Abstract
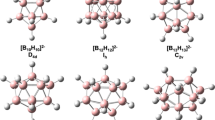
The work summarizes the data on currently known dimeric boron cluster anions with the general formula [B20H18]2−. Their preparation, structure, and the methods of structural modification are considered. The data on the reactivity of boron dimeric clusters in redox, substitution, complexation, and condensation reactions are presented. The isomerism of dimeric polyhedral boron cluster anions associated with the formation of substituted derivatives and coordination compounds is considered. The methods of reversible transformation of isomers in salts and coordination compounds in solutions and single crystals are discussed.
Similar content being viewed by others
References
W. N. Lipscomb. Boron Hydrides. New York, Amsterdam: W. H. Benjamin, 1963.
E. L. Muetterties, J. H. Balthis, Y. T. Chia, W. H. Knoth, and H. C. Miller. Inorg. Chem., 1964, 3, 444.
E. L. Muetterties and W. H. Knoth. Polyhedral Boranes. Dekker, New York, 1968.
N. N. Greenwood and A. Earnshaw. Chemistry of the Elements (2nd ed.). Butterworth-Heinemann, 1997.
K. Wade. Adv. Inorg. Chem. Radiochem., 1978, 18, 1.
J. Aihara. J. Am. Chem. Soc., 1978, 100, 3339.
Boron Hydride Chemistry. / Ed. E. L. Muetterties. Acad. Press: New York, 1975.
A. Kaczmarczyk, R. D. Dobrott, and W. N. Lipscomb. Proc. Natl. Acad. Sci. USA, 1962, 48, 729.
B. L. Chamberland and E. L. Muetterties. Inorg. Chem., 1964, 3, 1450.
M. F. Hawthorne, R. L. Pilling, P. F. Stokely, and P. M. Garrett. J. Am. Chem. Soc., 1963, 85, 3704.
E. A. Malinina, V. V. Avdeeva, L. V. Goeva et al. Russ. J. Inorg. Chem., 2011, 56(5), 687. https://doi.org/10.1134/S0036023611050160].
M. F. Hawthorne and R. L. Pilling. J. Am. Chem. Soc., 1966, 88, 3873.
Z. B. Curtis, C. Young, R. Dickerson, and A. Kaczmarczyk. Inorg. Chem., 1974, 13, 1760.
W. N. Lipscomb and M. F. Hawthorne. J. Am. Chem. Soc., 1962, 84, 3026.
F. Li, K. Shelly, C. B. Knobler, and M. F. Hawthorne. Angew. Chem., Int. Ed., 1998, 37, 1865.
M. F. Hawthorne, K. Shelly, and F. Li. Chem. Commun., 2002, 547.
M. F. Hawthorne, R. L. Pilling, and P. F. Stokely. J. Am. Chem. Soc., 1965, 87, 1893.
M. F. Hawthorne, R. L. Pilling, P. F. Stokely, and P. M. Garrett. J. Am. Chem. Soc., 1963, 85, 3704.
R. A. Watson-Clark, C. B. Knobler, and M. F. Hawthorne, J. Am. Chem. Soc., 1966, 35, 2963.
M. F. Hawthorne, R. L. Pilling, and P. M. Garrett. J. Am. Chem. Soc., 1965, 87, 4740.
K. Shelly, F. Li, R. A. Watson-Clark, M. F. Hawthorne. In: Advances in Neutron Capture Therapy. Volume II, Chemistry and Biology. / Eds. B. Larsson, J. Crawford, and R. Weinrich. Elsevier Science: Amsterdam, 1997, 30–34.
D. A. Feakes, K. Shelly, C. B. Knobler, and M. F. Hawthorne. Proc. Natl. Acad. Sci. USA, 1994, 91, 3029.
E. M. Georgiev, K. Shelly, D. A. Feakes, J. Kuniyoshi, S. Romano, and M. F. Hawthorne. Inorg. Chem., 1996, 35, 5412.
F. Li, K. Shelly, R. R. Kane, C. B. Knobler, and M. F. Hawthorne. Angew. Chem., Int. Ed., 1996, 35, 2646.
F. Li, K. Shelly, R. R. Kane, C. B. Knobler, and M. F. Hawthorne. J. Am. Chem. Soc., 1996, 118, 6506.
I. B. Sivaev, A. V. Prikaznov, and D. Naoufal. Collect. Czech. Chem. Commun., 2010, 75, 1149.
K. Yu. Zhizhin, A. P. Zhdanov, and N. T. Kuznetsov. Russ. J. Inorg. Chem., 2010, 55, 2089.
K. Shelly, D. A. Feakes, and M. F. Hawthorne. Proc. Natl. Acad. Sci. USA, 1992, 89, 9039.
E. A. Il’inchik, T. M. Polyanskaya, M. K. Drozdova, K. G. Myakishev, V. N. Ikorskii, and V. V. Volkov. Russ. J. Gen. Chem., 2005, 75, 1545.
V. V. Avdeeva, E. A. Malinina, L. V. Goeva et al. Dokl. Chem., 2017, 474, 141.
Zh. Zhank. Acta Cryst., 1989, 45, 333.
D. R. Pearson. J. Am. Chem. Soc., 1963, 85, 3533.
E. A. Malinina, V. V. Avdeeva, L. V. Goeva, and N. T. Kuznetsov. Russ. J. Inorg. Chem., 2010, 55, 2148.
V. V. Avdeeva, E. A. Malinina, I. B. Sivaev, V. I. Bregadze, and N. T. Kuznetsov. Crystals, 2016, 6, 60.
E. O. Firsova, V. V. Avdeeva, V. I. Privalov et al. Dokl. Chem., 2015, 465, 291.
V. V. Avdeeva, M. I. Buzin, E. A. Malinina, N. T. Kuznetsov, and A. V. Vologzhanina. Cryst. Eng. Comm., 2015, 17, 8870.
V. V. Avdeeva, I. N. Polyakova, L. V. Goeva et al. Dokl. Chem., 2011, 437, 63.
V. V. Avdeeva, A. V. Vologzhanina, M. I. Buzin, A. O. Dmitrienko, P. V. Dorovatovskii, E. A. Malinina, N. T. Kuznetsov, E. D. Voronova, and Y. V. Zubavichus. Chem. Eur. J., 2017, 23, 16819.
J. Zhang, M. Zhang, Y. Zhao, B. Chen, and C.-C. Sun. J. Comput. Chem., 2006, 27, 1817.
H. Yanjiu and Y. Yingyong. Chem. Res. Application (Chinese), 2008, 20, 16.
E. Bernhardt, D. J. Brauer, M. Finze, and H. Willner. Angew. Chem. Int. Ed., 2007, 46, 2927.
I. B. Sivaev, V. I. Bregadze, and S. Sjöberg. Collect. Czech. Chem. Commun., 2002, 67, 679.
M. Vlasse, M. Boiret, R. Naslain, J. S. Kasper, and K. Ploog. Chimie du solide. Compt. Rend. C, 1978, 287, 27.
M. Vlasse, R. Naslain, J. S. Kasper, and K. Ploog. J. Solid State Chem., 1979, 28, 289.
M. Vlasse, R. Naslain, J. S. Kasper, and K. Ploog. J. Less-Common Metals, 1979, 67, 1.
H. L. Yakel. Boron Rich Solids. In: AIP Conf. Proc., New York, 1986, 140, 97–108.
J. K. Burdett, E. Canadell. Inorg. Chem., 1991, 30, 1991.
W. Hayami. J. Solid State Chem., 2015, 221, 378.
J. S. Kasper, M. Vlasse, and R. Naslain. J. Solid State Chem., 1977, 20, 281.
I. Higashi, T. Sakurai, and T. Atoda. J. Solid State Chem., 1977, 20, 67.
R. E. Hughes, M. E. Leonowicz, J. T. Lemley, and L.-T. Tai. J. Am. Chem. Soc., 1977, 99, 5507.
I. Higashi. J. Solid State Chem., 2000, 154, 168.
N. E. Miller, J. A. Forstner, and E. L. Muetterties. Inorg. Chem., 1964, 3, 1690.
J. H. Enemark, L. B. Friedman, J. A. Hartsuck, and W. N. Lipscomb. J. Am. Chem. Soc., 1966, 88, 3659.
J. H. Enemark, L. B. Friedman, and W. N. Lipscomb. Inorg. Chem., 1966, 5, 2165.
O. Shameema, B. Pathak, and E. D. Jemmis. Inorg. Chem., 2008, 47, 4375.
P. Kaur, S. D. Perera, T. Jelinek, B. Stibr, J. D. Kennedy, W. Clegg, and M. Thornton-Pett. Chem. Commun., 1997, 2, 217.
L. B. Friedman, R. D. Dobrott, and W. N. Lipscomb. J. Am. Chem. Soc., 1963, 85, 3505.
N. E. Miller and E. L. Muetterties. J. Am. Chem. Soc., 1963, 85, 3506.
R. D. Dobrott, L. B. Friedman, and W. N. Lipscomb. J. Chem. Phys., 1964, 40, 866.
L. B. Friedman. Studies on Boron Hydrides. Harvard University, 1966.
N. E. Miller. Eicosaborane(16) preparation. In: du Pont de Nemours, E.I. and Co. 1968. US 3,404,959.
D. Hnyk, J. Holub, T. Jelinek, J. Machacek, and M. G. S. Londesborough. Collect. Czech. Chem. Commun., 2010, 75, 1115.
F. Schlueter and E. Bernhardt. Z. Anorg. Allg. Chem., 2012, 638, 594.
F. Schlüter. Die Chemie der closo-Borate [BnHn]2− (n = 6–9, 11) und [B21H18]−. Bergische Universität Wuppertal, 2012.
S. M. Eyrilmez, E. Bernhardt, J. Z. Davalos, M. Lepsik, P. Hobza, K. I. Assaf, W. M. Nau, J. Holub, J. M. Oliva-Enrich, J. Fanfrlik, and D. Hnyk. Phys. Chem. Chem. Phys., 2017, 19, 11748.
A. R. Pitochelli and M. F. Hawthorne. J. Am. Chem. Soc., 1962, 84, 3218.
F. P. Olsen, R. C. Vasavada, and M. F. Hawthorne. J. Am. Chem. Soc., 1968, 90, 3946.
A. A. Attia, A. Lupan, and R. Bruce King. Phys. Chem. Chem. Phys., 2016, 18, 11707.
W. N. Lipscomb. J. Less-Common Met., 1981, 82, 1.
M. M. Balakrishnarajan and E. D. Jemmis. J. Am. Chem. Soc., 2000, 122, 4516.
E. D. Jemmis, M. M. Balakrishnarajan, and P. D. Pancharatna. J. Am. Chem. Soc., 2001, 123, 4313.
E. D. Jemmis, M. M. Balakrishnarajan, and P. D. Pancharatna. Inorg. Chem., 2001, 40, 1730.
E. D. Jemmis, M. M. Balakrishnarajan, and P. D. Pancharatna. Chem. Rev., 2002, 102, 93.
E. D. Jemmis and E. G. Jayasree. Acc. Chem. Res., 2003, 36, 816.
E. D. Jemmis and P. D. Pancharatna. Appl. Organomet. Chem., 2003, 17, 480.
E. D. Jemmis, B. Pathak, and A. Anoop. Inorg. Chem., 2005, 44, 7184.
O. Shameema and E. D. Jemmis. Comput. Inorg. Bioinorg. Chem., 2009, 539.
K. Vidya and E. D. Jemmis. J. Organomet. Chem., 2015, 798, 91.
I. B. Sivaev, A. Kayumov, A. B. Yakushev, K. A. Solntsev, and N. T. Kuznetsov. Koord. Khimiya, 1989, 15, 1466.
S. Koerbe, P. J. Schreiber, and J. Michl. Chem. Rev., 2006, 106, 5208.
C. Douvris and J. Michl. Chem. Rev., 2011, 113, PR179.
M. F. Hawthorne, D. C. Young, T. D. Andrews, D. V. Howe, R. L. Pilling, A. D. Pitts, M. Reintjes, L. F. Warren, and P. A. Wegner. J. Am. Chem. Soc., 1968, 90, 879.
V. Koprda and V. Scasnar. J. Radioanal. Chem., 1979, 51, 245.
T. Popova, A. Zaulet, F. Teixidor, R. Alexandrova, and C. Vinas. J. Organomet. Chem., 2013, 747, 229.
S. H. Strauss. Chem. Rev., 1993, 93, 927.
C. A. Reed. Acc. Chem. Res., 1998, 31, 133.
C. Knapp. Compr. Inorg. Chem. II, 2013, 1, 651.
I. M. Riddlestone, A. Kraft, J. Schaefer, and I. Krossing. Angew. Chem. Int. Ed., 2018, 57, 13982.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Strukturnoi Khimii, 2019, Vol. 60, No. 5, pp. 726-747.
Rights and permissions
About this article
Cite this article
Avdeeva, V.V., Malinina, E.A., Zhizhin, K.Y. et al. Structural Diversity of Dimer Clusters Based on the Octadecahydro-Eicosaborate Anion. J Struct Chem 60, 692–712 (2019). https://doi.org/10.1134/S0022476619050020
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476619050020