Abstract
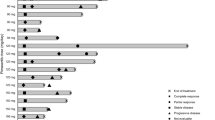
Malignant melanoma is rapidly increasing inthe United States. Metastatic diseaseresponds poorly to currently availablechemotherapy. Pyrazine diazohydroxide(PZDH) is a new agent inhibiting DNAsynthesis that is active in mouse tumormodels and human xenografts and lackscross resistance withmultiple standard agents. In this phase IItrial, patients with no prior chemotherapyor immunotherapyfor metastatic disease and performancestatus (SWOG) of 0–1, were treated withpyrazine diazohydroxide at a dose of 100 mg/m2/day by IV bolus injectionover 5–15 minutes for 5 consecutive daysevery 6 weeks. There were 23 eligiblepatients entered on this trial with 74%having PS of 0 and 91% having visceralmetastases. There were no confirmed anti-tumor responses. Theoverall response rate is 0% (95% CI 0%–15%). Median overall survival is sixmonths (95% CI 5-8months). The most common toxicities were hematologic and consisted of lymphopenia,thrombocytopenia, anemia, and leukopenia. Fatigue, and nausea and vomiting were thenext mostcommon toxicities. Pyrazine diazohydroxideby this dose and schedule has insufficientactivity in thetreatment of disseminated malignantmelanoma to warrant further investigation.
Similar content being viewed by others
References
Lotze MT, Dallal RM, Kirkwood JM, Flickinger JC: Cutaneous Melanoma. Ch 42.2 in Cancer: Principles and Practice of Oncology, Sixth Edition, edited by VT De Vita Jr, S Hellman, Rosenberg, SA. Lippincott Williams & Wilkins Publishers, Philadelphia, 2001
Division of Cancer Treatment, NCI. Annual report to the food and drug administration, pyrazine diazohydroxide (NSC 361456) December 1993
Plowman J, Haugwitz RD, Narayanan VL, Baker DC, Hand ES, Rampal JB, Safavy A: Preclinical antitumor activity of pyrazine diazohydroxide, NSC 361456. Proceedings of AACR 27: 275, 1986 (abstract)
Harrison Jr. SD, Plowman J, Dykes DJ, Waud WR, Griswold Jr. DP: Schedule dependence, activity against natural metastases, and cross-resistance of pyrazine diazohydroxide (sodium salt, NSC 361456) in preclinical models in vivo. Cancer Chemotherapy and Pharmacology 25: 425–429, 1990
Fiebig HH, Berger DP, Winterhalter BR, Plowman J: In vitro and in vivo evaluation of US-NCI compounds in human tumor xenografts. Cancer Treatment Reviews 17: 109–117, 1990
Brodfuehrer JI, Moore DJ, Melder DC, Wilke TJ, Powis G: In vitro cytotoxicity of pyrazine-2-diazohydroxide: specificity for hypoxic cells and effects of microsomal coincubation. Invest New Drugs 6: 3–9, 1988
Moore DJ, Brodfuehrer JI, Wilke TJ, Powis G: Disposition and metabolism of the antitumor agent pyrazine-2-diazohydroxide in mouse and beagle dog. Cancer Chemother Pharmacol 21: 269–273, 1988
Vogelzang NJ, Mick R, Janish L, Berezin F, Schilsky RL, Ratain MJ: Phase I and pharmacokinetic study of a new antineoplastic agent: Pyrazine diazohydroxide (NSC 361456). Cancer Res 54: 114–119, 1994
Vogelzang NJ, Mani S, Schilsky RL, Ansari RH, Taber D, Rhinehart SN, Garcia JC, Meyer SC, Mick R, Brockstein BE, Stadler WM, Ratain MJ, Vokes EE: Phase II and pharmacodynamic studies of pyrazine diazohydroxide (NSC 361456) in patients with advanced renal and colorectal cancer. Clin Cancer Res 4: 929–934, 1998
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Whitehead, R.P., Unger, J.M., Flaherty, L.E. et al. A Phase II Trial of Pyrazine Diazohydroxide in Patients with Disseminated Malignant Melanoma and no Prior Chemotherapy – Southwest Oncology Group Study. Invest New Drugs 20, 105–111 (2002). https://doi.org/10.1023/A:1014484821460
Issue Date:
DOI: https://doi.org/10.1023/A:1014484821460