Abstract
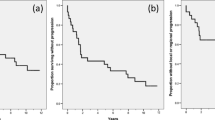
Fourteen patients with squamous cell carcinoma of the head and neck received 9-AC/DMA infusions of 850 mg/M2/day over 72 hours. Eligibility criteria included good performance status, advanced disease incurable by conventional means, no prior treatment of metastatic disease, and measurable lesions for objective response assessment. The infusions wererepeated at 21 day intervals until progression or prohibitive toxicity occurred.A median of 3 cycles (range 1–7) was given. No objective responses wereobserved. Median survival of the group was 6 months. Toxicity was hematologicwhich was modest and promptly reversible. 9-AC/DMA is inactive against this tumortype at the dose and schedule employed in this study.
Similar content being viewed by others
References
Hertzberg RP, Caranfa MJ, Hecht SM: On the mechanism of topoisomerase-I inhibition by camptothecin: evidence for binding to an enzyme-DNA complex. Biochem 28 (11): 4629–4638, 1989
Bendixen C, Thomsen B, Aisner J, Westergaard O: Camptothecin-stabilized topoisomerase I-DNA adducts cause premature termination of transcription. Biochem 29 (23): 5613–5619, 1990
Giovanella BC, Stehlin JS, Wall ME, Wani MC, Nicholas AW, Lu LF, Silber R, Potmesil M: DNA topoisomerase I-targeted chemotherapy of human colon cancer in xenografts. Science 246 (4933): 1046–1048, 1989
Rougier P, Bugat R, Douillard JY, Culine S, Sue E, Brunet P, Becouarn Y, Ychou M, Marty M, Extra JM, Bonneterre J, Adenis A, Seitz JF, Ganer G, Namer M, Conroy T, Negrier S, Merrouche Y, Burki F, Mousseau M, Herait P, Mahjoubi M: Phase II study of irinotecan in the treatment of advanced colorectal cancer in chemotherapy-naive patients and patients treated with flourouracil-based chemotherapy. J Clin Oncol 15: 251–260, 1997
Fuktioka M, Nitani H, Suzuki A, Motomiya M, Hasegawa K, Nishiwaki Y, Kuriyama T, Ariyoshi Y, Negoro S, Masuda N: A phase II study of CPT11, a new derivative of camptothecin, for previously untreated non-small cell lung cancer. J Clin Oncol 10 (1): 16–20, 1992
Masuda N, Fukuoka M, Kudoh S, Matsui K, Kusunoki Y, Takada M, Nakagawa K, Hirashima T, Tsukada H, Yana T: Phase I study of irinotecan and cisplatin with granulocyte colony-stimulating factor support for advanced non-small cell lung cancer. J Clin Oncol 12 (1): 90–96, 1994
Perez-Soler R, Fossella FV, Glisson BS, Lee JS, Murphy WK, Shin DM, Kemp BL, Lee JJ, Kane J, Robinson RA, Lippman SM, Kurie JM, Huber MH, Raber MN, Hong WK: Phase II study of topotecan in patients with advanced non-small cell lung cancer previously untreated with chemotherapy. J Clin Oncol 14 (2): 503–513, 1996
Lynch TJ, Kalish L, Strauss G, Elias A, Skarin A, Shulman LN, Posner M, Frei E III: Phase II study of topotecan in metastatic non-small cell lung cancer. J Clin Oncol 12 (2): 347–352, 1994
Robert F, Soong SJ, Wheeler RH: A phase II study of topotecan in patients with recurrent head and neck cancer. Identification of an active new agent. Am J Clin Oncol 20 (3): 298–302, 1997
Smith RE, Lew D, Rodriguez GI, Taylor SA, Schuller D, Ensley H: Evaluation of topotecan in patients with recurrent or metastatic squamous cell carcinoma of the head and neck. A phase II Southwest Oncology Group study. J Clin Oncol 14(4): 403–407, 1996
Vokes EE, Ansari RH, Masters GA, Hoffman PC, Klepsch A, Ratain MJ, Sciortino DF, Lad TE, Krauss S, Fishkin PAS, Golomb HM: A phase II study of 9-aminocamptothecin in advanced non-small cell lung cancer. Ann Oncol 9: 1085–1090, 1998
Dahut BC, Brillhart N, Takimoto C, Allegra C, Hamilton JM, Sorenson JM, Arbuck SM, Chen A, Grem J: A phase I trial of 9-aminocamptothecin (9-AC/DMA) in adult patients with solid tumors. Proc Am Soc Clin Oncol 13: 138, 1994
Gehan EA: The determination of the number of patients required in a preliminary and follow-up trial of a new chemotherapeutic agent. J Chronic Dis 13: 346–353, 1961
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lad, T., Rosen, F., Sciortino, D. et al. Phase II Trial of Aminocamptothecin (9-AC/DMA) in Patients with Advanced Squamous Cell Head and Neck Cancer. Invest New Drugs 18, 261–263 (2000). https://doi.org/10.1023/A:1006481924287
Issue Date:
DOI: https://doi.org/10.1023/A:1006481924287