Abstract
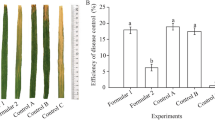
Chemical compounds, which are used on beans to manage common bacterial blight (CBB) disease, cause serious damage to natural ecosystems and often fail to control CBB. A greenhouse experiment was conducted to assess the effectiveness of foliar sprays of Bacillus subtilis str. QST 713 against CBB in a susceptible bean cultivar in comparison with copper hydroxide (CH) and its mixes. A concentration of 108 CFU mL− 1 for Xanthomonas axonopodis pv. phaseoli (Xap) was used to inoculate 21-day-old plants after 3 days of first-time treatment applications. The 2nd and 3rd time treatment applications were applied every 7 days after bacterial inoculation. The disease severity, disease infection rate, AUDPC and disease incidence were significantly reduced over the control treatment. The lowest disease severity, incidence and AUDPC were observed from the mix applications with str. QST713 and CH followed by str. QST 713 and CH alone compared to control treatments. The plants treated with mixes and str. QST 713 showed a reduction of the infection rate by 51.21 and 40.36%, and the disease incidence by 53 and 52.25%, respectively. In addition, the lowest bacterial population per gram of fresh weight of leaves was recorded in plants that had been treated with the mix treatments followed by str. QST713 and CH. Results suggest that integrated management with biocontrol agents is the best strategy for effective control of CBB in sustainable and organic agriculture.





Similar content being viewed by others
Data availability
All materials and data are available and have been presented in the manuscript.
References
Abbasi PA, Weselowski B (2014) Influence of foliar sprays of Bacillus subtilis QST 713 on development of early blight disease and yield of field tomatoes in Ontario. Can J Plant Pathol 36(2):170–178
Abbasi PA, Weselowski B (2015) Efficacy of Bacillus subtilis QST 713 formulations, copper hydroxide, and their tank mixes on bacterial spot of tomato. Crop Prot 74:70–76
Allen DJ, Lenne JM (1998) Disease as constraints to production of legumes in agriculture. In: Allen DJ, Lenne JM (eds) The pathology of food and pasture legumes. CAB International, Wallingford, pp 1–61
Ararsa L, Fikre L, Getachew A (2018) Evaluation of integrated management of common bacterial blight of common bean in Central Rift Valley of Ethiopia. Am J Phytomed Clin Ther 6:1:3
Baker SC, Stavely JR, Thomas CA, Sasser M, Mac. Fall SJ (1983) Inhibitory effect of Bacillus subtilis on Uromyces phaseoli and on development of rust pustules on bean leaves. Phytopathol 73:1148–1152
Balestra GM, Antonelli MG, Fabi A, Varvaro L (1999) Effectiveness of natural products for in vitro and in vivo control of epiphytic population of Pseudomonas syringae pv. tomato on tomato plants. J Plant Pathol 80(3):251
Balestra GM, Heydari A, Ceccarelli D, Ovidi E, Quattrucci A (2009) Antibacterial effect of Allium sativum and Ficus carica extracts on tomato bacterial pathogens. Crop Prot 28(10):807–811
Belachew K, Gebremariam M, Alemu K (2015) Integrated management of common bacterial blight (Xanthomonas axonopodis pv. Phaseoli) of common bean (Phaseolus vulgaries) in Kaffa, Southwest Ethiopia. Malays J Med Biol Res 2:147–152
Belete T, Bastas KK (2017) Common bacterial blight (Xanthomonas axonopodis pv. phaseoli) of beans with special focus on ethiopian condition. J Plant Pathol Microbiol 8:403
Borriss R (2011) Use of plant-associated Bacillus strains as biofertilizers and biocontrol agents in agriculture. In: Maheshwari D (ed) Bacteria in agrobiology: plant growth responses. Springer, Berlin, pp 41–76
Chen XH, Vater J, Piel J, Franke P, Scholz R, Schneider K, Koumoutsi A, Hitzeroth G, Gramme N, Strittmatter AW, Gottschalk G et al (2006) Structural and functional characterization of three polyketide synthase gene clusters in Bacillus amyloliquefaciens FZB42. J Bacteriol 188:4024–4036
Chen XH, Koumoutsi A, Scholz R, Schneider K, Vater J, Sussmuth R, Piel J, Borriss R (2009) Genome analysis of Bacillus amyloliquefaciens FZB42 reveals its potential for biocontrol of plant pathogens. J Biotechnol 140:27–37
CIAT (Centro International de Agricultura Tropical) (1998) Annual report. International centre for Tropical Agriculture, CIAT, Cali, Colombia, 39 pp
Corrêa BO, Soares VN, Sangiogo M, de Oliveira JR, Moura AB (2017) Interaction between bacterial biocontrol-agents and strains of Xanthomonas axonopodis pv. phaseoli effects on biocontrol efficacy of common blight in beans. AJMR 11(32):1294–1302
Donmez MF, Sahin F, Elkoca E (2013) Identification of bean genotypes from Turkey resistance to common bacterial blight and halo blight diseases. . Acta Sci Pol Hortorum Cultus 12(4):139–151
Dursun A, Donmez MF, Sahin F (2002) Identification of resistance to common bacterial blight disease on bean genotypes grown in Turkey. EJPP 108:811–813
Fetene S, Ayalew A (2016) Integrated management of common bacterial blight (Xanthomonas campestris pv. phaseoli) and its effect on seed yield of common bean (Phaseolus vulgaris L.). Int J Life Sci 4(3):336–348
Fortunati E, Mazzaglia A, Balestra GM (2019) Sustainable control strategies for plant protection and food packaging sectors by natural substances and novel nanotechnological approaches. J Sci Food Agric 99:986–1000
Fousia S, Paplomatas EJ, Tjamos SE (2016) Bacillus subtilis QST 713 confers protection to tomato plants against Pseudomonas syringae pv. tomato and induces plant defence-related genes. J Phytopathol 164:264–270
Gilardi G, Manker DC, Garibaldi A, Gullino ML (2008) Efficacy of the biocontrol agents Bacillus subtills and Ampelomyces quisqualis applied in combination with fungicides against powdery mildew of zucchini. JPDP 115(5):208–213
Gilardi G, Gullino ML, Garibaldi A (2010) Evaluation of spray programmes for the management of leaf spot incited by Pseudomonas syringae pv. syringae on tomato cv. Cuore di bue. Crop Prot 29:330–335
Haas D, Defago G (2005) Biological control of soil-borne pathogens by fluorescent Pseudomonas. Nat Rev Microbiol 3:307–319
Horvath DM, Stall RE, Jones JB, Pauly MH, Vallad GE, Dahlbeck D, Scott JW (2012) Transgenic resistance confers effective field level control of bacterial spot disease in tomato. PLoS One 7:1–9
Ibrahim YE, Saleh AA, El-Komy MH, Al-Saleh MA (2016) Bacillus subtilis QST 713, copper hydroxide, and their tank mixes for control of bacterial citrus canker in Saudi Arabia. J Cit Pathol 3(1):1–6
Kinsella K, Schulthess CP, Morris TF, Stuart JD (2009) Rapid quantification of Bacillus subtilis antibiotics in the rhizosphere. Soil Biol Biochem 41:374–379
Lahlali R, Peng G, McGregor L, Gossen BD, Hwang SF, McDonald M (2011) Mechanisms of the biofungicide Serenade (Bacillus subtilis QST713) in suppressing clubroot. Biocontrol Sci Technol 21(11):1351–1362
Lamichhane JR, Osdaghi E, Behlau F, Köhl J, Jones JB, Aubertot JN (2018) Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron Sustain Dev 38(3):28
Lugtenberg B, Kamilova F (2009) Plant-growth promoting rhizobacteria. Annu Rev Microbiol 63:541–556
Nakkeeran S, Fernando WGD, Siddiqui ZA (2005) Plant growth promoting rhizobacteria formulations and its scope in commercialization for the management of pests and diseases. In: Siddiqui ZA (ed) PGPR: Biocontrol and Biofertilization. Springer, Dordrecht, pp 257–296
Opio AF, Allen DJ, Teri JM (1996) Pathogenic variation in Xanthomonas campestris pv. phaseoli, the causal agent of common bacterial blight in Phaseolus beans. Plant Pathol 45:1126–1133
Paçe H, Vrapi H, Gixhari B (2016) Evaluation of some reduced-risk products for management of powdery mildew in greenhouse tomatoes. IJEES 4:505–508
Popovic T, Starovic M, Aleksic G, Zivkovic S, Josic D, Ignjatov M, Milovanovic P (2012) Response of different beans against common bacterial blight disease caused by Xanthomonas axonopodis pv.Phaseoli. Bulg J Agric Sci 18:701–707
Quattrucci A, Ovidi E, Tiezzi A, Vinciguerra V, Balestra GM (2013) Biological control of tomato bacterial speck using Punica granatum fruit peel extract. Crop Prot 46:18–22
Rahman MM, Ali ME, Ali MS, Rahman MM, Islam MN (2008) Hot water thermal treatment for controlling seed-borne mycoflora of maize. Int J Sustain Crop Prod 3:5–9
Roberts PD, Momol MT, Ritchie L, Olson SM, Jones JB, Balogh B (2008) Evaluation of spray programs containing famoxadone plus cymoxanil, acibenzolar-S-methyl, and Bacillus subtilis compared to copper sprays for management of bacterial spot on tomato. Crop Prot 27(12):1519–1526
Robledo-Buriticá1 J, Aristizábal-Loaiza1 JC, Ceballos-Aguirre N, Cabra-Cendales T (2018) Blackberry (Rubus glaucus Benth. cv. thornless) growth under semi-cover and field conditions. Acta Agron 67(2):258–263
Schuster ML, Coyne DP (1981) Source of Phaseolus species resistance and leaf and pod differential reactions to common blight. Hort Sci 18(6):901–903
Shanery G, Finney RE (1977) The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathol 67:1051–1056
Sundin GW, Jones AL, Fulbright DW (1989) Copper resistance in Pseudomonas syringae pv. syringae from cherry orchards and its associated transfer in vitro and in planta with a plasmid. Phytopathology 79:861 865
Sundin GW, Werner NA, Yoder KS, Aldwinckle HS (2009) Field evaluation of biological control of fire blight in the eastern United States. Plant Dis 93:386–394
Valarmathi P, Pareek SK, Priya V, Rabindran R, Chandrasekar G (2013) Compatibility of copper hydroxide (Kocide 3000) with biocontrol agents. IOSR-JAVS 3(6):28–31
Wheeler BEJ (1969) An introduction to plant diseases. Wiley, London., 347 pp
Zitter TA, Drennan JL, Mutschler MA, Kim MJ (2005) Control of early blight of tomato with genetic resistance and conventional and biological sprays. Acta Hortic 695:181–190
Zweerink M, Edison A (1987) Difficidin and oxydifficidin: novel broad-spectrum antibacterial antibiotics produced by Bacillus subtilis. III. Mode of action of difficidin. J Antibiot 40:1692–1697
Acknowledgements
It is our pleasure to acknowledge Scientific Research Projects Coordination Unit of Selcuk University, Turkey for partial funding to support this research. We also sincerely thank Prof. Dr. Ercan CEYHAN, head department of Field Crop, for allowing the use of his modern greenhouse. We extend our gratefulness to the staff members of Plant Protection Department, Selcuk University for helping us during data collection.
Funding
This project was supported by Scientific Research Project Coordination Unit (No.18201104) of Selcuk University, Turkey. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
First author TB was responsible for planning, design and implementation of the experiments, analyzed and interpreted data, and write and organized the manuscript. Second author KKB conceived of the study, participated in its design, revised the paper scientifically and made suitable changes. SF and GMB accurately revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Code availability
Not applicable.
Rights and permissions
About this article
Cite this article
Belete, T., Bastas, K.K., Francesconi, S. et al. Biological effectiveness of Bacillus subtilis on common bean bacterial blight. J Plant Pathol 103, 249–258 (2021). https://doi.org/10.1007/s42161-020-00727-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-020-00727-8