Abstract
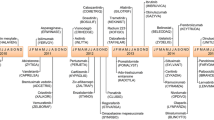
Toxicity is a major concern for anticancer drugs. These compounds present a narrow therapeutic index, with a small difference between the dose required for an antitumor effect and that responsible for unacceptable toxicity. Their recommended doses are determined according to the toxicity endpoint. Moreover, toxicity is observed earlier than the therapeutic effect, so, toxic effects represent a major endpoint for pharmacodynamic studies of cytotoxic drugs. Knowledge of toxicity patterns and main factors of toxicity of anticancer drugs is required before modeling data of these studies. Hematological toxicities represent the main toxicity of the cytotoxic. However, non-hematological toxicities have become more important than hematological toxicities as pharmacodynamic endpoints in some circumstances such as high-dose chemotherapy associated with bone marrow transplantation. This paper will describe the main toxicity of the cytotoxic drugs, and its factors of both inter- and intra-patient variability. The toxicity pattern of topotecan will be examined as an example. Knowledge of the toxicity pattern of a drug constitutes a prerequirement before modeling its pharmacodynamics.
Similar content being viewed by others
References
Hryniuk WM: Is more better? J Clin Oncol 4: 621–622, 1986.
Alberts DS, Dorr RT: New perspectives on an old friend: optimizing carboplatin for the treatment of solid tumors. Oncologist 3: 15–34, 1998
Gonzales-Vitale JC, Hayes DM, Cvitkovic E, Sternberg SS: The renal pathology in clinical trials of cis-platinum (II) diamminedichloride. Cancer 39: 1362–1371, 1977
Robert J: Clinical pharmacokinetics of epirubicin. Clin Pharmacokinet 26: 428–438, 1994
Schneider DT, Hilgenfeld E, Schwabe D, Behnisch W, Zoubek A, Wessalowski R, Gobel U: Acute myelogenous leukemia after treatment for malignant germ cell tumors in children. J Clin Oncol 17: 3226–3233, 1999
Macdonald JS: Toxicity of 5–fluorouracil. Oncology (Huntingt) 13: 33–34, 1999
Ratain MJ, Schilsky RL, Conley BA, Egorin MJ: Pharmacodynamics in cancer therapy. J Clin Oncol 8: 1739–1753, 1990
Drach D, Zhao S, Drach J, Mahadevia R, Gattringer C, Huber H, Andreeff M: Subpopulations of normal peripheral blood and bone marrow cells express a functional multidrug resistant phenotype. Blood 80: 2729–2734, 1992
Baum C, Peinert S, Carpinteiro A, Eckert HG, Fairbairn LJ: Genetic modification of haematopoietic cells for combined resistance to podophyllotoxins, other agents covered by MDR1–mediated efflux activity and nitrosoureas. Bone Marrow Transplant 25(suppl 2): S71–S74, 2000
Otsuki T, Sakaguchi H, Yamada O, Yawata Y, Ueki A: Effect of the nitrosourea anti-tumor chemotherapeutical agent MCNU on five human myeloma cell lines. Oncol Rep 5: 827–832, 1998
Schein PS, Bull JM, Doukas D, Hoth D: Sensitivity of human and murine hematopoietic precursor cells to 2–[3–(2–chloroethyl)-3–nitrosoureido]-D-glucopyranose and 1,3–bis(2–chloroethyl)-1–nitrosourea. Cancer Res 38: 257–260, 1978
Goldwasser F, Buthaud X, Gross M, Bleuzen P, Cvitkovic E, Voinea A, Jasmin C, Romain D, Misset JL: Decreased topotecan platelet toxicity with successive topotecan treatment cycles in advanced ovarian cancer patients. Anticancer Drugs 10: 263–265, 1999
Verwey J, de Vries J, Pinedo HM: Mitomycin C-induced renal toxicity, a dose-dependent side effect? Eur J Cancer Clin Oncol 23: 195–199, 1987
Pivot X, Guardiola E, Etienne M, Thyss A, Foa C, Otto J, Schneider M, Magne N, Bensadoun RJ, Renee N, Milano G: An analysis of potential factors allowing an individual prediction of cisplatin-induced anaemia. Eur J Cancer 36: 852–857, 2000
Pujol JL, Douillard JY, Riviere A, Quoix E, Lagrange JL, Berthaud P, Bardonnet-Comte M, Polin V, Gautier V, Milleron B, Chomy F, Chomy P, Spaeth D, Le Chevalier T: Dose-intensity of a four-drug chemotherapy regimen with or without recombinant human granulocyte-macrophage colony-stimulating factor in extensive-stage small-cell lung cancer: a multicenter randomized phase III study. J Clin Oncol 15: 2082–2089, 1997
Tjan-Heijnen VC, Postmus PE, Wagener DJ: Dose intensification of chemotherapy and the role of granulocyte colony stimulating factor and granulocyte macrophage colony stimulating factor in small cell lung cancer. Anticancer Drugs 8: 549–564, 1997
Newell DR: Pharmacokinetic determinants of the activity and toxicity of antitumour agents. Cancer Surv 8: 557–603, 1989
Egorin MJ, Reyno LM, Canetta RM, Jodrell DI, Swenerton KD, Pater JL, Burroughs JN, Novak MJ, Sridhara R: Modeling toxicity and response in carboplatin-based combination chemotherapy. Semin Oncol 21: 7–19, 1994
Canal P, Chatelut E, Guichard S: Practical treatment guide for dose individualisation in cancer chemotherapy. Drugs 56: 1019–1038, 1998
Gianni L, Kearns CM, Giani A, Capri G, Vigano L, Lacatelli A, Bonadonna G, Egorin MJ: Nonlinear pharmacokinetics and metabolism of paclitaxel and its pharmacokinetic/pharmacodynamic relationships in humans. J Clin Oncol 13: 180–190, 1995
Cvitkovic E, Bekradda M: Oxaliplatin: a new therapeutic option in colorectal cancer. Semin Oncol 26: 647–662, 1999
Pignon T, Lacarelle B, Duffaud F, Guillet P, Catalin J, Durand A, Monjanel S, Favre R: Pharmacokinetics of high-dose methotrexate in adult osteogenic sarcoma. Cancer Chemother Pharmacol 33: 420–424, 1994
Delepine N, Delepine G, Cornille H, Brion F, Arnaud P, Desbois JC: Dose escalation with pharmacokinetics monitoring in methotrexate chemotherapy of osteosarcoma. Anticancer Res 15: 489–494, 1995
Calvert H, Judson I, Van der Vijgh WJ: Platinum complexes in cancer medicine: pharmacokinetics and pharmacodynamics in relation to toxicity and therapeutic activity. Cancer Surv 17: 189–217, 1993
Perdaems N, Bachaud JM, Rouzaud P, Murris-Espin M, Hermant C, Mihura J, Lochon I, Houin G, Canal P, Chatelut E: Relation between unbound plasma concentrations and toxicity in a prolonged oral etoposide schedule. Eur J Clin Pharmacol 54: 677–683, 1998
Goa KL, Faulds D: Vinorelbine. A review of its pharmacological properties and clinical use in cancer chemotherapy. Drugs Aging 5: 200–234, 1994
Balis FM, Holcenberg JS, Bleyer WA: Clinical pharmacokinetics of commonly used anticancer drugs. Clin Pharmacokinet 8: 202–232, 1983
Peters GJ, Schornagel JH, Milano G: A. Clinical pharmacokinetics of anti-metabolites. Cancer Surv 17: 123–156, 1993
Schellens JH, Planting AS, Ma J, Maliepaard M, de Vos A, de Boer DM, Verweij J: Adaptive intrapatient dose escalation of cisplatin in patients with advanced head and neck cancer. Anticancer Drugs 12: 667–675, 2001
Joel SP, Shah R, Clark PI, Slevin ML: Predicting etoposide toxicity: relationship to organ function and protein binding. J Clin Oncol 14: 257–267, 1996
Stewart CF: Use of etoposide in patients with organ dysfunction: pharmacokinetic and pharmacodynamic considerations. Cancer Chemother Pharmacol 34(suppl): S76–S83, 1994
Reyno LM, Egorin MJ, Canetta RM, Jodrell DI, Swenerton KD, Pater JL, Burroughs JN, Novak MJ, Sridhara R: Impact of cyclophosphamide on relationships between carboplatin exposure and response or toxicity when used in the treatment of advanced ovarian cancer. J Clin Oncol 11: 1156–1164, 1993
Belani CP, Kearns CM, Zuhowski EG, Erkmen K, Hiponia D, Zacharski D, Engstrom C, Ramanathan RK, Capozzoli MJ, Aisner J, Egorin MJ: Phase I trial, including pharmacokinetic and pharmacodynamic correlations, of combination paclitaxel and carboplatin in patients with metastatic non-small-cell lung cancer. J Clin Oncol 17: 676–684, 1999
Calvert AH, Ghokul S, Al Azraqi A, Wright J, Lind M, Bailey N, Highley M, Siddiqui N, Lunec J, Sinha D, Boddy A, Roberts T, Fenwick J: Carboplatin and paclitaxel, alone and in combination: dose escalation, measurement of renal function, and role of the p53 tumor suppressor gene. Semin Oncol 26: 90–94, 1999
Bertrand R, O'Connor PM, Kerrigan D, Pommier Y: Sequential administration of camptothecin and etoposide circumvents the antagonistic cytotoxicity of simultaneous drug administration in slowly growing human colon carcinoma HT-29 cells. Eur J Cancer 28A: 743–748, 1992
Fischel JL, Etienne MC, Formento P, Milano G: Search for the optimal schedule for the oxaliplatin/5–fluorouracil association modulated or not by folinic acid: preclinical data. Clin Cancer Res 4: 2529–2535, 1998
Gauvin A, Bressolle F, Martineau P, Astre C, Pinguet F: In vitro schedule-dependent interaction between irinotecan and vinorelbine in NCI H460 non-small cell lung cancer cell line. Anticancer Res 22: 905–912, 2002
Perez EA, Buckwalter CA: Sequence-dependent cytotoxicity of etoposide and paclitaxel in human breast and lung cancer cell lines. Cancer Chemother Pharmacol 41: 448–452, 1998
McLeod HL: Clinically relevant drug-drug interactions in oncology. Br J Clin Pharmacol, 45: 539–544, 1998
Balis FM: Pharmacokinetic drug interactions of commonly used anticancer drugs. Clin Pharmacokinet 11: 223–235, 1986
Kohne CH, Grothey A, Bokemeyer C, Bontke N, Aapro M: Chemotherapy in elderly patients with colorectal cancer. Ann Oncol 12: 435–442, 2001
Tirelli U, Aapro M, Obrist R, Festen J, Scheider M, Fentiman I, Monfardini S: Cancer treatment and old people. Lancet 338: 114, 1991
Knoester PD, Underberg WJ, Beijnen JH: Clinical pharmacokinetics and pharmacodynamics of anticancer agents in pediatric patients (review). Anticancer Res 13: 1795–1808, 1993
Capizzi RL, Oster W: Protection of normal tissue from the cytotoxic effects of chemotherapy and radiation by amifostine: clinical experiences. Eur J Cancer 31A(suppl 1): S8–S13, 1995
Evans WE, Relling MV: Pharmacogenomics: translating functional genomics into rational therapeutics. Science 286: 487–491, 1999
Diasio RB, Johnson MR: The role of pharmacogenetics and pharmacogenomics in cancer chemotherapy with 5–fluorouracil. Pharmacology 61: 199–203, 2000
Levi F, Brienza S, Metzer G, Depres-Brummer P, Bertheault-Cvitkovic F, Zidani R, Adam R, Misset JL: Implications of chronobiology for 5–fluorouracil (5–FU) efficacy. Adv Exp Med Biol 339: 169–183, 1993
Levi F, Metzger G, Massari C, Milano G: Oxaliplatin: pharmacokinetics and chronopharmacological aspects. Clin Pharmacokinet 38: 1–21, 2000
Canal P, Sqalli A, de Forni M, Chevreau C, Pujol A, Bugat R, Roche H, Oustrin J, Houin G: Chronopharmacokinetics of doxorubicin in patients with breast cancer. Eur J Clin Pharmacol 40: 287–291, 1991
Hrushesky WJ: Circadian timing of cancer chemotherapy. Science 228: 73–75, 1985
Kerr DJ, Lewis C, O'Neil B, Lawson N, Blackie RG, Newell DR, Boxall F, Cox J, Rankin EM, Kaye SB: The myelotoxicity of carboplatin is influenced by the time of its administration. Hematol Oncol 8: 59–63, 1990
Smaaland R: Circadian rhythm of cell division. Prog Cell Cycle Res 2: 241–266, 1996
Dennis MJ, Beijnen JH, Grochow LB, van Warmerdam LJ: An overview of the clinical pharmacology of topotecan. Semin Oncol 24: S5–S5, 1997
Kollmannsberger C, Mross K, Jakob A, Kanz L, Bokemeyer C: Topotecan — A novel topoisomerase I inhibitor: pharmacology and clinical experience. Oncology 56: 1–12, 1999
Hochster H, Liebes L, Speyer J, Sorich J, Taubes B, Oratz R, Wernz J, Chachoua A, Blum RH, Zeleniuch-Jacquotte A: Effect of prolonged topotecan infusion on topoisomerase 1 levels: a phase I and pharmacodynamic study. Clin Cancer Res 3: 1245–1252, 1997
Gerrits CJ, Schellens JH, Burris H, Eckardt JR, Planting AS, van der Burg ME, Rodriguez GI, Loos WJ, van BV, Hudson I, Von Hoff DD, Verweij J: A comparison of clinical pharmacodynamics of different administration schedules of oral topotecan (Hycamtin). Clin Cancer Res 5: 69–75, 1999
Gerrits CJ, Burris H, Schellens JH, Planting AS, van den Burg ME, Rodriguez GI, van BV, Loos WJ, Hudson I, Fields S, Verweij J, Von Hoff DD: Five days of oral topotecan (Hycamtin), a phase I and pharmacological study in adult patients with solid tumours. Eur J Cancer 34: 1030–1035, 1998
Schellens JH, Creemers GJ, Beijnen JH, Rosing H, Boer-Dennert M, McDonald M, Davies B, Verweij J: Bioavailability and pharmacokinetics of oral topotecan: a new topoisomerase I inhibitor. Br J Cancer 73: 1268–1271, 1996
Jonker JW, Smit JW, Brinkhuis RF, Maliepaard M, Beijnen JH, Schellens JH, Schinkel AH: Role of breast cancer resistance protein in the bioavailability and fetal penetration of topotecan. J Natl Cancer Inst 92: 1651–1656, 2000
Rowinsky EK, Kaufmann SH, Baker SD, Grochow LB, Chen TL, Peereboom D, Bowling MK, Sartorius SE, Ettinger DS, Forastiere AA, Donehower RC: Sequences of topotecan and cisplatin: phase I, pharmacologic, and in vitro studies to examine sequence dependence. J Clin Oncol 14: 3074–3084, 1996
O'Dwyer PJ, LaCreta FP, Haas NB, Halbherr T, Frucht H, Goosenberg E, Yao KS: Clinical, pharmacokinetic and biological studies of topotecan. Cancer Chemother Pharmacol 34(suppl): S46–S52, 1994
Grochow LB, Rowinsky EK, Johnson R, Ludeman S, Kaufmann SH, McCabe FL, Smith BR, Hurowitz L, DeLisa A, Donehower RC, Noe DA: Pharmacokinetics and pharmacodynamics of topotecan in patients with advanced cancer. Drug Metab Dispos 20: 706–713, 1992
O'Reilly S, Rowinsky EK, Slichenmyer W, Donehower RC, Forastiere AA, Ettinger DS, Chen TL, Sartorius S, Grochow LB: Phase I and pharmacologic study of topotecan in patients with impaired renal function. J Clin Oncol 14: 3062–3073, 1996
Gallo JM, Laub PB, Rowinsky EK, Grochow LB, Baker SD: Population pharmacokinetic model for topotecan derived from phase I clinical trials. J Clin Oncol 18: 2459–2467, 2000
Montazeri A, Boucaud M, Lokiec F, Pinguet F, Culine S, Deporte-Fety R, Albin N, Laguerre B, Goupil A, Bugat R, Canal P, Chatelut E: Population pharmacokinetics of topotecan: intraindividual variability in total drug. Cancer Chemother Pharmacol 46: 375–381, 2000
Zamboni WC, Houghton PJ, Johnson RK, Hulstein JL, Crom WR, Cheshire PJ, Hanna SK, Richmond LB, Luo X, Stewart CF: Probenecid alters topotecan systemic and renal disposition by inhibiting renal tubular secretion. J Pharmacol Exp Ther 284: 89–94, 1998
Rosing H, van Zomeren DM, Doyle E, ten Bokkel WW, Schellens JH, Bult A, Beijnen JH: Quantification of topotecan and its metabolite N-desmethyltopotecan in human plasma, urine and faeces by high-performance liquid chromatographic methods. J Chromatogr B Biomed Sci Appl 727: 191–203, 1999
Rosing H, van Zomeren DM, Doyle E, Bult A, Beijnen JH: O-glucuronidation, a newly identified metabolic pathway for topotecan and N-desmethyl topotecan. Anticancer Drugs 9: 587–592, 1998
Zamboni WC, Gajjar AJ, Heideman RL, Beijnen JH, Rosing H, Houghton PJ, Stewart CF: Phenytoin alters the disposition of topotecan and N-desmethyl topotecan in a patient with medulloblastoma. Clin Cancer Res 4: 783–789, 1998
Montazeri A, Culine S, Laguerre B, Pinguet F, Lokiec F, Albin N, Goupil A, Deporte-Fety R, Bugat R, Canal P, Chatelut E: Individual adaptive dosing of topotecan in ovarian cancer. Clin Cancer Res 8: 394–399, 2002
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chatelut, E., Delord, JP. & Canal, P. Toxicity Patterns of Cytotoxic Drugs. Invest New Drugs 21, 141–148 (2003). https://doi.org/10.1023/A:1023565227808
Issue Date:
DOI: https://doi.org/10.1023/A:1023565227808