Abstract
Purpose: A large part of infertility treatment involves the use of exogenous gonadotropins. The last decade has seen a progressive switch from human menopausal gonadotropin (hMG), the original gonadotropin product, to progressively more costly products, primarily or exclusively containing follicle-stimulating hormone (FSH). Though obviously at least in part driven by marketing efforts of pharmaceutical companies, this switch has received relatively little scrutiny despite its obvious cost implications. We therefore investigated whether a switch back to a generic or less costly hMG-driven ovulation induction protocol would affect patient outcome after ovulation induction and, by implications, with other assisted reproductive technologies.
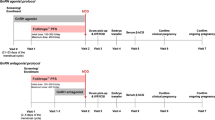
Methods: We prospectively studied clinical pregnancy rates in a large number of consecutive ovulation induction cycles in a well-defined patient population (group 1) which, after October of 1997, had been switched from a predominantly FSH to an hMG-driven protocol, based on an institutional formulary change. Until a transition period (between July and September 1997), this patient population had been on a primarily FSH-driven protocol (between July 1996 and June 1997). In parallel, we evaluated a second patient population (group 2), which was managed by the same physicians outside of formulary requirements and remained almost exclusively on principally FSH-driven ovulation induction cycles.
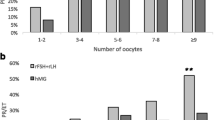
Results: FSH- and hMG-driven ovulation induction protocols did not differ in pregnancy outcome during the prospective study period. Group 1 patients, however, demonstrated a significant increase in pregnancy rates after the switch from FSH to hMG stimulation had taken place (P = 0.02), while group 2 patients demonstrated no change in pregnancy rate during the same time period.
Conclusions: Generic hMG products do not adversely affect pregnancy rates in comparison to more costly FSH products in routine ovulation induction cycles and should be considered an appropriate alternative to more expensive FSH products.
Similar content being viewed by others
REFERENCES
American Society for Reproductive Medicine Practice committee report: Induction of ovarian follicle development and ovulation with exogenous gonadotropins, 1998, pp 1-11
Jones HW, Toner JP: The infertile couple. New Engl J Med 1993;329:1710-1715
Venturoli S, Paradisi R, Fabbri R, et al.: Comparison between human urinary follicle-stimulating hormone and human menopausal gonadotropin treatment in polycystic ovary. Obstet Gynecol 1984; 63:6-11
Pratt DE, VanderLaan BF, Dudkiewicz A, et al.: A managed care provider's approach towards mandated infertility coverage: the Illinois Family Building Act. J Assist Reprod Genet 1994; 11:433-438
Karande VC, Rao R, Pratt D, et al.:A randomized prospective comparison between intrauterine insemination and fallopian sperm perfusion for the treatment of infertility. Fertil Steril 1995; 654:638-640
Pratt DE: A computer system for outcome assessment and utilization review in reproductive medicine. Infertil Reprod Med Clin North Am 1998; 9:85-96
Cohen AW: Managed healthcare's approach to infertility. Infertil Reprod Med Clinics North Am 1998;9:21-30
Armstrong DT, Coff AK, Dorrington JH: Regulation of follicular estrogen biosynthesis. In Ovarian Follicular Development and Function, AR Midgley, WA Sadler, (eds.), New York, Raven Press, 1979, pp 159-182
Ben-Chetrit A, Gotlieb L, Wong PY, et al.: Ovarian response to recombinant human follicle-stimulating hormone in luteinizing hormone-depleted women: examination of the two cell, two gonadotropin theory. Fertil Steril 1998;69:59S-65S
Society for Assisted Reproductive Technology and the American Society for Reproductive Medicine: Assisted reproductive technology in the United States and Canada: 1995 results generated from the American Society for Reproductive Medicine/Society for Assisted Reproductive Technology Registry. Fertil Steril 1998;69:389-398
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gleicher, N., Karande, V. Generic Human Menopausal Gonadotropin (hMG) in Place of More Costly Follicle-Stimulating Hormone (FSH) for Routine Ovulation Induction. J Assist Reprod Genet 17, 489–495 (2000). https://doi.org/10.1023/A:1009477523136
Issue Date:
DOI: https://doi.org/10.1023/A:1009477523136