Abstract
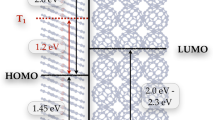
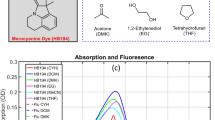
We present a study of the excited state relaxation dynamics of the photosensitizer P1 used in p-type dye-sensitized solar cells. Comparative femtosecond fluorescence upconversion measurements in solution and in films show that the dye undergoes a picosecond electronic relaxation from the bright Franck–Condon (FC) state to a low-emitting charge-transfer (CT) state in polar environment. The fluorescence is moderately quenched in solution and on the mesoporous Al2O3 isolator but dramatically more on NiO semiconductor. We assign this sub-picosecond process to the hole injection thus confirming that the electron transfer is from the FC state directly into the NiO valence band.
Graphic abstract









Similar content being viewed by others
References
Gong, J., Sumathy, K., Qiao, Q., & Zhou, Z. (2017). Renewable and Sustainable Energy Reviews, 68, 234–246. https://doi.org/10.1016/j.rser.2016.09.097
Tian, L., Tyburski, R., Wen, C., Sun, R., Abdellah, M., Huang, J., D’Amario, L., Boschloo, G., Hammarström, L., & Tian, H. (2020). Journal of the American Chemical Society, 142(43), 18668–18678. https://doi.org/10.1021/jacs.0c08886
Martín, C., Ziółek, M., & Douhal, A. (2016). Journal of Photochemistry and Photobiology C, 26, 1–30. https://doi.org/10.1016/j.jphotochemrev.2015.12.001
Ponseca, C. S., Chábera, P., Uhlig, J., Persson, P., & Sundström, V. (2017). Chemical Reviews, 117(16), 10940–11024. https://doi.org/10.1021/acs.chemrev.6b00807
Maffeis, V., Dogan, H., Cassette, E., Jousselme, B., & Gustavsson, T. (2019). The Journal of Physical Chemistry Letters, 10(17), 5076–5081. https://doi.org/10.1021/acs.jpclett.9b01947
Morandeira, A., Boschloo, G., Hagfeldt, A., & Hammarström, L. (2005). The Journal of Physical Chemistry B, 109(41), 19403–19410. https://doi.org/10.1021/jp053230e
Gibson, E. A., Smeigh, A. L., Le Pieux, L., Fortage, J., Boschloo, G., Blart, E., Pellegrin, Y., Odobel, F., Hagfeldt, A., & Hammarström, L. (2009). Angewandte Chemie, 48(24), 4402–4405. https://doi.org/10.1002/anie.200900423
Nikolaou, V., Charisiadis, A., Charalambidis, G., Coutsolelos, A. G., & Odobel, F. (2017). Journal of Materials Chemistry A, 5, 21077–21113. https://doi.org/10.1039/C7TA06500K
Wood, C. J., Summers, G. H., Clark, C. A., Kaeffer, N., Braeutigam, M., Carbone, L. R., D’Amario, L., Fan, K., Farré, Y., Narbey, S., Oswald, F., Stevens, L. A., Parmenter, C. D. J., Fay, M. W., La Torre, A., Snape, C. E., Dietzek, B., Dini, D., Hammarström, L., … Gibson, E. A. (2016). Physical Chemistry Chemical Physics, 18(16), 10727–10738. https://doi.org/10.1039/c5cp05326a
Qin, P., Wiberg, J., Gibson, E. A., Linder, M., Li, L., Brinck, T., Hagfeldt, A., Albinsson, B., & Sun, L. (2010). The Journal of Physical Chemistry C, 114(10), 4738–4748. https://doi.org/10.1021/jp911091n
Zhang, L., Boschloo, G., Hammarström, L., & Tian, H. (2016). Physical Chemistry Chemical Physics, 18(7), 5080–5085. https://doi.org/10.1039/c5cp05247e
Schmidt, B., Sobotta, C., Malkmus, S., Laimgruber, S., Braun, M., Zinth, W., & Gilch, P. (2004). The Journal of Physical Chemistry A, 108(20), 4399–4404. https://doi.org/10.1021/jp0495747
Fakis, M., Stathatos, E., Tsigaridas, G., Giannetas, V., & Persephonis, P. (2011). The Journal of Physical Chemistry C, 115(27), 13429–13437. https://doi.org/10.1021/jp201143n
Maffeis, V., Brisse, R., Labet, V., Jousselme, B., & Gustavsson, T. (2018). The Journal of Physical Chemistry A, 122(25), 5533–5544. https://doi.org/10.1021/acs.jpca.8b05177
Brisse, R., Praveen, C., Maffeis, V., Bourgeteau, T., Tondelier, D., Berthelot, T., Geffroy, B., Gustavsson, T., Raimundo, J. M., & Jousselme, B. (2018). Sustainable Energy & Fuels, 2, 648–654. https://doi.org/10.1039/C7SE00474E
Angulo, G., Grampp, G., & Rosspeintner, A. (2006). Spectrochimica Acta Part A, 65(3–4), 727–731. https://doi.org/10.1016/j.saa.2006.01.007
Brouwer, A. M. (2011). Pure and Applied Chemistry, 83(12), 2213–2228. https://doi.org/10.1351/PAC-REP-10-09-31
Gustavsson, T., Cassara, L., Gulbinas, V., Gurzadyan, G., Mialocq, J. C., Pommeret, S., Sorgius, M., & van der Meulen, P. (1998). The Journal of Physical Chemistry A, 102(23), 4229–4245. https://doi.org/10.1021/jp980282d
Lemmetyinen, H., Tkachenko, N. V., Valeur, B., Hotta, J., Ameloot, M., Ernsting, N. P., Gustavsson, T., & Boens, N. (2014). Pure and Applied Chemistry, 86(12), 1969–1998. https://doi.org/10.1515/pac-2013-0912
Changenet-Barret, P., Emanuele, E., Gustavsson, T., Improta, R., Kotlyar, A. B., Markovitsi, D., Vayá, I., Zakrzewska, K., & Zikich, D. (2010). The Journal of Physical Chemistry C, 114(34), 14339–14346. https://doi.org/10.1021/jp102106d
Changenet-Barret, P., Gustavsson, T., Markovitsi, D., Manet, I., & Monti, S. (2013). Physical Chemistry Chemical Physics, 15(8), 2937–2944. https://doi.org/10.1039/c2cp44056c
Joly, D., Pellejà, L., Narbey, S., Oswald, F., Chiron, J., Clifford, J. N., Palomares, E., & Demadrille, R. (2014). Scientific Reports, 4, 4033–4039. https://doi.org/10.1038/srep04033
Horng, M. L., Gardecki, J. A., Papazyan, A., & Maroncelli, M. (1995). The Journal of Physical Chemistry, 99(48), 17311–17337. https://doi.org/10.1021/j100048a004
Reynolds, L., Gardecki, J. A., Frankland, S. J. V., Horng, M. L., & Maroncelli, M. (1996). The Journal of Physical Chemistry, 100(95), 10337–10354. https://doi.org/10.1021/Jp953110e
Oum, K., Lohse, P. W., Klein, J. R., Flender, O., Scholz, M., Hagfeldt, A., Boschloo, G., & Lenzer, T. (2013). Physical Chemistry Chemical Physics, 15(15), 3906–3916. https://doi.org/10.1039/c3cp44095h
Ishow, E., Clavier, G., Miomandre, F., Rebarz, M., Buntinx, G., & Poizat, O. (2013). Physical Chemistry Chemical Physics, 15(33), 13922–13939. https://doi.org/10.1039/c3cp51480c
Yao, Z., Yang, L., Cai, Y., Yan, C., Zhang, M., Cai, N., Dong, X., & Wang, P. (2014). The Journal of Physical Chemistry C, 118(6), 2977–2986. https://doi.org/10.1021/jp412070p
Lefebvre, J.-F., Sun, X.-Z., Calladine, J. A., George, M. W., & Gibson, E. A. (2014). ChemComm, 50, 5258–2560. https://doi.org/10.1039/c3cc46133e
Black, F. A., Clark, C. A., Summers, G. H., Clark, I. P., Towrie, M., Penfold, T., George, M. W., & Gibson, E. A. (2017). Physical Chemistry Chemical Physics, 19(11), 7877–7885. https://doi.org/10.1039/c6cp05712h
Morandeira, A., Fortage, J., Edvinsson, T., Le, L., Pleux, E., Blart, E., Boschloo, G., Hagfeldt, A., Hammarström, L., & Odobel, F. (2008). The Journal of Physical Chemistry C, 112(5), 1721–1728. https://doi.org/10.1021/jp077446n
Le Pleux, L., Smeigh, A. L., Gibson, E., Pellegrin, Y., Blart, E., Boschloo, G., Hagfeldt, A., Hammarström, L., & Odobel, F. (2011). Energy and Environmental Science, 4(6), 2075–2084. https://doi.org/10.1039/c1ee01148k
Ziółek, M., Martín, C., Sun, L., & Douhal, A. (2012). The Journal of Physical Chemistry C, 116(50), 26227–26238. https://doi.org/10.1021/jp3097988
Ziółek, M., Martín, C., Cohen, B., Garcia, H., & Douhal, A. (2011). The Journal of Physical Chemistry C, 115(47), 23642–23650. https://doi.org/10.1021/jp208274x
Giannouli, M., & Fakis, M. (2011). Journal of Photochemistry and Photobiology, 226(1), 42–50. https://doi.org/10.1016/j.jphotochem.2011.10.013
Wiberg, J., Marinado, T., Hagberg, D. P., Sun, L., Hagfeldt, A., & Albinsson, B. (2010). The Journal of Physical Chemistry B, 114(45), 14358–14363. https://doi.org/10.1021/jp1002963
Oum, K., Flender, O., Lohse, P. W., Scholz, M., Hagfeldt, A., Boschloo, G., & Lenzer, T. (2014). Physical Chemistry Chemical Physics, 16(16), 8019–8029. https://doi.org/10.1039/c3cp55298e
Funding
This work was supported by the CEA program DSM Energie, contract PHLUMVIR—E113-9.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
Not applicable.
Data availability
All data reported in the Figures and the Tables can be obtained from the authors upon request.
Code availability
An executable version of the home-made fitting program used can be obtained from the authors upon request. All the Origin 8.1SR2 scripts used in data treatment (log-normal fitting of the TRFS) are available upon request.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Pushing the limits of flash photolysis to unravel the secrets of biological electron and proton transfer—a topical issue in honour of Klaus Brettel.
Supplementary Information
Below is the link to the electronic supplementary material.
43630_2021_98_MOESM1_ESM.docx
Supplementary file1 The Supporting Information contains the following figures; fluorescence decays recorded by TCSPC for P1 in toluene, THF, EtOH and ACN, total fluorescence intensities and mean positions of the fluorescence band as obtained from lognorm fitting for P1 in toluene, THF, EtOH and ACN. Depth profiles of NiO and Al2O3 films, photographs of films, absorption and fluorescence spectra of P1 dye-sensitized NiO and Al2O3 films, fluorescence decays of P1 dye-sensitized NiO and Al2O3 films recorded by TCSPC, lognorm-fitted parameters of total fluorescence intensities and mean positions of the fluorescence band, Radiative and non-radiative rate constants as well as the radiative lifetimes for P1 in toluene, THF, EtOH and ACN, the radiative rate constant and radiative lifetime of P1 in THF (DOCX 668 kb)
Rights and permissions
About this article
Cite this article
Maffeis, V., Jousselme, B. & Gustavsson, T. Hole injection from P1 dye hot-excited states in p-type dye-sensitized films: a fluorescence study. Photochem Photobiol Sci 20, 1257–1271 (2021). https://doi.org/10.1007/s43630-021-00098-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-021-00098-8