Abstract
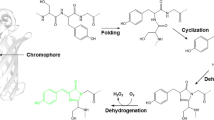
mKate red-to-green photoconversion is a non-canonical type of phototransformation in fluorescent proteins, with a poorly understood mechanism. We have hypothesized that the daughter mKate2 protein may also be photoconvertible, and that this phenomenon would be connected with mKate(2) chromophore photoreduction. Indeed, upon the intense irradiation of the protein sample supplemented by sodium dithionite, the accumulation of green as well as blue spectral forms is enhanced. The reaction was shown to be reversible upon the reductant’s removal. However, an analysis of the fluorescence microscopy data, absorption spectra, kinetics and time-resolved fluorescence spectroscopy revealed that the short-wavelength spectral forms of mKate(2) exist before photoactivation, that their fractions increase light-independently after dithionite addition, and that the conversion is facilitated by the photobleaching of the red chromophore form.








Similar content being viewed by others
Availability of data and material
Supplementary information is available along with an online-version of the manuscript at https://doi.org/10.1007/s43630-021-00060-8. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Chudakov, D. M., Matz, M. V., Lukyanov, S., & Lukyanov, K. A. (2010). Fluorescent proteins and their applications in imaging living cells and tissues. Physiological Reviews, 90(3), 1103–1163. https://doi.org/10.1152/physrev.00038.2009
Mishin, A. S., Belousov, V. V., Solntsev, K. M., & Lukyanov, K. A. (2015). Novel uses of fluorescent proteins. Current Opinion in Chemical Biology, 27, 1–9. https://doi.org/10.1016/j.cbpa.2015.05.002
Cardarelli, F. (2020). Back to the future: Genetically encoded fluorescent proteins as inert tracers of the intracellular environment. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms21114164
Aglyamova, G. V., Hunt, M. E., Modi, C. K., & Matz, M. V. (2011). Multi-colored homologs of the green fluorescent protein from hydromedusa Obelia sp. Photochemical & Photobiological Sciences: Official journal of the European Photochemistry Association and the European Society for Photobiology, 10(8), 1303–1309. https://doi.org/10.1039/c1pp05068k
Lambert, G. G., Depernet, H., Gotthard, G., Schultz, D. T., Navizet, I., Lambert, T., & Shaner, N. C. (2020). Aequorea’s secrets revealed: New fluorescent proteins with unique properties for bioimaging and biosensing. PLoS Biology, 18(11), e3000936. https://doi.org/10.1371/journal.pbio.3000936
Matz, M. V., Fradkov, A. F., Labas, Y. A., Savitsky, A. P., Zaraisky, A. G., Markelov, M. L., & Lukyanov, S. A. (1999). Fluorescent proteins from nonbioluminescent Anthozoa species. Nature biotechnology, 17(10), 969–973. https://doi.org/10.1038/13657
Salih, A., Larkum, A., Cox, G., Kühl, M., & Hoegh-Guldberg, O. (2000). Fluorescent pigments in corals are photoprotective. Nature, 408(6814), 850–853. https://doi.org/10.1038/35048564
Wiedenmann, J., Schenk, A., Röcker, C., Girod, A., Spindler, K.-D., & Nienhaus, G. U. (2002). A far-red fluorescent protein with fast maturation and reduced oligomerization tendency from Entacmaea quadricolor (Anthozoa, Actinaria). Proceedings of the National Academy of Sciences of the United States of America, 99(18), 11646–11651. https://doi.org/10.1073/pnas.182157199
Karasawa, S., Araki, T., Nagai, T., Mizuno, H., & Miyawaki, A. (2004). Cyan-emitting and orange-emitting fluorescent proteins as a donor/acceptor pair for fluorescence resonance energy transfer. The Biochemical journal, 381(Pt 1), 307–312. https://doi.org/10.1042/BJ20040321
Shagin, D. A., Barsova, E. V., Yanushevich, Y. G., Fradkov, A. F., Lukyanov, K. A., Labas, Y. A., & Matz, M. V. (2004). GFP-like proteins as ubiquitous metazoan superfamily: Evolution of functional features and structural complexity. Molecular Biology and Evolution, 21(5), 841–850. https://doi.org/10.1093/molbev/msh079
Deheyn, D. D., Kubokawa, K., McCarthy, J. K., Murakami, A., Porrachia, M., Rouse, G. W., & Holland, N. D. (2007). Endogenous green fluorescent protein (GFP) in amphioxus. The Biological Bulletin, 213(2), 95–100. https://doi.org/10.2307/25066625
Lambert, T. J. (2019). FPbase: A community-editable fluorescent protein database. Nature Methods, 16(4), 277–278. https://doi.org/10.1038/s41592-019-0352-8
Lukyanov, K. A., Chudakov, D. M., Lukyanov, S., & Verkhusha, V. V. (2005). Innovation: Photoactivatable fluorescent proteins. Nature Reviews. Molecular Cell Biology, 6(11), 885–891. https://doi.org/10.1038/nrm1741
Adam, V., Berardozzi, R., Byrdin, M., & Bourgeois, D. (2014). Phototransformable fluorescent proteins: Future challenges. Current Opinion in Chemical Biology, 20, 92–102. https://doi.org/10.1016/j.cbpa.2014.05.016
Chang, H., Zhang, M., Ji, W., Chen, J., Zhang, Y., Liu, B., & Xu, T. (2012). A unique series of reversibly switchable fluorescent proteins with beneficial properties for various applications. Proceedings of the National Academy of Sciences of the United States of America, 109(12), 4455–4460. https://doi.org/10.1073/pnas.1113770109
Jablonski, A. E., Vegh, R. B., Hsiang, J.-C., Bommarius, B., Chen, Y.-C., Solntsev, K. M., & Dickson, R. M. (2013). Optically modulatable blue fluorescent proteins. Journal of the American Chemical Society, 135(44), 16410–16417. https://doi.org/10.1021/ja405459b
Pennacchietti, F., Serebrovskaya, E. O., Faro, A. R., Shemyakina, I. I., Bozhanova, N. G., Kotlobay, A. A., & Testa, I. (2018). Fast reversibly photoswitching red fluorescent proteins for live-cell RESOLFT nanoscopy. Nature Methods, 15(8), 601–604. https://doi.org/10.1038/s41592-018-0052-9
Chudakov, D. M., Verkhusha, V. V., Staroverov, D. B., Souslova, E. A., Lukyanov, S., & Lukyanov, K. A. (2004). Photoswitchable cyan fluorescent protein for protein tracking. Nature Biotechnology, 22(11), 1435–1439. https://doi.org/10.1038/nbt1025
Zhang, M., Chang, H., Zhang, Y., Yu, J., Wu, L., Ji, W., & Xu, T. (2012). Rational design of true monomeric and bright photoactivatable fluorescent proteins. Nature Methods, 9(7), 727–729. https://doi.org/10.1038/nmeth.2021
Patterson, G. H., & Lippincott-Schwartz, J. (2002). A photoactivatable GFP for selective photolabeling of proteins and cells. Science, (New York, N.Y.), 297(5588), 1873–1877. https://doi.org/10.1126/science.1074952
Chudakov, D. M., Belousov, V. V., Zaraisky, A. G., Novoselov, V. V., Staroverov, D. B., Zorov, D. B., & Lukyanov, K. A. (2003). Kindling fluorescent proteins for precise in vivo photolabeling. (pp. 191–194). United States. https://doi.org/10.1038/nbt778
Adam, V., Moeyaert, B., David, C. C., Mizuno, H., Lelimousin, M., Dedecker, P., & Hofkens, J. (2011). Rational design of photoconvertible and biphotochromic fluorescent proteins for advanced microscopy applications. Chemistry & Biology, 18(10), 1241–1251. https://doi.org/10.1016/j.chembiol.2011.08.007
Solovyev, I. D., Gavshina, A. V., & Savitsky, A. P. (2019). Novel phototransformable fluorescent protein SAASoti with unique photochemical properties. International Journal of Molecular Sciences, 20(14), 3399. https://doi.org/10.3390/ijms20143399
Betzig, E., Patterson, G. H., Sougrat, R., Lindwasser, O. W., Olenych, S., Bonifacino, J. S., & Hess, H. F. (2006). Imaging intracellular fluorescent proteins at nanometer resolution. Science (New York, N.Y.), 313(5793), 1642–1645. https://doi.org/10.1126/science.1127344
Rust, M. J., Bates, M., & Zhuang, X. (2006). Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nature Methods, 3(10), 793–795. https://doi.org/10.1038/nmeth929
Nienhaus, K., & Nienhaus, G. U. (2014). Fluorescent proteins for live-cell imaging with super-resolution. Chemical Society Reviews, 43(4), 1088–1106. https://doi.org/10.1039/c3cs60171d
Li, H., & Vaughan, J. C. (2018). Switchable fluorophores for single-molecule localization microscopy. Chemical Reviews, 118(18), 9412–9454. https://doi.org/10.1021/acs.chemrev.7b00767
Acharya, A., Bogdanov, A. M., Grigorenko, B. L., Bravaya, K. B., Nemukhin, A. V., Lukyanov, K. A., & Krylov, A. I. (2017). Photoinduced chemistry in fluorescent proteins: Curse or blessing? Chemical Reviews, 117(2), 758–795. https://doi.org/10.1021/acs.chemrev.6b00238
Bogdanov, A. M., Mishin, A. S., Yampolsky, I. V., Belousov, V. V., Chudakov, D. M., Subach, F. V., & Lukyanov, K. A. (2009). Green fluorescent proteins are light-induced electron donors. Nature Chemical Biology, 5(7), 459–461. https://doi.org/10.1038/nchembio.174
Subach, O. M., Patterson, G. H., Ting, L.-M., Wang, Y., Condeelis, J. S., & Verkhusha, V. V. (2011). A photoswitchable orange-to-far-red fluorescent protein, PSmOrange. Nature Methods, 8(9), 771–777. https://doi.org/10.1038/nmeth.1664
Gorbachev, D. A., Petrusevich, E. F., Kabylda, A. M., Maksimov, E. G., Lukyanov, K. A., Bogdanov, A. M., & Mishin, A. S. (2020). A general mechanism of green-to-red photoconversions of GFP. Frontiers in Molecular Biosciences, 7, 176. https://doi.org/10.3389/fmolb.2020.00176
Herman, P., Holoubek, A., & Brodska, B. (2019). Lifetime-based photoconversion of EGFP as a tool for FLIM. Biochimica et Biophysica Acta. General Subjects, 1863(1), 266–277. https://doi.org/10.1016/j.bbagen.2018.10.016
Kremers, G.-J., Hazelwood, K. L., Murphy, C. S., Davidson, M. W., & Piston, D. W. (2009). Photoconversion in orange and red fluorescent proteins. Nature Methods, 6(5), 355–358. https://doi.org/10.1038/nmeth.1319
Verkhusha, V. V., Chudakov, D. M., Gurskaya, N. G., Lukyanov, S., & Lukyanov, K. A. (2004). Common pathway for the red chromophore formation in fluorescent proteins and chromoproteins. Chemistry & Biology, 11(6), 845–854. https://doi.org/10.1016/j.chembiol.2004.04.007
Shaner, N. C., Campbell, R. E., Steinbach, P. A., Giepmans, B. N. G., Palmer, A. E., & Tsien, R. Y. (2004). Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nature Biotechnology, 22(12), 1567–1572. https://doi.org/10.1038/nbt1037
Pletneva, N., Pletnev, S., Tikhonova, T., Popov, V., Martynov, V., & Pletnev, V. (2006). Structure of a red fluorescent protein from Zoanthus, zRFP574, reveals a novel chromophore. Acta Crystallographica Section D, Biological Crystallography, 62(Pt 5), 527–532. https://doi.org/10.1107/S0907444906007852
Pakhomov, A. A., & Martynov, V. I. (2007). Chromophore aspartate oxidation-decarboxylation in the green-to-red conversion of a fluorescent protein from Zoanthus sp. 2. Biochemistry, 46(41), 11528–11535. https://doi.org/10.1021/bi700721x
Pletneva, N., Pletnev, V., Tikhonova, T., Pakhomov, A. A., Popov, V., Martynov, V. I., & Pletnev, S. (2007). Refined crystal structures of red and green fluorescent proteins from the button polyp Zoanthus. Acta Crystallographica. Section D, Biological Crystallography, 63(Pt 10), 1082–1093. https://doi.org/10.1107/S0907444907042461
Kim, S. E., Hwang, K. Y., & Nam, K. H. (2019). Spectral and structural analysis of a red fluorescent protein from Acropora digitifera. Protein Science: A Publication of the Protein Society, 28(2), 375–381. https://doi.org/10.1002/pro.3540
Marchant, J. S., Stutzmann, G. E., Leissring, M. A., LaFerla, F. M., & Parker, I. (2001). Multiphoton-evoked color change of DsRed as an optical highlighter for cellular and subcellular labeling. Nature Biotechnology, 19(7), 645–649. https://doi.org/10.1038/90249
Snellenburg, J. J., Laptenok, S., Seger, R., Mullen, K. M., & van Stokkum, I. H. M. (2012). Glotaran: A java-based graphical user interface for the R package TIMP. Journal of Statistical Software, Articles, 49(3), 1–22. https://doi.org/10.18637/jss.v049.i03
Subach, F. V., & Verkhusha, V. V. (2012). Chromophore transformations in red fluorescent proteins. Chemical Reviews, 112(7), 4308–4327. https://doi.org/10.1021/cr2001965
Subach, O. M., Gundorov, I. S., Yoshimura, M., Subach, F. V., Zhang, J., Grüenwald, D., & Verkhusha, V. V. (2008). Conversion of red fluorescent protein into a bright blue probe. Chemistry & Biology, 15(10), 1116–1124. https://doi.org/10.1016/j.chembiol.2008.08.006
Reid, B. G., & Flynn, G. C. (1997). Chromophore formation in green fluorescent protein. Biochemistry, 36(22), 6786–6791. https://doi.org/10.1021/bi970281w
Cloin, B. M. C., De Zitter, E., Salas, D., Gielen, V., Folkers, G. E., Mikhaylova, M., & Kapitein, L. C. (2017). Efficient switching of mCherry fluorescence using chemical caging. Proceedings of the National Academy of Sciences of the United States of America, 114(27), 7013–7018. https://doi.org/10.1073/pnas.1617280114
Heppert, J. K., Dickinson, D. J., Pani, A. M., Higgins, C. D., Steward, A., Ahringer, J., & Goldstein, B. (2016). Comparative assessment of fluorescent proteins for in vivo imaging in an animal model system. Molecular Biology of the Cell, 27(22), 3385–3394. https://doi.org/10.1091/mbc.E16-01-0063
Chattoraj, M., King, B. A., Bublitz, G. U., & Boxer, S. G. (1996). Ultra-fast excited state dynamics in green fluorescent protein: Multiple states and proton transfer. Proceedings of the National Academy of Sciences of the United States of America, 93(16), 8362–8367. https://doi.org/10.1073/pnas.93.16.8362
Shcherbo, D., Merzlyak, E. M., Chepurnykh, T. V., Fradkov, A. F., Ermakova, G. V., Solovieva, E. A., & Chudakov, D. M. (2007). Bright far-red fluorescent protein for whole-body imaging. Nature Methods, 4(9), 741–746. https://doi.org/10.1038/nmeth1083
Shcherbo, D., Murphy, C. S., Ermakova, G. V., Solovieva, E. A., Chepurnykh, T. V., Shcheglov, A. S., & Chudakov, D. M. (2009). Far-red fluorescent tags for protein imaging in living tissues. The Biochemical Journal, 418(3), 567–574. https://doi.org/10.1042/BJ20081949
Cranfill, P. J., Sell, B. R., Baird, M. A., Allen, J. R., Lavagnino, Z., de Gruiter, H. M., & Piston, D. W. (2016). Quantitative assessment of fluorescent proteins. Nature Methods, 13(7), 557–562. https://doi.org/10.1038/nmeth.3891
Shemiakina, I. I., Ermakova, G. V., Cranfill, P. J., Baird, M. A., Evans, R. A., Souslova, E. A., & Shcherbo, D. (2012). A monomeric red fluorescent protein with low cytotoxicity. Nature Communications, 3, 1204. https://doi.org/10.1038/ncomms2208
Dean, K. M., Lubbeck, J. L., Binder, J. K., Schwall, L. R., Jimenez, R., & Palmer, A. E. (2011). Analysis of red-fluorescent proteins provides insight into dark-state conversion and photodegradation. Biophysical Journal, 101(4), 961–969. https://doi.org/10.1016/j.bpj.2011.06.055
Manna, P., & Jimenez, R. (2015). Time and frequency-domain measurement of ground-state recovery times in red fluorescent proteins. The Journal of Physical Chemistry. B, 119(15), 4944–4954. https://doi.org/10.1021/acs.jpcb.5b00950
Klementieva, N. V., Pavlikov, A. I., Moiseev, A. A., Bozhanova, N. G., Mishina, N. M., Lukyanov, S. A., & Mishin, A. S. (2017). Intrinsic blinking of red fluorescent proteins for super-resolution microscopy. Chemical Communications (Cambridge, England), 53(5), 949–951. https://doi.org/10.1039/c6cc09200d
Acknowledgements
This study was supported by the Russian Science Foundation (RSCF), Grant № 20-14-00255. The funders of the study had no part in the writing, analysis, interpretation, or decision to submit the paper. Experiments were partially carried out using equipment provided by the IBCH core facility (CKP IBCH, supported by Russian Ministry of Education and Science, Grant RFMEFI62117X0018).
Funding
This study was supported by the Russian Science Foundation (RSCF), Grant № 20–14-00255.
Author information
Authors and Affiliations
Contributions
EAP: investigation, formal analysis, software; ASM: validation, investigation; KAL: conceptualization, supervision, resources; EGM: methodology, validation, formal analysis, investigation, visualization; AMB: conceptualization, investigation, project administration, funding acquisition. The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Pushing the limits of flash photolysis to unravel the secrets of biological electron and proton transfer - a topical issue in honour of Klaus Brettel.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Protasova, E.A., Mishin, A.S., Lukyanov, K.A. et al. Chromophore reduction plus reversible photobleaching: how the mKate2 “photoconversion” works. Photochem Photobiol Sci 20, 791–803 (2021). https://doi.org/10.1007/s43630-021-00060-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-021-00060-8