Abstract
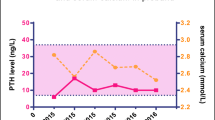
Vitamin D–mediated hypercalcemia may be due to benign or malignant conditions. Recently, loss-of-function mutations in the gene CYP24A1, which encodes 25-hydroxyvitamin D3-24-hydroxylase, have been described which result in reduced degradation of the active 1,25-(OH)2 D3, causing hypercalcemia and hypercalciuria. We describe four patients in whom we identified a novel CYP24A1 homozygous germline mutation (NM_000782.4: c.323A>G; p.(His108Arg). Three of the four patients had a long history of recurrent renal stones. One patient had nephrocalcinosis with significant renal impairment. The other three patients had intermittent hypercalcemia, which in two female patients predominantly occurred during pregnancy with severe gestational hypercalcemia. Germline CYP24A1 mutations are a rare but important cause of vitamin D–mediated hypercalcemia and demonstrate a core phenotype of renal stones, hypercalciuria, and PTH-independent hypercalcemia. However, even in the setting of a homozygous CYP24A1 germline mutation, hypercalcemia may be intermittent with a normal PTH level during the periods of normocalcemia resulting in the need for a high degree of clinical suspicion. Measurement of vitamin D metabolites, in particular the 25-(OH)D3/24,25-(OH)2D3 ratio which is elevated in patients with homozygous CYP24A1 germline mutations, may help provide a clue as to the underlying diagnosis. In women with a history of recurrent calcium-containing renal stones or nephrocalcinosis, we would recommend measurement of serum calcium levels prior to conception and each trimester due to the potential for severe adverse effects on mother and baby if an unidentified CYP24A1 mutation is present.



Similar content being viewed by others
References
Palmer M, Jakobsson S, Akerstrom G, Ljunghall S. Prevalence of hypercalcaemia in a health survey: a 14-year follow-up study of serum calcium values. Eur J Clin Investig. 1988;18:39–46.
Schlingmann KP, Kaufmann M, Weber S, Irwin A, Goos C, John U, et al. Mutations in CYP24A1 and idiopathic infantile hypercalcemia. N Engl J Med. 2011;365:410–21.
Jones G, Prosser DE, Kaufmann M. 25-Hydroxyvitamin D-24-hydroxylase (CYP24A1): its important role in the degradation of vitamin D. Arch Biochem Biophys. 2012;523:9–18.
Molin A, Baudoin R, Kaufmann M, Souberbielle JC, Ryckewaert A, Vantyghem MC, et al. CYP24A1 mutations in a cohort of hypercalcemic patients: evidence for a recessive trait. J Clin Endocrinol Metab. 2015;100:E1343–52.
Woods GN, Saitman A, Gao H, Clarke NJ, Fitzgerald RL, Chi NW. A young woman with recurrent gestational Hypercalcemia and acute pancreatitis caused by CYP24A1 deficiency. J Bone Miner Res. 2016;31:1841–4.
Hedberg F, Pilo C, Wikner J, Torring O, Calissendorff J. Three sisters with heterozygous gene variants of CYP24A1: maternal hypercalcemia, new-onset hypertension, and neonatal hypoglycemia. J Endocr Soc. 2019;3:387–96.
Kwong WT, Fehmi SM. Hypercalcemic pancreatitis triggered by pregnancy with a CYP24A1 mutation. Pancreas. 2016;45:e31–2.
Tebben PJ, Singh RJ, Kumar R. Vitamin D-mediated hypercalcemia: mechanisms, diagnosis, and treatment. Endocr Rev. 2016;37:521–47.
Kaufmann M, Gallagher JC, Peacock M, Schlingmann KP, Konrad M, DeLuca HF, et al. Clinical utility of simultaneous quantitation of 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D by LC-MS/MS involving derivatization with DMEQ-TAD. J Clin Endocrinol Metab. 2014;99:2567–74.
Schwarz JM, Cooper DN, Schuelke M, Seelow D. MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods. 2014;11:361–2.
Ng PC, Henikoff S. Predicting deleterious amino acid substitutions. Genome Res. 2001;11:863–74.
Hawkes CP, Li D, Hakonarson H, Meyers KE, Thummel KE, Levine MA. CYP3A4 induction by rifampin: an alternative pathway for vitamin D inactivation in patients with CYP24A1 mutations. J Clin Endocrinol Metab. 2017;102:1440–6.
Jacobs TP, Kaufman M, Jones G, Kumar R, Schlingmann KP, Shapses S, et al. A lifetime of hypercalcemia and hypercalciuria, finally explained. J Clin Endocrinol Metab. 2014;99:708–12.
Tebben PJ, Milliner DS, Horst RL, Harris PC, Singh RJ, Wu Y, et al. Hypercalcemia, hypercalciuria, and elevated calcitriol concentrations with autosomal dominant transmission due to CYP24A1 mutations: effects of ketoconazole therapy. J Clin Endocrinol Metab. 2012;97:E423–7.
Colussi G, Ganon L, Penco S, De Ferrari ME, Ravera F, Querques M, et al. Chronic hypercalcaemia from inactivating mutations of vitamin D 24-hydroxylase (CYP24A1): implications for mineral metabolism changes in chronic renal failure. Nephrol Dial Transplant. 2014;29:636–43.
Sayers J, Hynes AM, Srivastava S, Dowen F, Quinton R, Datta HK, et al. Successful treatment of hypercalcaemia associated with a CYP24A1 mutation with fluconazole. Clin Kidney J. 2015;8:453–5.
Garcia Rodriguez LA, Duque A, Castellsague J, Perez-Gutthann S, Stricker BH. A cohort study on the risk of acute liver injury among users of ketoconazole and other antifungal drugs. Br J Clin Pharmacol. 1999;48:847–52.
Paul RG, Elston MS, Gill AJ, Marsh D, Beer I, Wolmarans L, et al. Hypercalcaemia due to parathyroid carcinoma presenting in the third trimester of pregnancy. Aust N Z J Obstet Gynaecol. 2012;52:204–7.
Kovacs CS. Maternal mineral and bone metabolism during pregnancy, lactation, and post-weaning recovery. Physiol Rev. 2016;96:449–547.
Dinour D, Davidovits M, Aviner S, Ganon L, Michael L, Modan-Moses D, et al. Maternal and infantile hypercalcemia caused by vitamin-D-hydroxylase mutations and vitamin D intake. Pediatr Nephrol. 2015;30:145–52.
Shah AD, Hsiao EC, O'Donnell B, Salmeen K, Nussbaum R, Krebs M, et al. Maternal hypercalcemia due to failure of 1,25-dihydroxyvitamin-D3 catabolism in a patient with CYP24A1 mutations. J Clin Endocrinol Metab. 2015;100:2832–6.
Dinour D, Beckerman P, Ganon L, Tordjman K, Eisenstein Z, Holtzman EJ. Loss-of-function mutations of CYP24A1, the vitamin D 24-hydroxylase gene, cause long-standing hypercalciuric nephrolithiasis and nephrocalcinosis. J Urol. 2013;190:552–7.
Author information
Authors and Affiliations
Contributions
MSE—conception and design of the project, arranging testing and interpretation of results, drafting the manuscript and critical revision, and approval of the final version.
SDT—acquisition of data (biochemistry) and interpretation, revising the manuscript for critically important intellectual content, and final approval of the version to be published.
SA—acquisition of data (genetics) and interpretation, revising the manuscript for critically important intellectual content, and final approval of the version to be published.
JS—acquisition of data (genetics) and interpretation, revising the manuscript for critically important intellectual content, and final approval of the version to be published.
JVC—conception and design of the project, revising the manuscript for critically important intellectual content, and final approval of the version to be published.
JAUT—testing and interpretation of results, revising the manuscript for critically important intellectual content, and final approval of the version to be published.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
The local institutional review board (Waikato District Health Board) granted permission for the study. Written, informed consent was obtained from all patients for investigation and publication. Ethics approval for gene sequencing was given by the Newcastle and North Tyneside Research Ethics Committee, UK 2003/163.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Medicine
Rights and permissions
About this article
Cite this article
Elston, M.S., Du Toit, S., Alkanderi, S. et al. Variable Phenotypes Seen with a Homozygous CYP24A1 Mutation: Case Report. SN Compr. Clin. Med. 2, 995–1002 (2020). https://doi.org/10.1007/s42399-020-00351-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42399-020-00351-8