Abstract
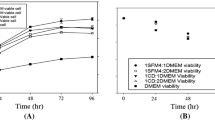
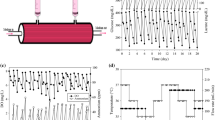
This paper analyses the performance of MAbMaxTM/TricentricTM, a new generation hollow fibre bioreactor, for hybridoma growth and antibody productivity, the down stream processing of monoclonal antibody harvests throughout the run and the further control of antibody quality consistency. Handling and process parameters were optimised using a mouse hybridoma, IgG1K secretor, and then confirmed with several other hybridomas. Cells were kept at optimal viability during an unusually long period of time and a continuously high production of antibodies was detected over several months. Foetal bovine serum concentration was reduced to 1\% and the effects of weaning of cells from serum were monitored in terms of cell metabolism and antibody productivity. Antibody harvests collected at regular intervals throughout the run (2 to 12 weeks) were purified using affinity chromatography on a recombinant protein A/G matrix and then analysed in terms of antigen binding properties, isoelectric forms and oligosaccharide structures, in order 1) to control antibody quality consistency as a function of time and serum concentration and 2) to compare antibody characteristics as a function of culture conditions, in vitro bioreactor cultivation versus in vivo mouse ascite cultivation.
Similar content being viewed by others
References
Abel CA, Spiegelberg HL and Grey HM (1968) The carbohydrate content of fragments and polypeptide chains of human gamma-myeloma proteins of different heavy chain subclasses. Biochemistry 7: 1271.
Coco-Martin JM, Brunink F, van der Velden-de Groot TAM and Beuvery EC (1992) Analysis of glycoforms present in two mouse IgG2a monoclonal antibody preparations. J Immunol Meth 155: 241–248
Davis JM, Lavender CM, Bowes KJ, Hanak JA, Combridge BS and Kingsland SL (1995) Human therapeutic monoclonal anti-D antibody produced in long-term hollow fibre culture. I — The relationship between antibody productivity and metabolic parameters. in Beuvery EC, Griffiths JB and Zeijlemaker WP (ed.) Animal Cell Technology: Developments towards the 21st Century (pp. 149–153), Kluwer Academic Publishers, Dordrecht, Boston, London.
Hamilton RG, Reimer CB and Rodkey LS (1987a) Quality control of murine monoclonal antibodies using isoelectric focusing affinity immunoblot analysis. Hybridoma 6: 205–217
Hamilton RG, Roebber M, Reimer CB and Rodkey LS (1987b) Isoelectric focusing-affinity immunoblot analysis of mouse monoclonal antibodies to the four human IgG subclasses. Electrophoresis 8: 127–134
Kessler N, Bertrand S and Aymard M (1993) Stability of a murine hybridoma is dependent on the clonal line and culture media. In Vitro Cell Dev Biol 29A: 203–207
Knazek RA, Gullino PM, Kohler PO and Dedrick RL (1972) Cell culture on artificial capillaries: an approach to tissue growth. In Vitro 178: 65
Kurkela R, Fraune E and Vihko P (1993) Pilot scale production of murine monoclonal antibodies in agitated, ceramic-matrix or hollow fibre cell culture systems. Biotechniques 15: 674–683
Leibiger H and Marx U (1995) Identification of carbohydrate structures of glycoproteins by a novel method utilizing a lectin-ELISA in Beuvery EC, Griffiths JB and Zeijlemaker WP (eds.) Animal Cell Technology: Developments towards the 21st Century (pp 409–413), Kluwer Academic Publishers, Dordrecht, Boston, London
Moellering BJ, Tedesco JL, Townsend RR, Hardy MR, Scott RW and Prior CP (1990) Electrophoretic differences in a MAb expressed in three media. BioPharm 3: 30–38
Painter RH and Freedman MH (1971) Isolation and characterization of electrophoretically homogeneous rabbit anti-hapten antibody populations. II A survey of the isoelectric properties of anti-hapten antibodies directed against charged and uncharged haptens. J Biol Chem 246: 66–92
Patel TP, Parekh RB, Moellering BJ and Prior CP (1992) Different culture methods lead to differences in glycosylation of a murine IgG monoclonal antibody. Biochem J 285: 839–845
Reisfeld RA (1967) Heterogeneity of rabbit light polypeptide chains. Cold Spring Harbor Symposium Quant Biol 32: 291–298
Tachibana H, Shirahata S and Murakami H (1992) Generation of specificity variant antibodies by alteration of carbohydrate in light chain of human monoclonal antibodies. Biochemical Biophysical Research Communication 89: 625–632
Tarakan JP and Chau PC (1986) A radial hollow fibre bioreactor for large scale culture of mammalian cells. Biotech Bioeng 28: 329–342
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kessler, N., Thomas, G., Gerentes, L. et al. Hybridoma growth in a new generation hollow fibre bioreactor: antibody productivity and consistency. Cytotechnology 24, 109–119 (1997). https://doi.org/10.1023/A:1007922004714
Issue Date:
DOI: https://doi.org/10.1023/A:1007922004714