Abstract
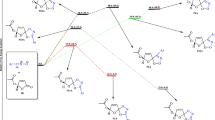
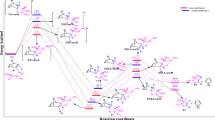
Using quantum-chemical calculations we have carried out an analysis of frontier molecular orbital interactions in the [2+3] cycloaddition reactions of C,C,N-triphenyl- and Z-C,N-diphenylnitrones with β- and α-substituted nitroethylenes. In all of the examples studied the orbital effects lead to the formation of the corresponding 4-nitroisoxazolidines.
Similar content being viewed by others
REFERENCES
A. Markowska, R. Jasinski, and A. Baranski, Czasopismo Techn. PK (Chemia), No. 1, 28 (2003).
K. Fukui, Theory of Orientation and Stereoselection, Springer-Verlag, Berlin (1975).
I. Fleming, Hranicni Orbitaly a Reakce v Organicke Chemii, NTL, Prague(1983).
R. Jasinski, A. Ciezkowska, and A. Baranski, Khim. Geterotsikl. Soedin., 239 (2004).
A. Baranski, R. Jasinski, and M. Bujak, Polish J. Chem., 76, 145 (2002).
R. Jasinski and A. Baranski, Czasopismo Techn. PK (Chemia), No. 4, 1 (2001).
K. N. Houk, A. Bimanand, D. Mukherjee, J Sims, Ch. Yau-Ming, D. C. Kaufman, and L. N. Domelsmith, Heterocycles, 7, 293 (1977).
A. Baranski, R. Jasinski, and K. Zurowski, J. Phys. Org. Chem., 16, 279 (2003).
A. Baranski and R. Jasinski, Khim. Geterotsikl. Soedin., 1670 (2001).
A. Baranski, R. Jasinski, and A. Markowska in Abstract of Papers of the Fifth International Conference. Theoretical and Experimental Backgrounds of Development of New High Performing Chemical Technologies and Equipment, Ivanovo (Russia) (2001), p. 118.
J. J. P. Steward, MOPAC-93 Manual, Fujitsu Limited, Tokyo (1993).
M. J. Dewar, E. G. Zoebisch, E. F. Haely, and J. J. P. Steward, J. Amer. Chem. Soc., 107, 3902 (1985).
R. Sustmann, Tetrahedron Lett., 2117 (1971).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jasinski, R., Ciezkowska, A., Lyubimtsev, A. et al. Synthesis and Properties of Azoles and Their Derivatives. 54. The Regioselectivity of [2+3] Cycloaddition Of C,C,N-Triphenyl- and Z-C,N-Diphenylnitrones With β- and α-Substituted Nitroethylenes in Light of Frontier Molecular Orbital Theory. Chemistry of Heterocyclic Compounds 40, 203–210 (2004). https://doi.org/10.1023/B:COHC.0000027893.83553.c2
Issue Date:
DOI: https://doi.org/10.1023/B:COHC.0000027893.83553.c2