. V. Shubin, D. V. Korneev, and A. A. Vedyagin. Industr. & Eng. Chem. Res., 2018, in press. DOI: https://doi.org/10.1021/acs.iecr.8b02186.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Strukturnoi Khimii, 2019, Vol. 60, No. 5, pp. 814-822.
Rights and permissions
About this article
Cite this article
Sukhikh, A.S., Khranenko, S.P., Komarov, V.Y. et al. [NiEn3]MoO4: Features of the Phase Transition and Thermal Decomposition in the Presence of Lithium Hydride. J Struct Chem 60, 780–788 (2019). https://doi.org/10.1134/S002247661905010X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002247661905010X