Abstract
Purpose: To investigate the difference of in vitro and in vivo grown oocytes, we compared maturation, fertilization, development, and maternal gene expression from both in vitro and in vivo grown mouse oocytes.
Methods: The preantral follicles isolated from 12-day-old mice were cultured on Transwell-COL membrane inserts. After culture, maturation, fertilization, and developmental rates were assessed. RT-PCR (reverse transcription—polymerase chain reaction) was performed to examine the expression of β-actin, GDF-9, and IGF-II in matured oocytes.
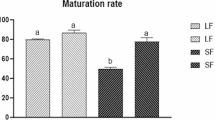
Results: No difference in the nuclear maturation was detected between in vitro and in vivo grown oocytes, but the mean oocyte diameter of the in vitro group was smaller than that of the in vivo group. The fertilization rate was significantly lower in the in vitro group than in the in vivo group (p < 0.05). The capacities of in vitro grown oocyte to cleave and develop to blastocysts were significantly lower than those of the in vivo grown oocytes (p < 0.001). Moreover, blastocyst of in vitro group had fewer total cells than those of in vivo group (p < 0.05). In regards to the expression of genes in mature oocytes, growth differentiation factor-9 (GDF-9) expression was similar between the two groups, but β-actin was significantly reduced in the in vitro group compared to the in vivo group. Particularly, the expression of insulin-like growth factor II (IGF-II) was not found in the in vitro grown oocytes.
Conclusions: These results showed that in vitro grown oocytes did not have the same developmental capacity as in vivo grown oocytes. We assume that the aberrant expression of maternal-derived genes in the in vitro grown oocytes may cause the poor embryo viability.
Similar content being viewed by others
REFERENCES
Gosden RG, Telfer E: Numbers of follicles and oocytes in mammalian ovaries and their allometric relationships. J Zool 1987;211:169–175
Schroeder AC, Eppig JJ: The developmental capacity of mouse oocytes that matured spontaneously in vitro is normal. DevBiol 1984;102:493–497
Vanderhyden BC, Armstrong DT: Role of cumulus cells and serum in the in vitro maturation, fertilization and subsequent development of rat oocytes. Biol Reprod 1989;40:720–728
Staigmiller RB, Moor RM:Effect of follicle cells on the maturation and developmental competence of ovine oocytes matured outside the follicle. Gamete Res 1984;9:221–229
Goto K, Kajihara Y, Kosaka S, Koba M, Nakanishi Y, Ogawa K: Pregnancies after co-culture of cumulus cells with bovine embryos derived from in-vitro fertilization of in-vivo matured follicular oocytes. J Reprod Fertil 1988;83:753–758
Mattioli M, Bacci ML, Galeati G, Seren E: Developmental competence of pig oocytes matured and fertilized in vitro. The-riogenology 1989;31:1201–1207
Cha KY, Choi DH, Koo JJ, Han JJ, Ko JJ, Yoon TK: Pregnancy after in vitro fertilization of human follicular oocytes collected from nonstimulated cycles, their culture in vitro and their transfer in a donor oocyte program. Fertil Steril 1991;55:109–113
Nayudu PL, Osborn SM: Factors influencing the rate of pre-antral and antral growth of mouse ovarian follicles in vitro. J Reprod Fertil 1992;95:349–362.
Boland NI, Gosden RG: Effects of epidermal growth factor on the growth and differentiation of cultured mouse ovarian follicles. J Reprod Fertil 1994;101:369–374
Cortvrindt R, Smitz J, Van Steirteghem AC: In-vitro maturation, fertilization and embryo development of immature oocytes from early preantral follicles from prepuberal mice in a simplified culture system. Hum Reprod 1996;11:2656–2666
Figueiredo JR, Hulshof SCJ, Van den Hurk R, Bevers MM, Nusgens B, Beckers JF: Development of a combined new me-chanical and enzymatic method for the isolation of intact pre-antral follicles from fetal, calf and adult bovine ovaries. Theri-ogenology 1993;40:789–799
Hirao Y, Nagai T, Kubo M, Miyano T, Miyake M, Kato S: In vitro growth and maturation of pig oocytes. J Reprod Fertil 1994;100:333–339
Roy SK, Treacy BJ: Isolation and long-term culture of human preantral follicles. Fertil Steril 1993;59:783–790
Eppig JJ, Schroeder AC: Capacity of mouse oocytes from pre-antral follicles to undergo embryogenesis and development to live young after growth, maturation and fertilization in vitro. Biol Reprod 1989;41:268–276
Spears N, Boland NI, Murray AA, Gosden RG: Mouse oocytes derived from in vitro grown primary ovarian follicles are fertile. Hum Reprod 1994;9:527–532
Schroeder AC, Schultz RM, Kopf GS, Taylor FR, Backer RB, Eppig JJ: Fetuin inhibits zona pellucida hardening and conversion of ZP2 to ZP2f during spontaneous mouse oocyte maturation in vitro in the absence of serum. Biol Reprod;1990;43: 891–897
Hirao Y, Miyano T, Kato S: Fertilization of in vitro grown mouse oocytes. Theriogenology 1990;34:1071–1077
Whitten WK, Biggers JD: Complete development in vitro of the preimplantation stage of the mouse in a simple, chemically defined medium. J Reprod Fertil 1968;17:399–401
Smitz J, Cortvrintz A, Van Steirteghem A: Normal oxygen atmosphere is essential for the solitary long-term culture of early preantral mouse follicles. Mol Reprod Dev 1996;45: 466–475
Eppig JJ, Wigglesworth K: Factors affecting the developmental competence of mouse oocytes grown in vitro oxygen concentration. Mol Reprod Dev 1995;42:447–456
Zuccotti M, Piccinelli A, Giorgi Rossi P, Garagna S, Redi CA: Chromatin organization during mouse oocyte growth. Mol Re-prod Dev 1995;41:479–485
Wickramasinghe D, Ebert KM, Albertini DF: Meiotic competence acquisition is associated with the appearance of M-phase characteristics in growing mouse oocytes. Dev Biol 1991;143:162–172
Hartshorne GM, Sargent IL, Barlow DH: Meiotic progression of mouse oocytes throughout follicle growth and ovulation in vitro. Hum Reprod 1994;9:352–359
Telford NA, Watson AJ, Schultz GA: Transition from maternal to embryonic control in early mammalian development: A comparison of several species. Mol Reprod Dev 1990;26: 90–100
Latham KE, Solter D: Effect of egg composition on the developmental capacity of androgenetic mouse embryos. Development 1991;113:561–568
Chastant S, Christians E, Campion E, Renard JP: Quantitative control of gene expression by nucleocytoplasmic interactions in early mouse embryos: Consequence for reprogramming by nuclear transfer. Mol Reprod Dev 1996;44:423–432
McGrath SA, Esquela SF, Lee S-J: Oocyte-specific expression of growth differentiation factor-9. Mol Endocrinol 1995;9: 131–136
Dong J, Albertini DF, Nishimori K, Rajendra Kumar T, Lu N, Matzuk MM:Growth differentiation factor-9 is required during early ovarian folliculogenesis. Nature 1996;383:531–535
Carabatsos MJ, Elvin J, Matzuk MM, Albertini DF: Characterization of oocyte and follicle development in growth differentiation factor-9-deficient mice. Dev Biol 1998;204:373–384
Hayashi M, McGee EA, Min G, Klein D, Rose UM, Van Duin M, Hsueh AJ: Recombinant growth differentiation factor-9 (GDF-9) enhances growth and differentiation of cultured early ovarian follicles. Endocrinology 1999;140:1236–1244
Harvey MB, Kaye PL: IGF-2 stimulates growth and metabolism of early mouse embryos. Mech Dev 1992;38:169–173
Ueda K, Ganem D: Apoptosis is induced by N-myc expression in hepatocytes, a frequent event in hepadnavirus oncogenesis and is blocked by insulin-like growth factor II. J Virol 1996;70:1375–1383
Stojanov T, Alechna S, O'Neill C: In-vitro fertilization and culture of mouse embryos in vitro significantly retards the on-set of insulin-like growth factor-II expression from the zygotic genome. Mol Hum Reprod 1999;5:116–124
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, DH., Ko, DS., Lee, HC. et al. Comparison of Maturation, Fertilization, Development, and Gene Expression of Mouse Oocytes Grown In Vitro and In Vivo. J Assist Reprod Genet 21, 233–240 (2004). https://doi.org/10.1023/B:JARG.0000042008.83699.cc
Issue Date:
DOI: https://doi.org/10.1023/B:JARG.0000042008.83699.cc