Abstract
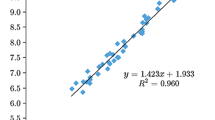
The plate dent test is one of the simplest tools for fast determination of the detonation pressure. The test is based on the observation that the detonation pressure correlates with the depth of the dent produced by a detonating explosive on a metal witness plate. The present study is aimed at developing a model for estimating the dent depth, which is used not only to obtain the detonation pressure, but also to evaluate the brisance relative to a reference explosive. It is shown that the experimental dent depth values for CHNO and CHNOClF explosives can be successfully reproduced by a model based on few parameters, namely: loading density, number of moles of gaseous detonation products per gram of the explosive, and average molecular weight of the gaseous products, where the number of moles and the mean molecular weight of the gaseous products are calculated according to the [H2O–CO2] arbitrary decomposition assumption. Furthermore, the predicted values of the dent depth and the Kamlet–Jacobs method are used to estimate the detonation pressure for 37 explosives. The results show that the pressures obtained on the basis of the dent depth values are in better agreement with experimental/thermochemical code data than the predictions of the Kamlet–Jacobs method.
Similar content being viewed by others
References
A. T. Nielsen, “Caged Polynitramine Compound,” US Patent No. 5 693 794 (1988).
M.-X. Zhang, P. E. Eaton, and R. Gilardi, “Heptaand Octanitrocubanes,” Angew. Chem., Int. Ed. 39 (2), 401–404 (2000).
J. Geith, T. M. Klapötke, J. Weigand, and G. Holl, “Calculation of the Detonation Velocities and Detonation Pressures of Dinitrobiuret (DNB) and Diaminotetrazolium Nitrate (HDAT-NO3),” Propel., Explos., Pyrotech. 29 (1), 3–8 (2004).
H. Gao and J. n. M. Shreeve, “Azole-Based Energetic Salts,” Chem. Rev. 111 (11), 7377–7436 (2011).
D. Fischer, T. M. Klapötke, and J. Stierstorfer, “Oxalylhydrazinium Nitrate and Dinitrate-Efficiency Meets Performance,” J. Energ. Mater. 32 (1), 37–49 (2014).
T. M. Klapötke and T. G. Witkowski, “Covalent and Ionic Insensitive High-Explosives,” Propel., Explos., Pyrotech. 41 (3), 470–483 (2016).
T. M. Klapötke, M. Leroux, P. C. Schmid, and J. Stierstorfer, “Energetic Materials Based on 5,5'-Diamino-4,4'-dinitramino-3,3'-bi-1,2,4-triazole,” Chem.-Asian J. 11 (6), 844–851 (2016).
M. Sućeska, Test Methods for Explosives (Springer-Verlag, New York, 1995).
L. C. Smith, “On Brisance, and a Plate-Denting Test for the Estimation of Detonation Pressure,” Report LADC-6267 (Los Alamos Scientific Laboratory, 1963).
C.L. Mader, Numerical Modeling of Explosives and Propellants (CRC Press, Boca Raton, 2008).
M. Suceska, “Calculation of Detonation Properties by EXPLO5 Computer Program,” Mater. Sci. Forum 465/466, 325–330 (2004).
L. E. Fried, W. M. Howard, and P. C. Souers, CHEETAH 2.0 User’s Manual, UCRL-MA-117541, Rev. 5 (Lawrence Livermore National Laboratory, 1998).
A. Smirnov, D. Lempert, T. Pivina, and D. Khakimov, “Basic Characteristics for Estimation Polynitrogen Compounds Efficiency,” Cent. Eur. J. Energ. Mater. 8 (4), 233–247 (2011).
J. R. Stine, “On Predicting Properties of Explosives-Detonation Velocity,” J. Energ. Mater. 8 (1/2), 41–73 (1990).
M. H. Keshavarz, A. Zamani, and M. Shafiee, “Predicting Detonation Performance of CHNOFCl and Aluminized Explosives,” Propel., Explos., Pyrotech. 39 (5), 749–754 (2014).
M. H. Keshavarz, M. Kamalvand, M. Jafari, and A. Zamani, “An Improved Simple Method for the Calculation of the Detonation Performance of CHNOFCl, Aluminized and Ammonium Nitrate Explosives,” Cent. Eur. J. Energ. Mater. 13 (2), 381–396 (2016).
M. J. Kamlet and S. J. Jacobs, “Chemistry of Detonation. I. A Simple Method for Calculating Detonation Properties of C–H–N–O Explosives,” J. Chem. Phys. 48 (1), 23–35 (1968).
D. R. Hardesty and J. E. Kennedy, “Thermochemical Estimation of Explosive Energy Output,” Combust. Flame 28, 45–59 (1977).
M. J. Kamlet and M. Finger, “An Alternative Method for Calculating Gurney Velocities,” Combust. Flame 34, 213–214 (1979).
J. M. Short, F. H. Helm, M. Finger, and M. J. Kamlet, “The Chemistry of Detonations. VII. A Simplified Method for Predicting Explosive Performance in the Cylinder Test,” Combust. Flame 43, 99–109 (1981).
H. Hornberg and F. Volk, “The Cylinder Test in the Context of Physical Detonation Measurement Methods,” Propel., Explos., Pyrotech. 14 (5), 199–211 (1989).
T. R. Gibbs and P. Popolato, LASL Explosive Property Data, (Univ. of California Press, Berkeley, 1980).
D. Chavez, T. M. Klapötke, D. Parrish, et al., “The Synthesis and Energetic Properties of 3,4-bis(2,2,2-trinitroethylamino)furazan (BTNEDAF),” Propel., Explos., Pyrotech. 39 (5), 641–648 (2014).
M. Göbel and T. M. Klapötke, “Development and Testing of Energetic Materials: The Concept of High Densities Based on the Trinitroethyl Functionality,” Adv. Funct. Mater. 19 (3), 347–365 (2009).
A. Elbeih, J. Pachman, S. Zeman, et al., “Detonation Characteristics of Plastic Explosives Based on Attractive Nitramines with Polyisobutylene and Poly(methyl methacrylate) Binders,” J. Energ. Mater. 30 (4), 358–371 (2012).
J. M. Veauthier, D. E. Chavez, B. C. Tappan, and D. A. Parrish, “Synthesis and Characterization of Furazan Energetics ADAAF and DOATF,” J. Energ. Mater. 28 (3), 229–249 (2010).
N. Fischer, D. Fischer, T. M. Klapötke, et al., “Pushing the Limits of Energetic Materials—the Synthesis and Characterization of Dihydroxylammonium 5,5'-bistetrazole-1,1'-diolate,” J. Mater. Chem. 22 (38), 20418–20422 (2012).
L. Liu, Y. Zhang, S. Zhang, and T. Fei, “Heterocyclic Energetic Salts of 4,4',5,5'-tetranitro-2,2'-biimidazole,” J. Energ. Mater. 33 (3), 202–214 (2015).
A. A. Dippold, M. Feller, and T. M. Klapötke, “5,5'-Dinitrimino-3,3'-methylene-1H-1,2,4-bistriazole—A Metal Free Primary Explosive Combining Excellent Thermal Stability and High Performance,” Cent. Eur. J. Energ. Mater. 8 (4), 261–278 (2011).
NIST Chemistry WebBook. NIST Standard Reference Database Number 69 (National Institute of Standards and Technology, Gaithersburg MD, 20899); http://webbook.nist.gov.
M. J. Kamlet and C. Dickinson, “Chemistry of Detonations. III. Evaluation of the Simplified Calculational Method for Chapman–Jouguet Detonation Pressures on the Basis of Available Experimental Information,” J. Chem. Phys. 48 (1), 43–50 (1968).
B. M. Dobratz and P. C. Crawford, “LLNL Explosives Handbook Properties of Chemical Explosives and Explosive Simulants,” Report No. UCRL-52997-CHG. 2 (Lawrence Livermore National Laboratory, 1985).
M. H. Keshavarz “Theoretical Prediction of Detonation Pressure of CHNO High Energy Materials,” Indian J. Eng. Mater. Sci. 14 (1), 77–80 (2007).
M. Jafari and M. H. Keshavarz, “Simple Approach for Predicting the Heats of Formation of High Nitrogen Content Materials,” Fluid Phase Equilib. 415, 166–175 (2016).
B. M. Rice, S. V. Pai, and J. Hare, “Predicting Heats of Formation of Energetic Materials using Quantum Mechanical Calculations,” Combust. Flame 118 (3), 445–458 (1999).
D. Frem, “Predicting the Plate Dent Test Output in Order to Assess the Performance of Condensed High Explosives,” J. Energ. Mater. 35 (1), 20–28 (2017).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D. Frem.
Published in Fizika Goreniya i Vzryva, Vol. 54, No. 6, pp. 85–94, November–December, 2018.
Rights and permissions
About this article
Cite this article
Frem, D. The Use of the [H2O–CO2] Arbitrary Decomposition Assumption to Predict the Performance of Condensed High Explosives. Combust Explos Shock Waves 54, 704–711 (2018). https://doi.org/10.1134/S0010508218060102
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0010508218060102