Abstract
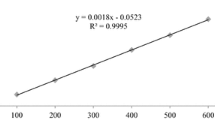
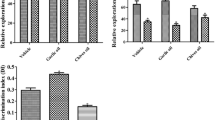
Some compounds derived from plants have been known to possess estrogenic properties and can thus alter the physiology of higher organisms. Genistein and daidzin are examples of these phytoestrogens, which have recently been the subject of extensive research. In this study, genistein and daidzin were found to enhance the acetylcholinesterase (AChE) activity of the rat neuronal cell line PC12 at concentrations as low as 0.08 μM by binding to the estrogen receptor (ER). Results have shown that this enhancement was effectively blocked by the known estrogen receptor antagonist tamoxifen, indicating the involvement of the ER in AChE induction. That genistein and daidzin are estrogenic were confirmed in a cell proliferation assay using the human breast cancer cell line MCF7. This proliferation was also blocked by tamoxifen, again indicating the involvement of the ER. On the other hand, incubating the PC12 cells in increasing concentrations of 17 β-estradiol (E2) did not lead to enhanced AChE activity, even in the presence of genistein or daidzin. This suggests that mere binding of an estrogenic compound to the ER does not necessarily lead to enhanced AChE activity. Moreover, the effect of the phytoestrogens on AChE activity cannot be expressed in the presence of E2 since they either could not compete with the natural ligand in binding to the ER or that E2 down-regulates its own receptor. This study clearly suggests that genistein and daidzin enhance AChE activityin PC12 cells by binding to the ER; however, the actual mechanism of enhancement is not known.
Similar content being viewed by others
References
Barnes S and Peterson TG (1995) Biochemical targets of the isoflavone genistein in tumor cell lines. Proc Soc Exp Biol Med 208: 103–108.
Donger C, Krejci E, Serradell AP, Eymard B, Bon S, Nicole S, Chateau D, Gary F, Fardeau M, Massoulie J and Guicheney P (1998) Mutation in the human acetylcholinesterase-associated collagen gene, COLQ, is responsible for congenital myasthenic syndrome with end-plate acetylcholinesterase deficiency (Type Ic). Am J Human Genetics 63: 967–975.
Greene LA and Rukenstein A (1981) Regulation of acetylcholinesterase activity by nerve growth factor. J Biol Chem 256: 6363–6367.
Greenspan RJ, Finn Jr JA and Hall JC (1980) Acetylcholinesterase mutants in Drosophila and their effects on the structure and function of the central nervous system. J Comp Neurology 189: 741–774.
Honma Y, Okabe KJ, Kasukabe T, Hozumi M, Kajigaya S, Suda T and Miura Y (1991) Induction by some protein kinase inhibitors of differentiation of a mouse megakaryoblastic cell line established by coinjection with Abelson murine leukemia virus and recombinant SV40 retrovirus. Cancer Res 51: 4649–4655.
Lucas CA, Czlonkowska A and Kreutzberg GW (1980) Regulation of acetylcholinesterase by nerve growth factor in the pheochromocytoma PC12 cell line. Neuroscience Lett 18: 333–337.
Martin PM, Horwitz KB, Ryan DS and McGuire WL (1978) Phytoestrogen interaction with estrogen receptors in human breast cancer cells. Endocrinology 103: 1860–1867.
Miksicek RJ (1993) Commonly occurring plant flavonoids have estrogenic activity. Mol Pharmacol 44: 37–43.
Minic J, Molgo J, Karlsson E and Krejci E (2002) Regulation of acetylcholine release by muscarinic receptors at the mouse neuromuscular junction depends on the activity of acetylcholinesterase. Eur J Neuroscience 15: 439–448.
Nobuo K, Lin TS, Fukudome T, Kimura J, Sakamoto T, Kaji R and Shibasaki H (2002) Pathophysiology of weakness in a patient with congenital end-plate acetylcholinesterase deficiency. Muscle & Nerve 25: 585–592.
Okafuji K, Kaku K, Seguchi M, Tanaka H, Azuno Y and Kaneko T (1995) Effects of activin A/erythroid differentiation factor on erythroid and megakaryocytic differentiations of mouse erythroleukemia (friend) cells: Evidence for two distinct modes of cell response. Exp Hematol 23: 210–216.
Pradines A, Magazin M, Schiltz P, LeFur G, Caput D and Ferrara P (1995) Evidence for nerve growth factor-potentiating activities of the nonpeptidic compound SR57746 in PC12 cells. J Neurochem 64: 1954–1964.
Rocch P, Ferreri AM, Simone G, Magrini E, Cavallazzi L and Paolucci G (1995) Inhibitors of protein kinases induce differentiation in human neuroblastoma cell lines. Anticancer Res 15: 1381–1385.
Shepard TH, Pyne GE, Kirschvink JF and McLean M (1960) Soybean goiter: Report of three cases. N Engl J Med 262: 1099–1103.
Sohrabji F, Greene LA, Miranda RC and Toran-Allerand CD (1994) Reciprocal regulation of estrogen and NGF receptors by their ligands in PC12 cells. J Neurobiol 25: 974–988.
Tham DM, Gardner CD and Haskell WL (1998) Potential health benefits of dietary phytoestrogens: A review of the clinical, epidemiological, and mechanistic evidence. J Clin Endocrinol Metab 83: 2223–2235.
Verdeal K and Ryan DS (1979) Naturally-occurring estrogens in plant foodstuffs – A review. J Food Protection 42: 577–583.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Isoda, H., Talorete, T.P.N., Kimura, M. et al. Phytoestrogens genistein and daidzin enhance the acetylcholinesterase activity of the rat pheochromocytoma cell line PC12 by binding to the estrogen receptor. Cytotechnology 40, 117–123 (2002). https://doi.org/10.1023/A:1023903220539
Issue Date:
DOI: https://doi.org/10.1023/A:1023903220539