Abstract
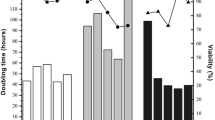
The recombinant a and bsubunits for human coagulation factor XIII were transfected into Chinese hamster ovary (CHO) cells. CHO cells were amplified and selected with methotrexate in adherent cultures containing serum, and CHO 1-62 cells were later selected in protein-free medium. To develop a recombinant factor XIII production process in a suspension culture, we have investigated the growth characteristics of CHO cells and the maintenance of factor XIII expression in the culture medium. Suspension adaptation of CHO cells was performed in protein-free medium, GC-CHO-PI, by two methods, such as serum weaning and direct switching from serum containing media to protein-free media. Although the growth of CHO cells in suspension culture was affected initially by serum depletion, cell specific productivity of factor XIII showed only minor changes by the direct switching to protein-free medium during a suspension culture. As for the long-term stability of factor XIII, CHO 1-62 cells showed a stable expression of factor XIII in protein-free condition for 1000 h. These results indicate that the CHO 1-62cells can be adapted to express recombinant human factor XIII in a stable maimer in suspension culture using a protein-free medium. Our results demonstrate that enhanced cell growth in a continuous manner is achievable for factor XIII production in a protein-free medium when a perfusion bioreactor culture system with a spin filter is employed.
Similar content being viewed by others
References
Bekkari H., Sekkat D., Straczek J., Hess K., Belleville-Nabet F. and Nabet P. 1994. Expression of secreted recombinant human insulin-like growth factor-II (IGF-II) in Chinese hamster ovary cells. J. Biotechnol. 36: 75–83.
Boedeker B.G.D. 1992. The manufacturing of recombinant factor VIII - KOGENATE. Transfusion Med. Rev. 6: 256–260.
Bröker M., Bäuml O., Göttig A., Ochs J., Bodenbenner M. and Amann E. 1991. Expression of the human blood coagulation protein factor XIII in Saccharomyces cerevisia: dependence of the expression levels from host-vector systems and medium conditions. Appl. Microbiol. Biotechnol. 34: 756–764.
Castro P.M.L., Ison A.P., Hayter P.M. and Bull A.T. 1995. The macroheterogeneity of recombinant human interferon-r produced by Chinese hamster ovary cells is affected by the protein and lipid content of the culture medium. Biotechnol. Appl. Biochem. 21: 87–100.
Gandor C., Leis C., Fiechter A. and Asselbergs F.A.M. 1995. Amplification and expression of recombinant genes in serum-independent Chinese hamster ovary cells. FEBS Letters 377: 290–294.
Gray P.P., Crowley J.M. and Marsden W.L. 1990. Growth of a recombinant Chinese hamster ovary cell line and high-level expression in protein-free medium. In: Murakami H. (ed.), Trends in Animal Cell Culture Technology. Kodansha Ltd., Tokyo, pp. 265–270.
Hayter P.M., Curling E.M.A., Baines A.J., Jenkins N., Salmon I., Strange P.G. et al. 1992. Glucose-limited chemostat culture of Chinese hamster ovary cells producing recombinant human interfirron-γ. Biotechnol. Bioeng. 39: 327–335.
Hoyer L.W. 1981. The factor VIII complex: structure and function. Blood 58: 1–13.
Ichinose A., McMullen B.A., Fujikawa K. and Davie B.W. 1986. Amino acid sequence of the b subunit of human factor XIII, a protein composed of ten peptide segments. Biochemistry 25: 4633–4638.
Jin B.R., Ryu C.J., Park S.S., Uk N.G., Hong H.J. and Han M.I. 1993. Cloning, expression and characterization of a murine-human chimeric antibody wish specificity for pre-S2 surface antigen of Hepatitis B virus. Mole. Immuno. 30: 1647–1654.
Kaczmarek E., Liu Y., Berse B., Chen C.S. and McDonagh J. 1995. Biosynthesis of plasma factor XIII: evidence for transcription and translation in hepatoma cells. Biochim. Biophys. Acta. 1247: 127–134.
Kaetsu H., Hashiguchi T., Foster D. and Ichinose A. 1996. Expression and release of the a and b subunits for human coagulation factor XIII in Baby Hamster Kidney (BHK) cells. J. Biochem. 119: 961–969.
Kaufman R.J., Wasley L.C. and Dorner A.J. 1988. Synthesis, processing, and secretion of recombinant human factor VIII expressed in mammalian cells. J. Biol. Chem. 263: 6352–6362.
Kaufman R.J., Wasley L.C., Spiliotes A.J., Goggels S.D., Latt S.A., Larsen G.R. et al. 1985. Coamplification and coexpression of human tissue-type plasminogen activator and murine dihydrofolate reductase sequences in Chinese hamster ovary cells. Mole. Cell. Biol. 5: 1750–1759.
Koehler U., Abken H., Grummt F., Wienberg J. and Weidle U.H. 1995. A novel type of unstable homogeneously staining region with a head-to-tail arrangement: spontaneous decay and reintegration of DNA elements into a plethora of new chromosomal sites. Cytogenet. Cell. Genet. 68: 33–38.
Konstantinov K.B., Tsai Y.-S., Moles D. and Matanguihan R. 1996. Control of long-term perfusion Chinese hamster ovary cell culture by glucose Auxostat. Biotechnol. Prog. 12: 100–109.
Lee K.H., Sburlati A., Renner W.A. and Bailey J.E. 1996. Deregulated expression of cloned transcription factor E2F-1 in Chinese hamster ovary cells shifts protein patterns and activates growth in protein-free medium. Biotechnol. Bioeng. 50: 273–279.
Linz M., Zeng A.-P., Wagner R. and Deckwer W.-D. 1997. Stoi-chiometry, kinetics, and regulation of glucose and amino acid metabolism of a recombinant BHK cell line in batch and continu-ous cultures. Biotechnol. Pro. 13: 453–463.
Lucas B.K., Giere L.M., DeMarco R.A., Shen A., Chisholm V. and Crowley C.W. 1996. High-level production of recombinant proteins in CHO cells using a dicistronic DHFR intron expression vector. Nucleic Acids Res. 24: 1774–1779.
Miele M., Bonatti S., Menichini P., Ottaggio L. and Abbondandolo A. 1989. The presence of amplitied regions affects the stability of chromosomes in drug-resistant Chinese hamster cells. Mutation. Res. 219: 171–178.
Renner W.A., Lee K.H., Hatzimanikatis V., Bailey J.E. and Eppen-berger H.M. 1995. Recombinant cyclin E expression activates proliferation and obviates surface attachment of Chinese hamster ovary (CHO) cells in protein-tree medium. Biotechnol. Bioeng. 47: 476–482.
Ruiz J.C. and Wahl G.H. 1990. Chromosomal destabilization during gene amplification. Mole. Cell. Biol. 10: 3056–3066.
Schwartz M.L., Pizzo S.V., Hill R.L. and Mckee P.A. 1973. Human factor XIII from plasma and platelets. J. Biol. Chem. 248: 1395–1407.
Searles J.A., Todd P. and Kompala D.S. 1994. Viable cell recycle with an inclined settler in the perfusion cultures of suspended recombinant Chinese hamster ovary cells. Biotechnol. Bioeng. 10: 198–206.
Sinacore M.S., Charlebois T.S., Harrison S., Brennan S., Richards T., Hamilton M. et al. 1996. CHO DUKX cell lineages pre-adaptated to growth in serum-free suspension culture enable rapid development of cell culture processes for the manufacture of recombinant proteins. Biotechnol. Bioeng. 52: 518–528.
Tharaud C., Ribet A.-M., Costes C. and Gaillardin C. 1992. Secretion of human blood coagulation factor XIlIa by the yeast Yarrowia lipolytica. Gene. 121: 111–119.
Tonouchi N., Koyama N. and Miwa K. 1992. A CHO strain producing high-level human IL-6 with the 39 deletion construct. J. Biotechnol. 22: 283–293.
Zang M., Trautrnwrn H., Gandor C., Messi F., Asselhergs F., Leist C. et al. 1995. Production of recombinant proteins in Chinese hamster ovary cells using a protein-free cell culture medium. Bio/Technology 13: 389–392.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chun, BH., Bang, WG., Park, YK. et al. Stable expression of recombinant human coagulation factor XIII in protein-free suspension culture of Chinese hamster ovary cells. Cytotechnology 37, 179–187 (2001). https://doi.org/10.1023/A:1020555918441
Issue Date:
DOI: https://doi.org/10.1023/A:1020555918441