Abstract
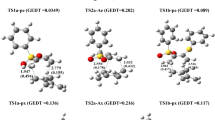
The relative reactivity (substrate selectivity) of five-membered heterocycles on electrophilic substitution (pyrrole >> furan > selenophene > thiophene) and their positional selectivity (furan > selenophene ≥ thiophene > pyrrole) are not consistent. The indicated differences in positional selectivity (α : β ratios) of the parent heterocycles show up essentially in orientation on electrophilic substitution reactions of their derivatives and the corresponding benzannelated systems. It was shown that the positional selectivity was reduced in a sequence corresponding to the change in relative stability of the onium states of the elements (O+ < Se+ ≤ S+ < N+) and reflects the predominant role of the heteroatoms in stabilizing the σ-complexes formed on β-substitution, in which the positive charge is distributed between the heteroatom and one α-carbon atom (in α -isom ers four atoms of the ring participate in delocalization of the charge). This interpretation was confirmed by quantum-chemical calculations carried out by the RHF/6-31G(d), MP2/6-31G(d)//RHF(6)-31G(d), and B3LYP/6-31G(d) ab initio methods.
Similar content being viewed by others
REFERENCES
L. I. Belen'kii, 16th Intern. Congr. Heterocycl. Chem., Abstracts, OP-V-20, Aug. 10-15, 1997, Montana State Univ. Boseman, USA.
L. I. Belen'kii, All-Russian Symposium on Chemistry of Organic Compounds of Silicon and Sulfur, Irkutsk, 3-6 Dec. 2001, Abstracts, 2001, p. 83.
L. I. Belen'kii,I. A. Suslov, and N. D. Chuvylkin, Practical Scientific Conference on the Current Problem of the Introduction of Chemical Processes into Agriculture, Tashkent, 24-26 September 2002, Abstracts, 2002, p. 6.
G. Marino, Adv. Heterocycl. Chem., 13, 235 (1971)
G. Marino, Khim. Geterotsikl. Soedin., 579 (1973).
L. I. Belen'kii, III Intern. Symp. on Furan Chemistry, Coll. of Papers, Smolenice, Czechoslovakia, 1979, p. 4.
L. I. Belen'kii, Khim. Geterotsikl. Soedin., 1587 (1980).
C. F. Candy,R. A. Jones, and P. H. Wright, J. Chem. Soc. C, 2563 (1970).
R. X. Xu,H. J. Anderson,N. J. Gogan,C. A. Loader, and R. McDonald, Tetrahedron Lett., 22, 4899 (1981).
P. Linda and G. Marino, J. Chem. Soc. (B), 392 (1968).
H. J. Anderson and S.-F. Lee, Can. J. Chem., 43, 409 (1965).
Ya. L. Gol'dfarb,Yu. B. Vol'kenshtein, and B. V. Lopatin, Zh. Obshch. Khim., 34, 969 (1964).
Z. N. Nazarova, Zh. Obshch. Khim., 24, 575 (1954).
E. Baum,L. I. Belen'kii,V. G. Kul'nevich, and T. E. Goldovskaya, Khim. Geterotsikl. Soedin., 662 (1982).
H. J. Anderson and L. C. Hopkins, Can. J. Chem., 42, 1279 (1964).
H. J. Anderson and L. C. Hopkins, Can. J. Chem., 44, 1831 (1966).
H. J. Anderson and C. W. Huang, Can. J. Chem., 45, 897 (1967).
C. E. Loader and H. J. Anderson, Tetrahedron, 25, 3879 (1969).
J. K. Groves,H. J. Anderson, and H. Nagy, Can. J. Chem., 49, 2427 (1971).
C. Jaureguiberry,M. C. Fournier-Zaluski,J. P. Chevallier, and B. Roques, Compt. Rend., 273, 276 (1971).
P. Fournari,M. Farnier, and C. Fournier, Bull. Soc. Chim. France, 283 (1972).
H. J. Anderson,C. R. Ricke,T. G. Costello,C. T. Loader, and G. H. Barnett, Can. J. Chem., 56, 654 (1978).
P. Barker,P. Gendler, and H. Rappoport, J. Org. Chem., 43, 4849 (1978).
Ya. L. Gol'dfarb and Yu. B. Vol'kenshtein, Dokl. Akad. Nauk SSSR, 128, 536 (1959).
S. G. Mairanovskii,N. V. Barashkova, and Yu. B. Vol'kenshtein, Izv. Akad. Nauk SSSR. Ser. Khim., 1539 (1965).
L. I. Belen'kii,Ya. L. Gol'dfarb, and G. P. Gromova, Izv. Akad. Nauk SSSR. Ser. Khim., 2733 (1973).
Ya. L. Gol'dfarb,A. P. Yakubov, and L. I. Belen'kii, Dokl. Akad. Nauk SSSR, 185, 941 (1969).
L. I. Belen'kii,I. B. Karmanova, and Ya. L. Gol'dfarb, Zh. Org. Khim., 7, 1743 (1971).
Ya. L. Gol'dfarb,I. B. Karmanova,Yu. B. Vol'kenshtein, and L. I. Belen'kii, Khim. Geterotsikl. Soedin., 1474 (1978).
Ya. L. Gol'dfarb,E. I. Novikova, and L. I. Belen'kii, Izv. Akad. Nauk SSSR. Ser. Khim., 1233 (1971).
Ya. L. Gol'dfarb,E. I. Novikova, and L. I. Belen'kii, Izv. Akad. Nauk SSSR. Ser. Khim., 2841 ( (1971).
L. I. Belen'kii,E. I. Novikova, and Ya. L. Gol'dfarb, Khim. Geterotsikl. Soedin., 1353 (1971).
D. M. Antonov,L. I. Belen'kii, and S. Gronowitz, J. Heterocycl. Chem., 32, 53 (1995).
G. Hart,L. D. R. Liljegren, and K. T. Pitts, J. Chem. Soc., 4267 (1961)
S. Clementi,P. Linda, and G. Marino, J. Chem. Soc. (B), 79 (1971).
Tran Quang Minh,F. Mantovani,P. Faller,L. Christiaens, and M. Renson, Bull. Soc. Chim. France, 3955 (1972).
G. Olah,K. K. Laali,Q. Wang, and G. K. S. Prakash, Onium Ions, Wiley, New York (1998), p. 509.
H. Meerwein in Houben-Weyl Methoden der Organischen Chemie, 4th Edition, Vol. VI, Part 3, G. Thieme Verlag, Stuttgart (1965), p. 327.
J. Goerdeler, in Houben-Weyl Methoden der Organischen Chemie, 4th Edition, Vol. IX, G. Thieme Verlag, Stuttgart (1955), p. 174.
H. Reinboldt, in Houben-Weyl Methoden der Organischen Chemie, 4th Edition, Vol. IX, G. Thieme Verlag, Stuttgart (1955), p. 917.
H. Meerwein, J. Prakt. Chem. [2], 154, 98 (1939).
L. I. Belen'kii and I. A. Abronin, Zh. Org. Khim., 17, 1129 (1981).
M. J. Frisch,G. W. Trucks,H. B. Schlegel,P. M. W. Gill,B. Johnson,M. A. Robb,J. R. Cheeseman,T. Keith,G. A. Petersson,J. A. Montgomery,K. Raghavachari,M. A. Al-Laham,V. G. Zakrzewski,J. V. Ortiz,J. B. Foresman,J. Cioslowski,B. B. Stefanov,A. Nanayakkara,M. Challacombe,C. Y. Peng,P. Y. Ayala,W. Chen,M. W. Wong,J. L. Andres,E. S. Replogle,R. Gomperts,R. L. Martin,D. J. Fox,J. C. Binkley,D. J. Defrees,J. Baker,J. P. Stewart,M. Head-Gordon,C. Gonzalez, and J. A. Pople, Gaussian 94, Revision E. 1, Gaussian Inc., Pittsburgh PA (1995).
A. R. Katritzky and A. F. Pozharskii, Handbook of Heterocyclic Chemistry, 2nd Edition, Pergamon, Amsterdam (2000), p. 61.
J. B. Foresman and A. E. Frish, Exploring Chemistry with Electronic Structure Methods, 2nd Edition, Gaussian Inc., Pittsburgh PA (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Belen'kii, L.I., Suslov, I.A. & Chuvylkin, N.D. Substrate and Positional Selectivity in Electrophilic Substitution Reactions of Pyrrole, Furan, Thiophene, and Selenophene Derivatives. Chemistry of Heterocyclic Compounds 39, 36–48 (2003). https://doi.org/10.1023/A:1023060406534
Issue Date:
DOI: https://doi.org/10.1023/A:1023060406534