Abstract
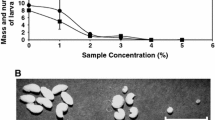
Larvae of the Mediterranean flour moth (Anagasta kuehniella Zeller) cause major losses in stored grains throughout the world. In this study pouterin, a lectin-like protein isolated from Pouteria torta (Mart.) Radlk. seeds, was tested for activity against A. kuehniella larvae. Pouterin did not produce significant effects on survival; however, at c. 1% w/w concentration in artificial diet, it produced a 71.4% reduction in average weight of the larvae. A dietary utilisation assay showed a reduction in efficiency of conversion of ingested food, efficiency of conversion of digested food and approximate digestibility as well as an increase in metabolic cost. Pouterin increased the level of trypsin in the midgut and faeces of larvae. We conclude that pouterin possesses an insecticidal effect against A. kuehniella larvae and this effect may be mediated by the binding of pouterin to chitin components of the peritrophic membrane, or glycosylated proteins in the insect midgut.
Similar content being viewed by others
References
Bate N. J. and Rothstein S. J. (1998) C6-volatiles derived from the lipoxygenase pathway induce a subset of defense-related genes. Plant Journal 16, 561–569.
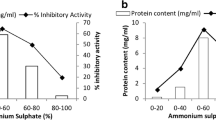
Boleti A. P. A., Freire M. G. M., Coelho M. B., Silva W., Baldasso P. A., Gomes V. M., Marangoni S., Novello J. C. and Macedo M. L. R. (2007) Insecticidal and antifungal activity of a protein from Pouteria torta seeds with lectin-like properties. Journal of Agricultural and Food Chemistry 55, 2653–2658.
Boleti A. P. A., Ventura C. A., Justo G. Z., Silva R. A., Sousa A. C. T., Ferreira C. V., Yano T. and Macedo M. L. R. (2008) Pouterin, a novel potential cytotoxic lectin-like protein with apoptosis-inducing activity in tumorigenic mammalian cells. Toxicon 51, 1321–1330.
Bradford M. M. (1976) A rapid and sensitive method for the quantification of microgram quantities for proteins utilizing the principie of protein dye binding. Anal. Biochem. 72, 248–254.
Carlini C. R. and Grossi-de-Sá M. F. (2002) Plant toxic proteins with insecticidal properties. A review on their potentialities as bioinsecticides. Toxicon 40, 1515–1539.
Coelho M. B., Marangoni S. and Macedo M. L. R. (2007) Insecticidal action of Annona coriacea lectin against the flour moth Anagasta kuehniella and the rice moth Corcyra cephalonica (Lepidoptera: Pyralidae). Comparative Biochemistry and Physiology, Part C 146, 406–414.
Erlanger F., Kokowsky N. and Cohen W. (1961) The preparation and properties of two chromogenic substrates of trypsin. Archives of Biochemistry and Biophysics 95, 271–278.
Fabre C., Causse H., Mourey L., Koninkx J., Riviere M., Puzo G., Samama J. P. and Rougé P. (1998) Characterization and sugar-binding properties of arcelin-1, an insecticidal lectin-like protein isolated from kidney bean (Phaseolus vulgaris L. cvRAZ-1) seeds. Biochemical Journal 329, 551–560.
Ferry N., Edwards M. G., Gatehouse J. A. and Gatehouse A. M. R. (2004) Plant-insect interactions: molecular approaches to insect resistance. Current Opinion in Biotechnology 15, 155–161.
Fitches E. and Gatehouse J. A. (1998) A comparison of the short and long term effects of insecticidal lectins on the activities of soluble and brush border enzymes of tomato moth larvae (Lacanobia oleracea). Journal of Insect Physiology 44, 1213–1224.
Hackman R. H. and Goldberg M. (1964) New substrate for use with chitinases. Anal. Biochem. 8, 397–401.
Koul O., Daniewski W. M., Multani J. S., Gumulka M. and Singh G. (2003) Antifeedant effects of the limonoids from Entandrophragma candolei (Meliaceae) on the gram pod borer Helicoverpa armigera (Lepidop-tera: Noctuidae). Journal of Agricultural and Food Chemistry 51, 7271–7275.
Laemmli V. K. (1970) Cleavage of structural proteins during the assembly of the bacteriophage T4. Nature 227, 680–685.
Macedo M. L. R., Fernandes K. V. S., Sales M. P. and Xavier-Filho J. (1993) Vicilins variants and the resistance of cowpea (Vigna unguiculata) seeds to the cowpea weevil (Callosobruchus maculatus). Comparative Biochemistry and Physiology C 105, 89–94.
Macedo M. L. R., Freire M. G. M., Novello J. C. and Marangoni S. (2002) Talisia esculenta lectin and larval development of Callosobruchus maculatus and Zabrotes subfasciatus (Coleoptera: Bruchidae). Biochimica et Biophysica Acta 1571, 83–88.
Macedo M. L. R., Damico D. C. S., Freire M. G. M., Toyama M. H., Marangoni S. and Novello J. C. (2003) Purification and characterization of an N-acetylglucosamine-binding lectin from Koelreuteria paniculata seeds and its effect on the larval development of Callosobruchus maculatus (Coleoptera: Bruchidae) and Anagasta kuehniella (Lepidoptera: Pyralidae). Journal of Agricultural and Food Chemistry 51, 2980–2986.
Macedo M. L. R., Freire M. G. M., Cabrini E. C., Toyama M. H., Novello J. C. and Marangoni S. (2003) A trypsin inhibitor from Peltophorum dubium seeds active against pest proteases and its effect on the survival of Anagasta kuehniella (Lepidoptera: Pyralidae). Biochimica et Biophysica Acta 1621, 170–181.
Macedo M. L. R., Freire M. G. M., Martins L. T. D., Martinez D. S., Gomes V. M., Smolka M. B., Toyama M. H., Marangoni S. and Coelho L. C. B. B. (2004) Novel protein from Labramia bojeri A. DC. seeds homologue to Kunitz-type trypsin inhibitor with lectin-like properties. Journal of Agricultural and Food Chemistry 52, 7548–7554.
Macedo M. L. R., Freire M. G. M., Silva M. B. R. and Coelho L. C. B. B. (2007) Insecticidal action of Bauhinia monandra leaf lectin (BmoLL) against Anagasta kuehniella (Lepidoptera: Pyralidae), Zabrotes subfasciatus and Callosobruchus maculatus (Coleoptera: Bruchidae). Comparative Biochemistry and Physiology 146, 486–498.
Michaud D., Faye L. and Yalle S. (1993) Electrophoretic analysis of plant cysteine and serine proteinases using gelatin-containing polyacrilamide gels and class-specific proteinase inhibitors. Electrophoresis 14, 94–99.
Mordue L. and Blackwell A. J. (1993) Azadirachtin: an update. Journal of Chemical Ecology 15, 121–128.
Powel K. S., Gatehouse A. M. R., Hilder V. A., Van Damme E. J. M., Peumans W. J., Boonjawat J., Horsham K. and Gatehouse J. A. (1995) Different antimetabolic effects of related lectins towards nymphal stages of Nilaparvata lugens. Entomologia Experimentalis et Applicata 75, 61–65.
Scriber J. M. and Slansky F. Jr (1981) The nutritional ecology of immature insects. Annual Review of Entomology 26, 183–211.
Tellan R. L., Wijffels G. and Willadsen P. (1999) Peritrophic matrix proteins. Insect Biochemistry and Molecular Biology 29, 87–101.
Uchoa A. F., DaMatta R. A., Retamal C. A., Albuquerque-Cunha J. M., Souza S. M., Samuels R. I., Silva C. P. and Xavier-Filho J. (2006) Presence of the storage seed protein vicilin in internal organs of larval Callosobruchus maculatus (Coleoptera: Bruchidae). Journal of Insect Physiology 52, 169–178.
Van Damme E. J. M., Peumans W. J., Barre A. and Rougé P. (1998) Plant lectins: a composite of several distinct families of structurally and evolutionary related proteins with diverse biological roles. Critical Reviews in Plant Sciences 17, 575–692.
Vasconcelos I. M. and Oliveira J. T. A. (2004) Antinutri-tional properties of plant lectins. Toxicon 44, 1737–1747.
Wearing C. H. (1998) Cross-resistance between azinphos-methyl and tebufenozide in the green headed leafroller, Planotortix octo. Pesticide Science 54, 203–211.
Wheeler D. A. and Isman M. B. (2001) Antifeedant and toxic activity of Trichilia americana extract against the larvae of Spodoptera litura. Entomología Experimentalis et Applicata 98, 9–16.
Zhu-Salzman K., Shade R. E., Koiwa H., Salzman R. A., Narasimhan M., Bressan R. A., Hasegawa P. M. and Murdock L. L. (1998) Carbohydrate binding and resistance to proteolysis control insecticidal activity of Griffonia simplicifolia lectin II. Proceedings of the National Academy of Sciences 95, 15123–15128.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Boleti, A.P.A., Kubo, C.E.G. & Macedo, M.L.R. Effect of pouterin, a protein from Pouteria torta (Sapotaceae) seeds, on the development of Anagasta kuehniella (Lepidoptera: Pyralidae). Int J Trop Insect Sci 29, 24–30 (2009). https://doi.org/10.1017/S1742758409299590
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1017/S1742758409299590