Abstract
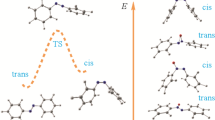
It has been shown by empirical, semiempirical, and nonempirical methods that the interconversion of the rings of 1,3,2-dioxaborinanes goes via transition state with the 2,5-twist form and differs from the monotypic substituted 1,3-dioxanes in having a lower potential barrier.
Similar content being viewed by others
REFERENCES
A. I. Gren' and V. V. Kuznetsov, Chemistry of Cyclic Esters of Boric Acids [in Russian], Naukova Dumka, Kiev (1988), 160 p.
V. V. Kuznetsov, Khim. Obsch. Khim., 69, 417 (1999).
V. V. Kuznetsov, A. I. Gren', A. V. Bogatsky, S. P. Egorova, and V. I. Sidorov, Khim. Geterotsikl. Soed., 26 (1978).
A. I. Gren', V. V. Kuznetsov, and K. S. Zakharov, Khim. Geterotsikl. Soed., 558 (1986).
HyperChem 5.02, Trial version. http://www.hyper.com
M. Salem, Electrons in Chemical Reactions [Russian translation], Mir, Moscow (1985), p. 42.
D. Carton, A. Pontier, M. J. Pouet, J. Soulie, and P. Cadiot, Tetrahedron Letters, 28, 2333 (1975).
N. S. Zefirov, V. A. Palyulin, and E. E. Dashevskaya, J. Phys. Org. Chem., 3, 143 (1990).
B. I. Ionin and B. A. Ershov, NMR Spectroscopy in Organic Chemistry [in Russian], Khimiya, Leningrad (1967), 326 p.
G. E. McAchran and S. G. Shore, Inorg. Chem., 5, 2044 (1966).
A. Finch, P. J. Gardner, and E. J. Pearn, Rec. Trav. Chim., 83, 1314 (1964).
V. J. Orville-Thomas (editor), Internal Rotation of Molecules [Russian translation], Mir, Moscow (1977), p. 355.
M. Anteunis, D. Tavernier, and T. Borremans, Heterocycles, 4, 293 (1976).
V. V. Kuznetsov, Physico-Chemical Inst., Academy of Sciences of Ukraine SSR, Odessa (1983). Deposited in VINITI 14.10.83, No. 5646–83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kuznetsov, V.V., Novikov, A.N. Pseudorotation and Barriers to Inversion of 1,3,2-Dioxaborinane Rings. Chemistry of Heterocyclic Compounds 39, 263–266 (2003). https://doi.org/10.1023/A:1023788912799
Issue Date:
DOI: https://doi.org/10.1023/A:1023788912799