Abstract
The hydrosystem Lac Togo-Lagoon of Aného is located in a watershed where phosphate mining of trace element-rich phosphorites takes place. This activity generates various types of wastes that are dumped into the environment without prior treatment. In addition, there are contributions of its tributaries after leaching of agricultural and urban soils. All this contributes to the degradation of the water quality of this ecosystem. The aim of this study is to assess the level of trace elements contamination of the waters from the hydrosystem Lake Togo-Lagoon of Aného and its spatio-seasonal variations. In the dry and rainy season, the physicochemical parameters of waters were measured according to French standards (AFNOR). The trace elements were determined by atomic absorption spectrometer coupled to a hydride and cold vapour generator. The spatial distribution was evaluated using maps which were performed by ordinary kriging interpolation method of ArcGIS 10.2.2 software. The results showed that the average dry season concentrations were significantly higher (p < 0.0001) than those of the rainy season except that of Zn. The average concentrations (in μg/l) observed for Cd: (39.28; 24.63), Pb: (216.82; 66.44), Cr: (197.00; 69.05) and Ni: (100.06; 66.50), respectively, in dry and rainy season were significantly higher than the WHO standards. However, overall trace elements contamination has been noted with polymetallic contamination indices which reached 4.14 in the dry season and 2.41 in the rainy season. This contamination is mainly of anthropogenic origin and comes from the intrusion of seawater loaded with phosphate effluents in the dry season and the leaching of mining and agricultural soils in the rainy season. The results proved that this lagoon ecosystem is polluted by trace elements mainly caused by phosphate mining. Thus, it deserves special attention for its better management for a sustainable development.
Similar content being viewed by others
1 Introduction
Heavy metal pollution has become a worldwide concern due to the increasing levels of pollution and its obvious impacts on human health. These are of great concern from the public health point of view, and being environmental pollutants it can occur naturally in the environment and can come from industrial or agricultural sources [1]. Thus, the contamination of aquatic ecosystems by toxic substances remains a serious more and more worryingly environmental problem [2]. Anthropogenic activities have been shown to be the main contributors to degradation of aquatic ecosystems’ quality whose sediments are real reservoirs of pollutant accumulation [3,4,5]. Trace elements from anthropogenic activities are frequently detected in sediments and the water column [6, 7]. However, metal contamination of sediments is a threat to water, living species and human health [3].
Trace elements are present in all compartments of the aquatic ecosystem. Thus, they pose serious ecological problems, both by their ubiquitous and persistent nature and by their high toxicity [1, 8]. Due to their non-biodegradability and high persistence, trace elements can be very harmful or even deadly [9] because of their ability to be bioaccumulated by aquatic organisms. This leads to an increase in their concentrations relative to those of the biotope and from one link to another in the food chain [10,11,12].
In Togo, the phosphorite mining activities, which are highly concentrated in trace elements [13,14,15], take place in the Lake Togo watershed. These activities release huge quantities of phosphate waste into the coastal region without any prior treatment [16, 17]. These wastes end up in coastal waters through runoff, leaching and atmospheric depositions. In addition, the Lake Togo-Lagoon of Aného hydrosystem mainly receives inputs from the Haho and Zio rivers after crossing mining, urban and agricultural soils where agrochemicals are used irrationally. Also, this water body is the subject of illegal traffic of petroleum products which are voluntarily or accidentally, dumped into the waters in order to balance the canoes during transport. This study aims to assess the level of trace elements contamination of the waters from the hydrosystem Lac Togo-Lagoon of Aného and its spatio-seasonal distributions. This will identify potential risks for fish species and local populations in order to better plan its management.
2 Materials and methods
2.1 Study area
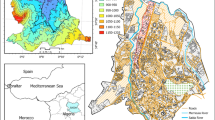
The Lake Togo-Lagoon of Aného Hydrosystem is a part of a lagoon system set in southeastern Togo. This lagoon system set is located between latitudes 6° 14′ 38″ and 6° 17′ 37″ North and longitudes 1° 23′ 33″ and 1° 37′ 38″ East between the villages of Dékpo and Sévatonou in the north-west and the city of Aného in the south-east. It covers a total area of 64 km2 and includes Lake Togo (46 km2), Lagoon of Togoville, which is parallel to the coast and has 13 km length with a width ranging from 150 to 900 m, Lake Zowla (6.55 km2), and the Lagoon of Aného in the Southeast [18]. It communicates almost permanently with the ocean in Aného. In addition, the Aného Lagoon communicates with the Mono River in the East via the Gbaga Channel. With respective lengths of 176 and 140 km and respective flow rates of 9.9 and 5.8 m3/s at the mouth, Zio and Haho are the main tributaries of the lagoon system [19]. The phosphorites mining activities take place in this watershed with the rejection of various kinds of mining wastes. This coastal area of the lagoon complex is under a subequatorial or Guinean climate with two rainy seasons and two dry seasons. Local people are mainly fishermen, farmers and pastoralists. Figure 1 shows the location of the study area and sampling points.
3 Sampling and laboratory analysis
The samplings were carried out at 30 random sites in two seasons: the dry season (March 2016) and the rainy season (October 2016) corresponding, respectively, to the low and high water periods. A total of 30 water samples per season were collected at 30 cm water depth using 0.5 L sterile polyethylene bottles. A reference site (6° 26′ 56,97″ N; 1° 04′ 44,46″ E) was selected and sampled far from the phosphorite mines in the Zio River in order to determine the level of trace elements contamination of lagoon waters. Water intended for the determination of trace elements was acidified to 1% with nitric acid in order to release the trace elements according to French standards (FD T90-523-3, NF EN ISO 5667-3). Temperature and pH were measured using a pH-meter Crison PH 25 (accuracy: ± 0.01 pH for pH and ± 0.1 °C for temperature). The total dissolved solids (TDS) and salinity were measured using a conductivity meter Knick Portamess 911 Cond (error: ± 0.5%). All these parameters were measured in situ. The samples intended for trace elements analysis were transported to the laboratory in coolers containing ice cubes and stored at an air conditioning room temperature (18 °C) and in the dark until analysis [20, 21]. Those used for orthophosphate (PO43−) analysis was not acidified and was refrigerated until analysis within 48 h. The PO43− was analyzed using a molecular absorption spectrometer HACH DR 3800. For this purpose, PhosVer® 3 with the acid persulfate digestion method was used. The elements Cd, Pb, Cr, Cu, Ni, Zn and Mn were analyzed using air/acetylene flame atomic absorption spectrometers (AAS), Varian AA 240 FS type at “Vasile Alecsandri” University of Bacau (Romania) and Thermo Electron S Series type at the University of Lomé (Togo). Concerning Hg and As, they were only analyzed at the University of Lomé using a hydride and cold vapour generator, Thermo Scientific VP 100 type coupled to the AAS Thermo Electron S Series according to French standards (NF T90-112, NF EN ISO 5961, NF EN 1233, NF EN 1483, NF EN ISO 11969). In fact, appropriate volumes of reagents were added to the water samples before analysis (ascorbic acid (10%) + potassium iodide (10%) + hydrochloric acid (37%) for As and hydroxylamine chloride (5%) + potassium permanganate (5%) + hydrochloric acid (37%) for Hg). The reductants used were sodium borohydride (1%) prepared with sodium hydroxide (0.1%) for As analysis and stannous chloride (10%) prepared with hydrochloric acid (30%) for Hg analysis. The mixture of hydrochloric acid (10%), the reductants and the prepared samples or standards is done by the hydride and cold vapour generator. The reaction between these reagents produces an arsenic hydride (AsH3) for As and elemental mercury (Hg0) for Hg. These elements are leaded to a “T” Cell placed on the burner of the spectrometer where they are normally read by the spectrometer using air/acetylene flame for As and no flame for Hg. The standard solutions used for the calibration contain the same reagent like the samples and ranged as follows: 0 µg/l, 1 µg/l, 2,5 µg/l and 5 µg/l. The detection limits of the hydride and cold vapour generator are 0.04 µg/l for As and 0.06 µg/l for Hg with a measurement error of ± 2%. The reagents used for this purpose are from Sigma-Aldrich and Merck.
4 Quality control
The analytical methods have been validated by an internal control. Thus, a laboratory blank sample was prepared. It consists of 1% acidified distilled water using the same acid used for the samples. For As and Hg analysis, all the reagents added to the samples were also added to the blank. These blanks were used in the calibration process of the spectrometer and were regularly measured during the analysis. This allowed to determine the possible contamination and to eliminate the quantization errors. Standard solutions of each trace element were regularly analyzed to verify the accuracy of the results and the internal fidelity of the method. In addition, standard solutions of pH, conductivity and PO43− were used to verify the accuracy of the measurements.
5 Data processing
The contamination indices (CI) and polymetallic contamination indices (PCI) were calculated relative to the reference site as follows [22,23,24]:
where Csample: concentration of the studied sample, Creference: concentration of the reference, n: number of elements. 1 < CI or PCI < 2: less or not contaminated, CI or PCI > 2: contaminated.
The study of spatio-seasonal variation of the trace element contents was carried out using distribution maps performed by the Ordinary Kriging Interpolation method of ArcGIS 10.2.2 software [25,26,27]. In order to determine the interrelationships between the different variables studied as well as the similarities between the different sampling sites, principal component analysis (PCA) and a Pearson correlation analysis were performed [20, 27]. The Student's t test was used to compare the concentrations between seasons. All these analyses were carried out using the STATISTICA 6.1 software.
6 Results
6.1 Physicochemical parameters of waters
The values of temperature, pH, TDS, salinity and orthophosphate of the water in the dry and rainy seasons are depicted in Fig. 2. The temperature values (T°C) vary from 26.5 °C in the rainy season to 33.33 °C in the dry season. The most frequent T°C values ranged from 30 to 30.5 °C and from 31 to 31.5 °C in the dry season whereas they oscillate between 28 and 28.5 °C in the rainy season. The lowest pH value (5.9) was obtained in the rainy season at site S17 while the highest (7.71) was recorded in the dry season at site S29. The most frequent pH values were between 6.8 and 7.2 in the dry season and between 6.8 and 7.4 in the rainy season.
The salinity (Sa) of the waters was between 0.90 g/l recorded at the site S7 during the rainy season and 34.80 g/l during the dry season at the site S29. The most frequent salinity values vary between 5 and 10 g/l then between 15 and 20 g/l in the dry season and between 1 and 2 g/l in the rainy season. The lowest value of total dissolved solids (TDS) (7.70 g/l) was recorded in rainy season at the site S7 while the highest (718 g/l) was recorded in the dry season at the site S29. The most frequent TDS values were between 0 and 200 g/l in the dry season and between 10 and 20 g/l in the rainy season. For orthophosphate (PO43−), their contents range from 0.25 mg/l in the rainy season at the site S7 to 2.20 mg/l in dry season at the site S29. The most frequent contents range from 0.6 to 0.8 mg/l in the dry season and from 0.3 to 0.4 mg/l and 0.6 to 0.7 mg/l in the rainy season.
6.2 Trace elements concentrations in water and quality degree
6.2.1 Spatio-seasonal variations in the trace element content of water
Table 1 indicates that Cd contents vary heterogeneously both in the dry season (CV = 51.59%) and rainy season (57.58%). They oscillate, respectively, between 23.20 and 92.30 μg/l with an average of 39.28 ± 20.27 μg/l and between 1.92 and 61.48 μg/l with an average of 24.63 ± 14.18 μg/l. A significant difference (t = 3.24; p = 0.0019) was noted between seasonal mean contents. The spatio-seasonal distribution of Cd contents (Fig. 3a, d) shows that in the dry season the concentrations increase from upstream to downstream with the highest contents recorded in the lagoon of Aného. This gradient is reversed in the rainy season with the highest concentrations in upstream located in northwestern Lake Togo.
The concentrations of Pb range from 151.05 to 348.35 μg/l with an average of 216.82 ± 52.96 μg/l in the dry season and from 5.36 to 124.24 μg/l with an average of 66. 44 ± 33.73 μg/l in the rainy season. The coefficient of variation (CV) is quite low (CV = 24.42%) in the dry season and quite high in the rainy season (CV = 50.77%). The average Pb content obtained during the dry season is very significantly higher than that of the rainy season (t = 13.12; p = 0.0001). The spatio-seasonal distribution of Pb contents (Fig. 3b, e) shows an increasing gradient from upstream to downstream in the dry season followed by an inversion of this gradient in the rainy season where the lowest levels have been observed in downstream in the Lagoon of Aného.
A heterogeneous variation (CV = 59.14%) of the Cr contents was observed in the dry season whereas it was fairly homogeneous (CV = 33.50%) in the rainy season. These contents ranged, respectively, from 41.10 to 396.90 μg/l with an average of 197 ± 116.51 μg/l and from 22.73 to 123.97 μg/l with an average of 69.05 ± 23.13 µg/l. These seasonal averages contents are very significantly different (t = 5.90; p = 0.0001). In the dry season, the lowest Cr levels were located in the south-east of Lake Togo while the highest are found in the lagoon of Aného (Fig. 3c, f). In the rainy season, a decreasing concentration gradient from upstream to downstream is noted with the highest concentrations recorded in northwestern Lake Togo.
The average Ni content obtained in the dry season (100.065 ± 9.48 μg/l) is significantly higher (t = 2.53; p = 0.014) than that of the rainy season (66.50 ± 41.47 μg/l). The Ni contents vary, respectively, from 15.60 to 185.40 μg/l and from 2.99 to 175.20 μg/l. These variations are heterogeneous both in the dry season (CV = 59.44%) and in the rainy season (62.36%). The lowest levels of Ni were located in the north-west of Lake Togo in the dry season and in lagoon of Aného during the rainy season (Fig. 4a, d). The highest contents were located in the south-east of Lake Togo and in the lagoon of Togoville during the dry season while in the rainy season they were located in the north-west and center of Lake Togo and in the lagoon of Togoville.
The Cu contents ranged from 85.11 to 126.93 μg/l in the dry season and from 49.60 to 77.54 μg/l in the rainy season. These variations are homogeneous both in dry season (CV = 9.39%) and in rainy season (CV = 9.57%). The average dry season concentration (105 ± 986 μg/l) was very significantly higher (t = 18.66, p = 0.0001) than that of the rainy season (65.24 ± 6.25 μg/l). The spatial distribution of Cu contents (Fig. 4b, e) shows that the lowest contents were located in southeastern Lake Togo during the dry season and in the south-west of Lake Togo during the rainy season. As for the highest contents, they were located in the lagoons of Togoville and Aného during the rainy season and only in the lagoon of Togoville in the dry season.
The average contents of Hg, As, Zn and Mn were, respectively, 0.58 ± 0.26 μg/l, 2.97 ± 1 μg/l, 11.48 μg/l and 38.86 μg/l in the dry season and 0.18 ± 0.13 μg/l, 1.95 ± 0.68 μg/l, 27.79 μg/l and 18.64 μg/l in the rainy season. They vary, respectively, from 0.19 to 0.96 μg/l, from 0.99 to 4.86 μg/l, from 3.60 to 35.40 μg/l and from 13.60 to 67.80 μg/l in the dry season and from 0.02 to 0.5 μg/l, from 0.86 to 3.23 μg/l, from 12.46 to 64.19 μg/l and from 1.50 to 56.52 μg/l in the rainy season. Unlike other concentrations that have a fairly homogeneous variation, those of Zn (CV = 69.48%) in the dry season, Hg (CV = 72.37%) and Mn (CV = 80.88%) in the rainy season vary in a heterogeneous way. The mean dry season concentrations are very significantly higher than those of the rainy season for elements Hg (7.39; p = 0.0001), As (t = 4.64; p = 0.0001) and Mn (t = 5.37; p = 0.0001). On the other hand, the average Zn contents in the dry season are very significantly lower than that of the rainy season (t = − 6.58, p = 0.0001).
During the dry season, the highest Hg concentrations were located in the Lake Togo with the maximum value at the site S17 in the southeastern zone (Fig. 4c, f). During the rainy season, an increasing concentration gradient from upstream to downstream was observed with the highest concentrations recorded in the Lagoon of Aného. The spatial distribution of As (Fig. 5a, d) is irregular with most of the water body covered by contents between 2.54 and 4.86 μg/l in the dry season and between 1, 49 and 3.23 μg/l in the rainy season.
During the dry season, an increasing gradient from upstream to downstream was observed for Zn contents with the highest concentrations recorded in the Lagoon of Aného. In the rainy season, high contents of Zn were found in southeastern Lake Togo and in the Lagoon of Togoville, while the lowest contents occupied the Lake Togo (Fig. 5b, e). The highest concentrations of Mn are located in central and northwestern Lake Togo, respectively, during the dry season and the rainy season (Fig. 5c, f). With the exception of Ni, Cu and Hg in the dry season then of Pb and Cu in the rainy season, the concentrations of all the other trace elements positively skew toward their lowest values. Indeed, their skewness values vary from 0 for Mn in the dry season to 1.94 for Zn in the rainy season. Thus, for these trace elements, the majority of the sites presents the lowest concentrations values. This skewness is more noticeable for Cd, Pb and Zn in the dry season and for Cr, Ni, Hg and Zn in the rainy season with skewness greater than 1 and confirmed by their median values which are lower than the average values. The kurtosis values indicate that in the dry season, most of the trace elements (Cr, Ni, Cu, Hg, As, Mn) present concentration values which are more dispersed and relatively far from their mean (kurtosis < 0). However, in the rainy season, the concentration values are less dispersed and relatively closer and more grouped around the averages for Cd, Cr, Ni, Cu, Hg, Zn, Mn (kurtosis > 0). The order of decreasing abundance of trace elements is as follows for each season: Pb > Cr > Cu > Ni > Cd > Mn > Zn > As > Hg, in the dry season and Cr > Ni > Pb > Cu > Zn > Cd > Mn > As > Hg, in the rainy season. It is found that during both seasons Hg and As are the most weakly represented elements.
6.2.2 Comparison of trace element concentrations with water quality standards
During the dry season, 100% of Cd and Pb levels were above the WHO standards, against 96.67% and 93.33%, respectively, in the rainy season (Fig. 6a, b). The pollution factors (PF) of Cd and Pb reached, respectively, 30.77 times and 34.84 times the WHO standards during the dry season at the site S30 in lagoon of Aného. On the other hand, these PF reached 20.49 times the standard for Cd and 12.42 times the standard for Pb, respectively, in the sites S1 and S5 of Lake Togo, in the rainy season. The Cr contents recorded in the dry season were 96.67% higher than the WHO standard against 90% in the rainy season (Fig. 6c), with the respective highest PF of 7.94 times the standard in the lagoon of Aného and 2.48 times the standard in the Lake Togo. In the dry season, 63.33% of Ni contents are above the WHO standard with PF reaching 2.65 times the standard against 40% in the rainy season with the highest PF of 2.58 times the standard (Fig. 6d). Table 1 shows that all contents of Cu, As, Zn and Mn recorded during the two seasons are well below the WHO standard for water intended for human consumption.
Table 2 presents Student t test results between seasonal mean contents and standards. It indicates that during the dry season, the average Cd, Pb, Cr and Ni contents of the water were significantly higher than the WHO standards while the average contents of Cu, Hg, As, Zn were significantly lower. In the rainy season, the mean concentrations of Cd, Pb and Cr were significantly higher than the WHO standards, whereas those of Cu, Hg, As, Zn and Mn were significantly lower. It is noted that there is no significant difference between the average Ni content and the WHO standard. However, it was slightly lower.
6.2.3 Trace elements contamination degree of waters
Table 3 indicates that in the dry season, the highest contamination indices (CI) ranged from 2.45 for Cu to 12.31 for Cd, while in the rainy season they ranged from 1.41 for Pb to 8.20 for Cd. During the dry season, all sites were contaminated by Cd. However, most sites were contaminated by Pb (90%), Cr (66.67%), Ni (56.67%), Cu (60%), Hg (50%) and Mn (60%). Only 40% and 20% of sites for As and Zn, respectively, had CI greater than 2. In the rainy season, the elements that had contaminated (CI > 2) the waters in most of the sites were Cd (60%) and Zn (96.67%). Thus, Cr (6.67%), Ni (23.33%), As (13.33%) and Mn (10%) only contaminated (CI > 2) in less than 25% of the sites studied.
The averages of contamination indices show that in the dry season the lagoon system waters were heavily contaminated by elements Cd, Pb, Cr, Ni, Cu and Mn (CI > 2). These values vary from 2.13 for Cu to 5.24 for Cd (Table 3). On the other hand, a low contamination was noted for Hg, As and Zn (1 < CI < 2). During the rainy season, Cd and Zn had strongly contaminated the waters of the hydrosystem (CI > 2). However, these waters were weakly contaminated by Cr, Ni, Cu and As (1 < CI < 2). These results show the following decreasing order of trace elements contamination: Cd > Cr > Pb > Ni > Mn > Cu > As > Hg > Zn in dry season and Zn > Cd > Ni > Cu > As > Cr > Mn > Pb > Hg in the rainy season.
The polymetallic contamination indices (PCI) representing the resultant of the combined effects of all trace elements studied were determined to illustrate their common impact by site. These values varied from 1.57 to 4.14 in the dry season and from 1.04 to 2.41 in the rainy season (Table 3). It is observed that 93, 33% of sites were highly contaminated by trace elements with PCI > 2 in the dry season against 16.67% in the rainy season. Thus, the waters were more contaminated by trace elements in the dry season than in the rainy season.
The spatial distribution of PCI values (Fig. 7) shows that during the dry season the metallic contamination of the waters from the hydrosystem Lake Togo-Lagoon of Aného presented an increasing gradient from upstream to downstream with the highest contaminations recorded in the lagoon of Aného. However, during the rainy season, a decreasing gradient of metallic contamination was noted with the highest contamination recorded in north-west of Lake Togo.
6.3 Typology of water trace elements contamination
6.3.1 Correlation matrix
The Pearson correlation matrices between the variables studied in the dry season (a) and rainy seasons (b) are presented in Table 4. It shows that in the dry season, significantly strong and positive correlations were obtained between the pH and Cd, Pb, Cr, Cu, Zn and between (TDS, Sa) and Cd, Pb, Cr, Ni, Cu, Zn. This was also observed between PO43− and Cd, Pb, Cr, Zn. However, the correlations noted between Hg and PO43− are significantly negative (r = − 0.39). In addition, significant correlations were recorded between Cd and Pb, Cr, Cu, Zn, between Pb and Cr, Cu, Zn, between Cr and Cu, Hg, Zn, between Ni and Cu, Mn then between Cu and Zn, Mn. During the rainy season, significantly negative correlations were obtained between (TDS, Sa) and Cd, Pb, Cr, Ni. However, the correlations between (TDS, Sa) and Cu, Hg were significantly positive. A considerable decrease in the correlations between the parameters (pH, PO43−) and the trace elements was observed. Concerning the trace elements in the rainy season, significant correlations were obtained between Cd and Pb, Cr, Ni, Cu, Hg, Mn, between Pb and Cu, Hg, between Cr and Ni, Hg, between Ni and Zn, between Cu and Hg then between Zn and Mn.
6.3.2 Eigen values and projection of variables and cases (sites) in the factorial plan F1 × F2
The Eigen values and the variances explained, resulting from the PCA, are presented in Table 5. This table shows that in the dry season, the first four factors explain 85.32% of the total variance with F1: 52.91%; F2: 17.36%; F3: 8.94%; F4: 6.11% while in the rainy season, they explain 74.11% of the total variance including F1: 35.88%; F2: 17.33%; F3: 13.36%; F4: 7.55%. The factorial plane (F1 × F2) holds 70.27% and 53.21% of the total information, respectively, in the dry season and in the rainy season. Therefore, these factorial axes hold most of the information contained in the data that can explain the chemical factors controlling the distribution of trace elements in the waters.
The projection of the variables in the F1 × F2 factorial plan is depicted in Fig. 8. In the dry season (Fig. 8a), the F1 axis (52.91%) is negatively defined by the parameters pH, Sa, TDS, PO43− and trace elements Cd, Pb, Cr and Zn. The F2 axis (17.36%) is defined in its negative part by Ni and Mn and in its positive part by Cu. In the rainy season (Fig. 8b), the axis F1 (35.88%) is defined in its negative part by Sa, TDS and Hg and in its positive part by Cd, Pb, Cr. The main contributors of the F2 axis (17.33%) are Ni, Cu, Zn. In addition, Table 5 indicates that the axes F3 and F4 are, respectively, defined by As in the positive part and by Hg in the negative part during the dry season. In the rainy season, the F3 axis is positively defined by pH and Mn while the F4 axis is positively defined by PO43−.
The projection of the cases (sites) in the F1 × F2 factorial plan (Fig. 9) allowed to distinguish three types of water in the Lake Togo-Lagoon of Aného hydrosystem during both seasons. In the dry season (Fig. 9a), the first type (G1) groups sites S1-S20 of Lake Togo. These waters are characterized by the lowest pH values, low mineralization and low concentrations of Cd, Pb, Cr, Zn. In addition, high contents of Ni, Hg and Mn have been noted in sites S11–S20 in southeastern Lake Togo while sites S1–S10 in northwestern Lake Togo are characterized by high contents of Cu. This zone of water is the most weakly influenced by marine waters. The second type (G2) includes sites S21–S26 located in the lagoon of Togoville and part of the lagoon of Aného whose waters are characterized by higher pH, medium mineralization, relatively high concentrations of Cd, Pb, Cr, Zn and high Cu contents. This group is the moderately influenced by marine waters. The third type (G3) is constituted by sites S27–S30 in the Lagoon of Aného. These waters are characterized by high pH values, high mineralization and high contents of Cd, Pb, Cr, Zn. This group is the nearest zone to the ocean. It is not only the most strongly influenced by the ocean but also the most threatened by the urban effluents of Aného city.
During the rainy season (Fig. 9b), the first type of water (G1) groups the S1–S7 sites in north-west of Lake Togo. These waters are characterized by low mineralization and very high contents of Cd, Pb, Cr, Ni, Mn. This group of water is the most influenced by the floodwaters of Zio and Haho rivers with no marine influence. The second type (G2) is formed by the waters of the sites S8–S25, which have rather low mineralization, rather high concentrations of Cd, Pb, Cr, Ni, Hg, Zn. These sites are moderately influenced by the ocean. The third type of water (G3) includes the sites S26–S30 located in the Lagoon of Aného. These waters are characterized by higher pH values, high mineralization, low contents of Cd, Pb, Cr, Ni and high contents of Hg and Cu. This group of sites are the most strongly influenced by marine waters and urban effluents.
7 Discussion
The spatial variations in salinity and TDS show an increase from upstream to downstream during both seasons. This highlights a strong marine influence on the waters of the Lake Togo-Lagoon of Aného hydrosystem by the intrusion of marine water which is very concentrated in dissolved salts. This phenomenon is more marked during the dry season [19, 28, 29]. In fact, the intrusion of seawater leading to the increase in salinity and TDS values is favored by low flow rate of the lagoon water, low water and high tides. These seawaters progressively go up to the upstream of the hydrosystem by diffusion favored by the mechanical mixing of the waters. These waters are unhealthy for human consumption according to WHO guidelines. In contrary to the pH, the total dissolved solids (TDS) and salinity values are higher than the WHO standard which is 1 g/l and 0.1 g/l, respectively [30, 31]. In addition, the orthophosphate (PO43−) values are higher than the French standards which is 0.5 mg/l [32]. These results are in agreement with those obtained in other coastal lagoons such as Ahémé Lake and Nokoué Lake in Bénin [33, 34] and Grand Lahou Lagoon in Côte d’Ivoire [35]. But these results were higher than those observed in lagoons of Lomé for TDS [36] One of the important ecological factors in aquatic ecosystems remain the temperature. It is involved in most physicochemical and biochemical reactions. Indeed, the temperature influences the solubility of dissolved salts and gazes such as oxygen. This may influence the distribution of chemical elements in aquatic environments. In addition, it has a catalytic effect on enzymes of living species [21, 37].
Trace elements have adverse consequences for aquatic ecosystems and human health. This has led several states, international organizations and environmental agencies to set threshold values for the protection of aquatic organisms and human health. Thus, in accordance with WHO standards, the waters of the Lake Togo- Lagoon of Aného hydrosystem are polluted by the traces elements Cd, Pb, Cr, Ni and are therefore unfit for human consumption. The average contents of Cd, Pb and Cr during both seasons and Ni during the dry season are significantly higher than the WHO standards (Table 2) set at 3, 10, 50 and 70 μg/l, respectively, for Cd, Pb, Cr and Ni. However, 40% of the sites studied have Ni contents above the standard during the rainy season (Fig. 6). It is noted that the Cu, Hg, As, Zn and Mn contents of these waters are in accordance with WHO standards which are, respectively, set at 2000, 6, 10, 3000 and 400 μg/l [30, 31]. With regard to the protection of aquatic life, the average concentrations of both seasons show that the waters of this lagoon complex can cause harmful effects, linked to the trace elements (Cd, Pb, Cr, Cu and Hg), for the aquatic life based on Canadian standards which are 0.2–1.8 μg/l, 1–7 μg/l, 2–20 μg/l, 2–4 μg/l and 0.1 μg/l, respectively. However, the average contents of Ni, As and Zn are safe in accordance with Canadian standards for the protection of aquatic life which are: Ni (25–150 μg/l), As (50 μg/l) and Zn (30 μg/l) [38].
In addition, the contamination indices calculated in relation to the reference site indicate that the lagoon waters are slightly to highly contaminated by all the trace elements except Pb, Hg and Mn in the rainy season. This is confirmed by the polymetallic contamination indices (PCI), which are all greater than 1 (Table 3). The recorded concentrations confirm those obtained by Razaie-Boroon et al. [39] in lake sediments and Ouro-Sama et al. [40] in the same ecosystem, for Cd and Pb. The concentrations of trace elements recorded in this study are higher than those obtained in the lagoon system of Lomé in Togo for Cd, Pb, Cr, Ni and Cu [36], in Kpeshie lagoons in Ghana for Cd, Pb and Ni [41], in Muni and Sakumo II Lagoons in Ghana for Cd, Pb and Cu [42], in the Lake Nokoué in Benin for Cd and Pb [43], in the estuary of Konkouré river in Guinea for Cd [44] and in the estuary of Bas Loukkos in Morocco for Cd, Pb, Cr, Cu and Zn [45]. On the other hand, these concentrations are lower than those recorded in the lagoon system of Lomé for Hg, As, Zn and Mn [36], in the Muni and Sakumo II lagoons for Cu, Zn and Mn [42], in the Lake Nokoué for Hg [43] and in the Konkouré river estuary for Pb, Ni, Zn and Hg [44]. It is observed that the Hg, Zn and Mn contents recorded in this study generally remained lower than those observed in the lagoon and estuarine ecosystems mentioned above, contrary to the Cd contents which are always higher. This Cd enrichment in waters of the hydrosystem compared to other lagoons and estuaries could be due to phosphate mining activities in the region. Indeed, it is known that the natural phosphorite of Hahotoe-Kpogamé is highly concentrated in trace elements, particularly the Cd [13, 14] (Table 6).
During the dry season, an increase in concentrations from upstream to downstream was observed for most of the trace elements with the exception of Zn. This spatial variation indicates that the trace elements contamination of the ecosystem mainly originates from the ocean via the intrusion of marine waters loaded with effluents discharged by the phosphates processing plant. This is favored by high tides and low water in the hydrosystem associated to the low flow rate of water (average from 2015 to 1016 = 1.76 m3/s) [53]. However, these effluents still contain significant amounts of phosphorites and trace elements [15, 54, 55]. This is confirmed by the strong correlation between the parameters (Sa, TDS, PO43−) and the traces elements (Cd, Pb, Cr, Cu, Zn) giving them a common origin (Table 4; Fig. 8a). On the other hand, the origin of this contamination becomes essentially continental, during the rainy season. This is due to the inflow of floodwaters from the Zio and Haho rivers and runoff after leaching of mining and agricultural soils characterized by the irrational use of agrochemicals (pesticides and fertilizers). The reduction in marine influence was possible because of the higher water flow rate whom average from 2015 to 2016 was 18.51 m3/s [53]. These agrochemicals can release trace elements such as Cd, Hg, Pb, Al, As, Cr, Cu, Mn, Ni, Zn and Sn [21, 56]. This continental origin of trace elements, in the rainy season, was concretized by the multivariate analyzes such as the principal component analysis (PCA) which showed a lack of correlation between the parameters originate from the ocean (TDS, Sa) and trace elements except Hg. These trace elements present negative correlations with the parameters (TDS, Sa). In addition to these sources, the trace elements of this lagoon complex can also come from the leaching of household waste, domestic effluents, atmospheric depositions, the combustion of fossil hydrocarbons by the phosphate plant and vehicles etc. [21, 57]. Also, it is necessary to add the illicit transport of adulterated petroleum products on the lagoon complex resulting in accidental or voluntary spills in the lagoon waters. Indeed, Biney et al. [56] report that the fuels contain the elements Cd. Ni, Hg, Cu, Fe, Mn, Pb, Cd. The strong correlations obtained between the traces elements Cd, Pb and Cr, during both seasons, could indicate a common source of contamination and a similar behavior of these elements in the physicochemical processes of the environment [3].
The increase in trace element concentrations in the dry season can be explained by the lack of precipitation and the evaporation phenomenon which leads to a decrease in the water level [52, 58]. In fact, the temperatures recorded in the dry season (29–33.3 °C) are significantly higher than those of the rainy season (26.5–28.8 °C). In addition, this could be explained by the phenomena of desorption and diffusion of the trace elements in the dissolved phase from the sediments due to the rather favorable physicochemical conditions recorded during this season (slightly acidic pH, high temperature and high salinity) [59]. Indeed, this phenomenon is favored by acid pH, high temperature, high salinity, high hardness and high oxygenation [60,61,62]. On the other hand, the decrease in trace element contents during the rainy season could be explained by the entry of floodwaters, which are less concentrated in trace elements, via the Zio and Haho rivers and the direct precipitation waters. This causes dilution of pre-existing lagoon waters strongly influenced by the ocean, followed by a decrease in concentrations [63, 64]. The seasonal variation of the origin of trace elements contamination has been evidenced by the PCA through the inversion of the positive correlation between the trace elements and the mineralization parameters (TDS, Sa) mainly brought by the ocean (Table 4; Fig. 8). In addition, PCA showed strong and positive correlations between some trace elements, particularly Cd with Pb and Cr during both seasons (Table 4; Fig. 8). This indicates that these trace elements exhibit the same physicochemical processes, and the same behaviors during their transport are from the same source of pollution [65, 66]. Thus, Cd, Pb and Cr come mainly from phosphate whatever the season is.
The contamination of aquatic ecosystems by trace elements poses a threat to aquatic life because of their bioaccumulation in the tissues of living species [10, 12, 21, 67]. In fact, a slowdown in the growth of young Pimephales promelas individuals whose parents had been exposed to 0.26 μg/l of inorganic Hg was observed. In addition, reproductive inhibition in the form of reduction in spawning has been noted [68]. This creates ecological and public health problems. Indeed, discharges of mining effluents into aquatic environments have caused adverse effects on living organisms, particularly fish in Buttle Lake in Canada [69]. In addition, bioaccumulation phenomenon of trace elements has been observed in aquatic living species in coastal zone of Togo [40, 70, 71]. However, the toxicity of the trace elements to the aquatic biocenosis depends on the physicochemical conditions of the environment (pH, redox potential, dissolved oxygen, conductivity, salinity). These parameters influence the transport, stability, bioavailability and distribution of different pollutants in the aquatic ecosystem [72, 73].
8 Conclusion
The waters of the Lake Togo-Lagoon of Aného hydrosystem are under the influence of anthropogenic activities which is reflected in the contamination of the waters by trace elements. Thus, these waters are contaminated by Cd, Pb, Cr and Ni with average concentrations that are significantly above WHO standards during both seasons. Concerning the average concentrations of Cu, Hg, As, Zn and Mn, they all remained below these standards during both seasons. However, there is a global metal contamination of the water compared to the reference site with polymetallic contamination index (PCI) greater than 1. This contamination is mainly from anthropogenic sources and is due to agricultural activities in the Lake Togo watershed and the various wastes discharged by the extractive phosphates industry. These pollutants enter the hydrosystem during the dry season, mainly through the intrusion of ocean water containing phosphate effluents and in the rainy season by fluvial inflow with floodwaters that have leached mining and agricultural soils. This state of contamination requires the implementation of a sustainable management strategy to preserve this ecosystem in order to avoid public health problems.
References
Smita Achary M, Satpathy KK, Panigrahi S, Mohanty AK, Padhi RK, Biswas S, Prabhu RK, Vijayalakshmi S, Panigrahy RC (2017) Concentration of heavy metals in the food chain components of the nearshore coastal waters of Kalpakkam, south east coast of India. Food Control 72:232–243
Gouin CA, Aka N, Adiaffi B, Bamba BS, Soro N (2016) Pollution saisonnière des sédiments de lagune par des métaux lourds (Cu, Pb et Zn) en zone tropicale humide: cas de la lagune Mondoukou (Sud-Est de la Côte d’Ivoire). Int J Biol Chem Sci 10(2):835–845
Yao KM, Metongo BS, Trokourey A, Boka Y (2009) Assessment of sediments contamination by heavy metals in a tropical lagoon urban area (Ebrié lagoon, Côte d’Ivoire). Eur J Sci Res 34(2):280–289
Chouti W, Mama D, Changotade O, Alapini F, Boukari M (2010) Etude des éléments traces métalliques contenus dans les sédiments de la lagune de Porto-Novo (Sud Bénin). J Appl Biosci 34:2186–2197
Traoré A, Soro G, Ahoussi KE, Bamba BS, Soro N, Biemi J (2014) Niveau de contamination en métaux lourds des sédiments d’une lagune tropicale: la lagune Aghien (Sud-Est de la Côte d’Ivoire). Afrique Science: Revue Internationale des Sciences et Technologie 10(3):73–88
Sun Y, Zhou Q, Xie X, Liu R (2010) Spatial, sources and risk assessment of heavy metal contamination of urban soils in typical regions of Shenyang, China. J Hazard Mater 174:455–462
Huang J, Ge X, Wang D (2012) Distribution of heavy metals in the water column, suspended particulate matters and the sediment under hydrodynamic conditions using an annular flume. J Environ Sci 24:2051–2059
Agah H, Saleh A, Bastami KD, Fumani NS (2016) Ecological risk, source and preliminary assessment of metals in the surface sediments of Chabahar Bay. Oman Sea Mar Pollut Bull 107(1):383–388
Boucheseiche C, Cremille E, Pelte T, Pojer K (2002) Pollution toxique et écotoxicologie: notion de base, Guide technique n°7. Bassin Rhône-Méditerranée-Corse Agence de l'Eau Rhône-Méditerranée-Corse, Lyon
Ogindo BA (2001) Heavy metal pollutants and their concentrations in Fish (Barbus Species) in Sosiani River. Kenya discovery and Innovation 13(3/4):178–183
Manda BK, Colinet G, André L, Manda AC, Marquet J-P, Micha J-C (2010) Evaluation de la contamination de la chaîne trophique par les éléments traces (Cu Co, Zn, Pb, Cd, U, V et As) dans le bassin de la Lufira Supérieure (Katanga/RD Congo). Tropicultura 28(4):246–252
Bastami KD, Afkhami M, Mohammadizadeh M, Ehsanpour M, Chambari S, Aghaei S, Esmaeilzadeh M, Neyestani MR, Lagzaee F, Baniamam M (2015) Bioaccumulation and ecological risk assessment of heavy metals in the sediments and mullet Liza klunzingeri in the northern part of the Persian Gulf. Mar Pollut Bull 94:329–334
Gnandi K (1998) Cadmium et autres polluants inorganiques dans les sols et sédiments de la Région côtière du Togo: une étude géochimique. Thèse de Doctorat, Université Friedrich-Alexander d’Erlangen-Nurenberg, Allemagne
Gnandi K, Tobschall HJ (2002) Heavy metals distribution of soils around mining sites of cadmium-rich marine sedimentary phosphorites of Kpogame and Hahotoe (Southern Togo). Environ Geol 41(5):593–600
Gnandi K, Rezaie B, Edorh AP (2009) The geochemical characterization of mine effluents from the phosphorite processing plant of Kpémé (southern Togo). Mine Water Environ 28:65–73
Gnandi K, Tozo K, Amouzouvi K, Baba G, Tchangbédji G, Killi K, Agbeko K (2006) Impact de l’exploitation minière sur la santé humaine: cas de la fluorose dentaire chez les enfants autours de l’usine de traitement des phosphates de Kpémé (Sud-Togo). Journal de la Recherche Scientifique de l'Université de Lomé, Série A 8(2):195–205
ONUDI/NSE (2007) Réduction des rejets des mines de phosphates du Togo dans le GCLME, Projet de démonstration du Togo. Rapport final. Organisation des Nations Unies pour le Développement Industriel/North-South Environment (ONUDI/NSE)
Millet B (1983) Etude de quelques caractéristiques hydrologiques et hydrochimiques du système lagunaire du lac Togo: années 1981 et, 1982nd edn. Edition ORSTOM, Lomé
Millet B (1986) Hydrologie et hydrochimie d’un milieu lagunaire tropical: le lac, Togo edn. Edition ORSTOM Collection Etudes et Thèses, Paris
El Morhit M, Fekhaoui M, Serghini A, El Blidi S, El Abidi A, Bennaakam R, Yahyaoui A, Jbilou M (2008) Impact de l’aménagement hydraulique sur la qualité des eaux et des sédiments de l’estuaire du Loukkos (côte atlantique, Maroc). Bulletin de l’Institut Scientifique, Rabat, Section Sciences de la Terre 30:39–47
Rodier J, Legube B, Merlet N (2009) L’analyse de l’eau 9è, édition edn. Dunod, Paris
Boust D, Jouanneau JM, Latouche C (1981) Méthodologie d’interprétation des teneurs totales en métaux traces contenus dans les sédiments estuariens et littoraux. Bulletin de l'Institut de Géologie Bassin d’Aquitain 30:71–86
Rosso A, Lafont M, Exinger A (1993) Etude des métaux lourds sur le peuplement d’oligochètes de l’Illet de ses effluents (Haut Rhin France). Ann Limnol 29:295–305
Bennasser LM, Fekhaoui M, Mameli O (2000) Assessment of the metallic contamination of the Low Sebou sediments. Ann Chim 90:637–644
Ghanbarpour MR, Goorzadi M, Vahabzade G (2013) Spatial variability of heavy metals in surficial sediments: Tajan River Watershed, Iran. Sustain Water Qual Ecol 1(2):48–58
Wani MA, Wani J, Bhat M, Kirmani N, Wani ZM, Bhat SN (2013) Mapping of soil micronutrients in kashmir agricultural landscape using ordinary kriging and indicator approach. J Indian Soc Remote Sens 41(2):319–329
Nshimiyimana F-X, Faciu M-E, El Blidi S, El Abidi A, Soulaymani A, Fekhaoui M, Lazar G (2016) Seasonal influence and risk assessment of heavy metals contamination in groundwater, Arjaat village, Morocco. Environ Eng Manag J 15(3):579–587
Konan KS, Kouassi AM, Adingra AA, Dongui BK, Gnakri D (2008) Variations saisonnières des paramètres abiotiques des eaux d'une lagune tropicale: la lagune de Grand-Lahou, Cote d'Ivoire. Eur J Sci Res 21(3):376–393
Kambiré O, Adingra A, Eblin S, Aka N, Kakou A, Koffi-Nevry R (2014) Caractérisation des eaux d'une lagune estuarienne de la Côte d’Ivoire: la lagune Aby. Larhyss J 20:95–110
WHO (2011) Guidelines for drinking-water quality, 4th edn. World Health Organization (WHO) chronicle
WHO (2008) Guidelines for Drinking-water quality: incorporating 1st and 2nd addenda, 3rd edn. World Health Organization (WHO) chronicle Genève, Suisse
MEDAD (2007) Synthèse des valeurs réglementaires pour les substances chimiques en vigueur dans l’eau, l’air et les denrées alimentaires en France au 1er décembre 2007. Ministère de l’Ecologie du Développement et de l’Aménagement Durable (MEDAD)/Direction de la Prévention des Pollutions et des Risque (DPPR), Rapport d'Etudes, N° DRC-07-86177-15736A, France
Dèdjiho CA, Mama D, Tomètin L, Nougbodé I, Chouti W, Sohounhloué CKD, Boukari M (2013) Évaluation de la qualité physico-chimique de certains tributaires d’eaux usées du lac Ahémé au Bénin. J Appl Biosci 70:5608–5616
Konan KS, Kouassi KL, Kouame KI, Kouassi A, Gnakri D (2013) Hydrologie et hydrochimie des eaux dans la zone de construction du chenal du port de pêche de Grand-Lahou, Côte d’Ivoire. Int J Biol Chem Sci 7(2):819–831
Zandagba J, Adandedji FM, Mama D, Chabi A, Afouda A (2016) Assessment of the physico-chemical pollution of a water body in a perspective of integrated water resource management: case study of Nokoué Lake. J Environ Prot 7(5):656–669
Ayah M, Grybos M, Tampo L, Bawa LM, Bril H, Djaneye-Boundjou G (2015) Qualité et pollution des eaux d’un hydrosystème littoral tropical: cas du système lagunaire de Lomé, Togo. Eur Sci J 11(15):95–119
Chapman D, Kimstach V (1996) Selection of water quality variables. In: Chapman D (ed) Water quality and assesments—a guide to use of biota, sediments and water in environmental monitoring, 2nd edn. E and FN Spon, London, pp 56–133
CCME (2008) Recommandations pour la qualité des eaux au Canada Conseil Canadien des Ministres de l’Environnement (CCME), Environnement Canada, Ottawa, Ontario, Canada
Razaie-Boroon MH, Gnandi K, Tomety-Mensah KF (2011) Presence and distribution of toxic trace elements in water and sediments of the southern Togo Rivers watershed, West Africa. Fresenius Environ Bull 20(7a):1853–1865
Ouro-Sama K, Solitoke HD, Gnandi K, Afiademanyo KM, Bowessidjaou EJ (2014) Évaluation et risques sanitaires de la bioaccumulation de métaux lourds chez des espèces halieutiques du système lagunaire togolais. VertigO-La revue électronique en sciences de l'environnement 14(2):2–18
Addo MA, Okley GM, Affum HA, Acquah S, Gbadago JK, Senu JK, Botwe BO (2011) Water quality and level of some heavy metals in water and sediments of Kpeshie Lagoon, Accra. Ghana Res J Environ Earth Sci 3(5):487–497
Tay CK, Asmah R, Biney CA (2009) Trace metal levels in water and sediment from the Sakumo II and Muni Lagoons, Ghana. West Afr J Appl Ecol 16:75–94
Degnon RG, Dahouenon-Ahoussi E, Adjou ES, Soumanou MM, Dolganova NV, Sohounhloue DCK (2012) Heavy metal contamination of the Nokoué Lake (Southern Benin) and the dynamic of their distribution in organs of some fish’s species (Mugil cephalus L. and Tilapia guineensis). J Anim Sci Adv 2(7):589–595
Onivogui G, Balde S, Bangoura K, Barry MK (2013) Evaluation des risques de pollution en métaux lourds (Hg, Cd, Pb Co, Ni, Zn) des eaux et des sédiments de l’estuaire du fleuve Konkouré (Rep. de Guinée). Afr Sci 9(3):36–44
El Morhit M (2009) Hydrochimie, éléments traces métalliques et incidences écotoxicologiques sur les différentes composantes d'un écosystème estuarien (Bas Loukkos). Thèse de Doctorat, Université Mohammed V (Rabat), Maroc
Solitoke HD (2019) Aspects de croissance et bioaccumulation des éléments traces chez un Bivalve (Crassostrea gasar, Dautzenberg, 1891) du complexe Lac Zowla- Lagune d’Aného au Sud du Togo. Thèse de Doctorat, Université de Lomé
Faciu ME, Lazar I, Ifrim I, Ureche C, Lazar G (2014) Exploratory spatial data analysis of heavy metals concentration in two sampling sites on Siret River. Environ Eng Manag J 13(9):2179–2186
Abdul-Razak A, Asiedu AB, Entsua-Mensah REM, deGraft-Johnson KAA (2009) Assessment of the water quality of the Oti River in Ghana. West Afr J Appl Ecol 15:1–12
Kayalto B, Mbofung CMF, Tchatchueng J-B, Ahmed A (2014) Contribution à l’évaluation de la contamination par les métaux lourds de trois espèces de poissons, des sédiments et des eaux du Lac Tchad. Int J Biol Chem Sci 8(2):468–480
Fahssi A, Yahya HSA, Touzani R, Chafi A (2016) Evaluation spatiotemporelle de la contamination métallique des ressources hydriques superficielles de la basse Moulouya (Nord-Est Maroc). J Mater Environ Sci 7(7):2404–2423
Zhang L, Shi Z, Zhang JP, Jiang Z, Wang F, Huang X (2015) Spatial and seasonal characteristics of dissolved heavy metals in the east and west Guangdong coastal waters, South China. Mar Pollut Bull 95:419–426
Chouti W, Mama D, Alapini F (2010) Etude des variations spatio-temporelles de la pollution des eaux de la lagune de Porto-Novo (sud Bénin). Int J Biol Chem Sci 4(4):1017–1029
Avumadi AMD (2019) Dynamique des apports fluviaux dissous et particulaires au Lac Togo : Bilans, origines et devenir, mécanismes et facteurs de contrôle. Thèse de Doctorat, Université de Lomé et Université de Toulouse
Gnandi K (2003) Les déchets miniers phosphatés, sources de la pollution marine au Togo. Journal de la Recherche Scientifique de l’Université de Lomé, Série A 7(2):75–90
Bawa LM, Djaneye-Boundjou G, Boyode BP, Assih BT (2007) Water quality evaluation from Lomé’s lagoon: Effects on heavy metals contamination on fishes. J Appl Sci Environ Manag 11(4):33–36
Biney C, Amzu AT, Calamari D, Kaba N, Mbome IL, Naeve H, Ochumba O, Osibanjo O, Radegonde V, Saad MAH (1994) Etude des métaux lourds. In: Clamari D, Naeve H (eds) Revue de la pollution dans l’environnement aquatique africain. Document technique du CPCA/FAO, pp 37–54. http://www.fao.org/docrep/005/V3640F/V3640F3604.htm#ch3644.3641
Bloundi MK (2005) Etude géochimique de la lagune de Nador (Maroc oriental): Impacts des facteurs anthropiques. Thèse de Doctorat, Université Mohammed V (Rabat) et ULP-EOST Strasbourg I, Maroc et France
Fianko JR, Osae S, Adomako D, Adotey DK, Serfor-Armah Y (2006) Assessment of heavy metal pollution of the Iture Estuary in the Central Region of Ghana. Environ Monit Assess 131:467–473
Van Den Berg CMG (1993) Complex formation and the chemistry of selected trace elements in estuaries. Estuaries 16:512–520
Förstner U, Wittmann GTW (1979) Metal Pollution in the aquatic environment. Springer, New York
Lau SSS, Chu LM (1999) Contamination release from sediment in a coastal wetland. Water Res 33(4):909–918
Rasmussen AD, Andersen O (1999) Cadmium transfer between water and sediments studied in laboratory experiments and with a computer model. Bull Environ Contam Toxicol 63:720–727
Audry S, Blanc G, Schäfer J (2004) Cadmium transport in the Lot-Garonne River system (France)-temporal variability and a model for flux estimation. Sci Total Environ 319:197–213
Mohiuddin KM, Otomo K, Ogawa Y, Shikazono N (2012) Seasonal and spatial distribution of trace elements in the water and sediments of the Tsurumi river in Japan. Environ Monit Assess 184(1):265–279
Chen B, Liang X, Xu W, Huang X, Li X (2012) The changes in trace metal contamination over the last decade in surface sediments of the Pearl River Estuary, South China. Sci Total Environ 439:141–149
Suresh G, Sutharsan P, Ramasamy V, Venkatachalapathy R (2012) Assessment of spatial distribution and potential ecological risk of the heavy metals in relation to granulometric contents of Veeranam lake sediments, India. Ecotoxicol Environ Saf 84:117–124
Hounkpatin AS, Edorh AP, Sèzonlin M, Guédénon P, Elégbédé B, Boni G, Dougnon V, Montcho S, Kéké E, Boko M (2012) Pollution of aquatic ecosystems by heavy metals at Ganvié’s lacustrine city (Benin). Int Res J Biotechnol 3(6):81–87
Snarski V, Olson G (1982) Chronic toxicity and bioaccumulation of mercuric chloride in the fathead minnow (Pimephales promelas). Aquattic Toxicol 2:143–156
Deniseger J, Erickson LJ, Austin A, Roch M, Clark MJ (1990) The effects of decreasing heavy metal concentrations on the biota of buttle lake, Vancouver Island, British Columbia. Water Resour 24:403–416
Gnandi K, Tchangbedji G, Baba G, Kili K, Abbé KD (2006) The impact of phosphate mine tailings on the bioaccumulation of heavy metals in marine fish and crustaceans from the coastal zone of Togo. Int J Mine Water Environ 25(1):56–62
Mélila M, Poutouli W, Amouzou KS, Tchangbédji G, Tchaou M, Doh A (2012) Evaluation de l’impact du rejet des déchets phosphates dans la mer sur la biodiversité marine dans trois localités côtières au Togo à partir des biomarqueurs du stress oxydatif chez Sphyraena barracuda (Heckel, 1843). Int J Biol Chem Sci 6(2):820–831
Bordin G (1991) Distribution et évolution saisonnière du cuivre dissous dans un écosystème estuarien macrotidal d'Europe occidentale (estuaire de l'Aulne, France). Acta Oceanol 14(5):445–456
Péna G, Picot B (1991) Métaux traces dans les sédiments d’une lagune méditerranéenne: l’étang de Thau. Acta Océanol 14(5):459–472
Acknowledgements
This study was co-funded by the International Foundation for Science (IFS) in Sweden (Scholarship: I-2-A -6056-1) and the Romanian Government through ''Eugen Ionescu'' Scholarship 2016 entirely managed by the Francophone University Agency (Agence Universitaire de la Francophonie (AUF)). Thus, the authors wish to thank these institutions. In addition, we express our gratitude to the Faculty of Engineering of “Vasile Alecsandri” University of Bacau (Romania) and to the Laboratory of Management, Treatment and Valorization of Waste of the University of Lomé (Togo).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no potential conflict of interest with this research and in the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ouro-Sama, K., Solitoke, H.D., Tanouayi, G. et al. Spatial and seasonal variation of trace elements contamination level of the waters from the hydrosystem Lake Togo-Lagoon of Aného (South of Togo). SN Appl. Sci. 2, 811 (2020). https://doi.org/10.1007/s42452-020-2593-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-020-2593-7