Abstract
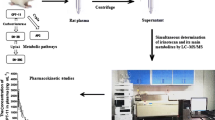
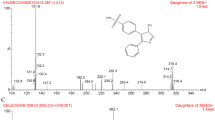
A specific, sensitive and robust LC-MS/MS method was developed and validated for the quantification of deoxyelephantopin in rat plasma using simvastatin as an internal standard as per regulatory guidelines of Bioanalytical Method Validation. Plasma sample was prepared through liquid-liquid extraction. Chromatographic separation was performed on an Agela-C18 analytical column (1.8 μm, 2.1 mm × 50 mm) with an isocratic mobile phase consisting of 0.05% formic acid (dissolved in acetonitrile) and water (55:45, v/v) at a flow rate of 0.5 ml/min. The column oven was maintained at 40 °C and the injection volume was 4 μl. Elution of deoxyelephantopin and the internal standard occurred at 5.1 and 6.3 min, respectively. The total chromatographic run time was 7.5 min. A linear response function was constructed in the concentration range of 13.2–2640 ng/ml. The intra- and inter-day precision and accuracy were in the range of 1.4–14.8% and −11.7 to 14.1%, respectively. The validated LC-MS/MS was successfully applied to the pharmacokinetic study of deoxyelephantopin after intravenous injection of 1, 2 and 4 mg/kg and oral administration of 7.5, 15 and 30 mg/kg deoxyelephantopin in rats. After oral and intravenous administration, the Cmax and AUC values of deoxyelephantopin increased dose-dependently.
Article PDF
Similar content being viewed by others
References
Anees, A., Alkarkhi, A.F.M., Hena, S., Khim, L.H., 2009. Extraction, separation and identification of chemical ingredients of Elephantopus scaber L. using factorial design of experiment. Int. J. Chem. 1, 36–49.
Andy, S.N., Chan, C.K., Kadir, H.A., 2018. Deoxyelephantopin from Elephantopus scaber modulates neuroinflammatory response through MAPKs and PI3K/Aktdependent NF-kB signaling pathways in LPS-stimulated BV-2 microglial cells. J. Funct. Foods 38, 221–231.
Behera, S.K., Misra, M.K., 2005. Indigenous phytotherapy for genitor-urinary diseases used by the Kandha tribe of Orissa. J. Ethnopharmacol. 102, 319–325.
Chan, C.K., Supriady, H., Goh, B.H., Kadir, H.A., 2015. Elephantopus scaber induces apoptosis through ROS-dependent mitochondrial signaling pathway in HCT116 human colorectal carcinoma cells. J. Ethnopharmacol. 168, 291–304.
Farha, A.K., Dhanya, S.R., Mangalam, S.N., Geetha, B.S., Latha, P.G., Remani, P., 2014. Deoxyelephantopin impairs growth of cervical carcinoma SiHa cells and induces apoptosis by targeting multiple molecular signaling pathways. Cell Biol. Toxicol. 30, 331–343.
Farha, A.K., Dhanya, S.R., Mangalam, S.N., Remani, P., 2015. Anti-metastatic effect of deoxyelephantopin from Elephantopus scaber in A549 lung cancer cells in vitro. Nat. Prod. Res. 29, 2341–2345.
Ho, W.Y., Yeap, S.K., Ho, C.L., Abdul Rahim, R., Alitheen, N.B., 2012. Hepatoprotective activity of Elephantopus scaber on alcohol-induced liver damage in mice. Evid. Based Complement. Alternat. Med., https://doi.org/10.1155/2012/417953.
Hiradeve, S.M., Rangari, V.D., 2014. Elephantopus scaber Linn.: a review on its ethnomedical, phytochemical and pharmacological profile. J. Appl. Biomed. 12, 49–61.
Hou, L., Huang, H., 2016. Immune suppressive properties of artemisinin family drugs. Pharmacol. Ther. 166, 123–127.
Huang, C.C., Lin, K.J., Cheng, Y.W., Hsu, C.A., Yang, S.S., Shyur, L.F., 2013. Hepatoprotective effect and mechanistic insights of deoxyelephantopin, a phyto-sesquiterpene lactone, against fulminant hepatitis. J. Nutr. Biochem. 24, 516–530.
Kabeer, F.A., Sreedevi, G.B., Nair, M.S., Rajalekshmi, D.S., Gopalakrishnan, L.P., Kunjuraman, S., Prathapan, R., 2013. Antineoplastic effects of deoxyelephantopin, a sesquiterpene lactone from Elephantopus scaber, on lung adenocarcinoma (A549) cells. J. Integr. Med. 11, 269–277.
Lee, W.L., Shyur, L.F., 2012. Deoxyelephantopin impedes mammary adenocarcinoma cell motility by inhibiting calpain-mediated adhesion dynamics and inducing reactive oxygen species and aggresome formation. Free Radic. Biol. Med. 52, 1423–1436.
Lachi-Silva, L., Sy, S.K.B., Voelkner, A., Sousa, J.P.B., Lopes, J.L.C., Silva, D.B., Lopes, N.P., Kimura, E., Derendorf, H., Diniz, A., 2015. Simultaneous characterization of intravenous and oral pharmacokinetics of lychnopholide in rats by transit compartment model. Planta Med. 81, 1121–1127.
Rajakumar, N., Shivanna, M.B., 2010. Traditional herbal medicinal knowledge in Sagar taluk of Shimoga district, Karnataka, India. Indian J. Nat. Prod. Resour. 1, 102–108.
Schepetkin, I.A., Kirpotin, L.N., Mitchell, P.T., Kishkentaev, S., Shaimerdenov, Z.R., Atazhanov, G.A., Adekenov, S.M., Quinn, M.T., 2018. The natural sesquiterpene lactones arglabin, grosheimin, agracin, parthenolide, and estafiatin inhibit T cell receptor (TCR) activation. Phytochemistry 146, 36–46.
Singh, S.D.J., Krishna, V., Mankani, K.L., Manjunatha, B.K., Vidya, S.M., Manohara, Y.N., 2005. Wound healing activity of the leafextracts and deoxyelephantopin isolated from Elephantopus scaber Linn. Indian J. Pharmacol. 37, 238–242.
Su, M., Chung, H.Y., Li, Y., 2011. Deoxyelephantopin from Elephantopus scaber L. induces cell-cycle arrest and apoptosis in the human nasopharyngeal cancer CNE cells. Biochem. Biophys. Res. Commun. 411, 342–347.
US Food and Drug Administration, 2001. Guidance for Industry: Bioanalytical Method Validation, http://www.fda.gov/downloads/Drugs/ Guidance/ucm070107.pdf (accessed 10.01.17).
Wu, J., Sun, L., Wang, J., 2014. Qualitative and quantitative analysis of deoxyelephantopin in Elephantopus scaber L. Anhui Med. Pharm. J. 18, 1862–1865.
Wu, C., Xiao, J., Wu, X., Wang, G., Zhou, G., Li, Y., 2013. Fingerprint study and content determination of three active components of Elephantopus scaber L. Lishizhen Med. Mater. Med. Res. 24, 1643–1645.
Zahari, Z., Jani, N.A., Amanah, A., Latif, M.N., Majid, M.I., Adenan, M.I., 2014. Bioassay-guided isolation of a sesquiterpene lactone of deoxyelephantopin from Elephantopus scaber Linn active on Trypanosome brucei rhodesience. Phytomedicine 21, 282–285.
Zou, J., Zhang, Y., Sun, J., Wang, X., Tu, H., Geng, S., Liu, R., Chen, Y., Bi, Z., 2017. Deoxyelephantopin induces reactive oxygen species-mediated apoptosis and autophagy in human osteosarcoma cells. Cell Physiol. Biochem. 42, 1812–1821.
Zhao, W., Liu, N., Lei, J., 2015. Simultaneous determination of schaftoside, isoschaftoside, deoxyelephantopin and 4, 5-dicaffeoylquinic acid in shennongcha granules by HPLC. Chin. Pharm. 18, 741–744.
Zhao, A.Q., Zhao, J.H., Zhang, S.Q., Pan, Y.Y., Huo, X.L., 2016. Determination of parthenolide in rat plasma by UPLC–MS/MS and its application to a pharmacokinetic study. J. Pharm. Biomed. Anal. 119, 99–103.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
KN, CG and HY carried out the data analyses. KN and ST designed the study and wrote the manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niu, K., Guo, C., Yan, H. et al. LC-MS/MS determination of deoxyelephantopin, a novel anti-tumor candidate in rat plasma and its application to a pharmacokinetic study in rats. Rev. Bras. Farmacogn. 28, 582–588 (2018). https://doi.org/10.1016/j.bjp.2018.06.001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.06.001