Abstract
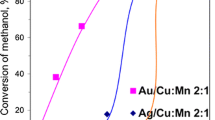
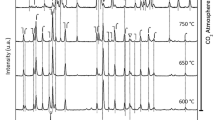
La2CuO4 is an active catalyst for the reduction of NO by CO. Under reaction conditions, the catalyst exhibits an activation which results in a lowering of the light‐off temperature by 80°C. XRD, TEM and EDX analysis carried out after the catalytic test indicate that the mixed oxide has been reduced to form a La2O3, Cu binary system. It seems that metallic copper species are the most active sites in the CO + NO reaction.
Similar content being viewed by others
References
R.J.H. Voorhoeve, in: Advanced Materials in Catalysis, eds. J.J. Burton and R.L. Garten (Academic Press, London, 1977) p. 129.
L.G. Tejuca, J.L.G. Fierro and J.M.D. Tascon, Adv. Catal. 36 (1989) 237.
K. Tabata and M. Misono, Catal. Today 8 (1990) 249.
N. Guilhaume, S.D. Peter and M. Primet, Appl. Catal. B 10 (1996) 325.
J.B. Goodenough and J.M. Longo, in: Landolt-Börnstein, Zahlenwerte und Funktionen aus Naturwissenschaft und Technik, New Series, Group III, Vol. 4 (Part a) (Springer, Berlin, 1970) p. 193.
M.P. Rosynek and D.T. Magnuson, J. Catal. 46 (1977) 402.
M.L. Rojas, J.L.G. Fierro, L.G. Tejuca and A.T. Bell, J. Catal. 124 (1990) 41.
J.A. Anderson and J.L.G. Fierro, J. Solid State Chem. 108 (1994) 305.
I. Halasz, A. Brenner, M. Shelef and K.Y.S. Ng, Catal. Lett. 11 (1991) 327.
K. Sakurai, Y. Okamoto, T. Imanaka and S. Teranishi, Bull. Chem. Soc. Jpn. 49 (1976) 1732.
N. Mizuno, M. Yamoto, M. Tanaka and M. Misono, Chem. Mater. 1 (1989) 232.
Rights and permissions
About this article
Cite this article
Peter, S., Garbowski, E., Guilhaume, N. et al. Catalytic properties of La2CuO4 in the CO + NO reaction. Catalysis Letters 54, 79–84 (1998). https://doi.org/10.1023/A:1019063502409
Issue Date:
DOI: https://doi.org/10.1023/A:1019063502409