Abstract
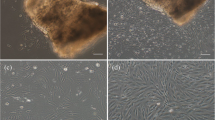
A primary cell line (designated as CCf) derived from caudal fin tissue of channel catfish, Ictalurus punctatus, was developed using explant techniques. The cell line grew fastest in media supplied with FBS and channel catfish serum. The duplication time of the cell line under optimal conditions was ∼56 h at a plating density of 1.1 × 105 cells/ml. The cell line has been propagated continuously for 25 passages (1:4 dilution per passage), cryopreserved, and recovered successfully at different passages. The cultured cells had fibroblastic morphology, and synthesized fibronectin and Type I and III collagens in the cytoplasm. The cell line maintained the normal diploid chromosome number (58) of channel catfish throughout the experiment. Nucleolus organizer regions were located on the short arms of a pair of medium-sized submetacentrics, which is typical for channel catfish. This study provides a method for acquiring a cell line from juvenile catfish without sacrifice, and is especially useful for early screening of valuable fishes.
Similar content being viewed by others
References
Amemiya CT, Bickham JW and Gold JR (1984) A cell culture technique for chromosome preparation in cyprinid fishes. Copeia 1984: 232-235.
Avella M, Berhaut J and Payan P (1994) Primary cuture of gill epithelial cells from the sea bass Dicentrarchus labrax. In VitroCell. Dev. Biol. 30A: 41-49.
Bouchard B, Fuller BB, Vijayasaradhi S and Houghton AN (1989) Induction of pigmentation in mouse fibroblasts by expression of human tyrosinase cDNA. J. Exp. Med. 169: 2029-2042.
Bowser PR and Plumb JA (1980) Fish cell lines: establishment of a line from ovaries of channel catfish. In VitroCell. Dev. Biol. 16: 363-368.
Freshney RI (1994) Culture of Animal Cells: a Manual of Basic Technique, 3rd ed. Wiley Liss, New York.
Fryer JL and Lannan CN (1994) Three decades of fish cell culture: a current listing of cell lines derived from fishes. J. Tissue Cult. Methods 16: 87-94.
Gay S, Matin GR, Muller PK, Timple R and Kuhn K (1976) Simultaneous synthesis of type I and III collagen by fibroblasts in culture. Proc. Natl. Acad. Sci. USA 73: 4037-4040.
Howel WM and Black DA (1980) Controlled silver staining of nucleolus organizer regions with a protective colloidal developer: a one step method. Experientia 36: 1014-1015.
Hynes R and Yamada KM (1982) Fibronectins: Multifuctional modular glycoproteins. J. Cell Biol. 95: 369-377.
Kang J and Caprio J (1995) In vivoresponses of single olfactory receptor neurons in the channel catfish, Ictalurus punctatus. J. Neurophysiol. 73: 172-177.
LeGrande WH, Dunham RA and Smitherman RO (1984) Karyology of three species of catfishes (Ictaluridae: Ictalurus) and four hybrid combinations. Copeia 1984: 873-878.
Miller NW and McKinney EC (1994) In vitroculture of fish leukocytes. In: Hochachka and Mommensen (eds) Biochemistry and Molecular Biology of Fishes (pp. 339-351). Elsevier Science B.V.
Miller NW, Chinchar VG and Clem LW (1994a) Development of leukocyte cell lines fromthe channel catfish (Ictalurus punctatus). J. Tissue Cult. Methods 16: 117-123.
Miller NW, Rycyzyn MA, Wilson MR, Warr GW, Naftel JP and Clem LW (1994b) Development and characterization of channel catfish long term B cell lines. J. Immunol. 152: 2180-2189.
Noga EJ (1980) Establishment of primary cell cultures using short-term trypsinization of organ fragments. J. Tissue Cult. Methods 6: 55-56.
Porter KR and Pappas GD (1959) Collagen formation by fibroblasts of the chick embryo dermis. J. Biophys. Biochem. Cytol. 5: 153-166.
Stickney RR (1993) Culture of Non-Salmonid Fishes. Boca Raton: CRC Press.
Thune RL, Stanley LA and Cooper RK(1993) Pathogenesis of warm-water bacterial fish pathogens. Ann. Rev. Fish Diseases, 37-68.
Vallejo AN, Ellsaesser CF, Miller NW and Clem LW (1991) Spontaneous development of functionally active long-term monocyte-like cell lines from channel catfish. In VitroCell. Dev. Biol. 27A: 279-286.
Wise GE, Lin F and Fan W (1992) Culture and characterization of dental follicle cells from rat molars. Cell Tissue Res. 267: 483-492.
Wohlschlag N, delos Snatos L and Dehn PF (1989) Preparation of primary cultures of adult and juvenile catfish (Ictalurus punctatus) hepatocytes. J. Tissue Cult. Methods 12: 53-55.
Wolters WR (1993) Channel catfish breeding and selection programs: Constraints and future prospects. In: Main, KL and Reynolds, B. (eds.). Selective Breeding of Fishes in Asia and the United States (pp. 82-95). The Oceanic Institute, Honolulu.
Wolters WR, Chrisman CL and Libey GS (1981). Lymphocyte culture for chromosomal analysis of channel catfish, Ictalurus puntatus. Copeia 1981: 503-504.
Yamada KM (1978) Immunological characterization of a major transformation-sensitive fibroblast cell surface glycoprotein: Localization, redistribution, and role in cell shape. J. Cell Biol. 78: 520-541.
Zhang Q (1996) Cytogenetic and molecular analysis of the channel catfish (Ictalurus punctatus) genome. Doctoral dissertation, Louisiana State University, Baton Rouge, 164 pages.
Zhang Q and Tiersch TR (1997) Chromosomal inheritance patterns of intergeneric hybrids of ictalurid catfishes: Odd diploid numbers with equal parental contributions. J. Fish Biol. In press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, Q., Cooper, R.K., Wolters, W.R. et al. Isolation, culture and characterization of a primary fibroblast cell line from channel catfish. Cytotechnology 26, 83–90 (1998). https://doi.org/10.1023/A:1007911619537
Issue Date:
DOI: https://doi.org/10.1023/A:1007911619537