Abstract
Purpose
The aims of this study were to investigate the correlation of sex hormone-binding globulin (SHBG) and the components of metabolic syndrome (MetS) and explore the ability of SHBG to predict MetS in newly diagnosed and untreated patients with polycystic ovary syndrome (PCOS).
Methods
Ninety-eight newly diagnosed and untreated patients with PCOS and 37 healthy volunteers were recruited. A receiver operating characteristic (ROC) curve was used to explore the best cutoff values of SHBG for predicting that the patients with PCOS would fulfill at least one abnormal index of MetS components, at least two abnormal indexes of MetS components, or MetS.
Results
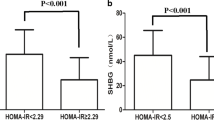
The numbers of patients with PCOS who fulfilled none, one, or two of the MetS criteria items and MetS were 33, 31, 19, and 15, respectively. SHBG was negatively correlated with BMI (r = − 0.615, P < 0.001), systolic blood pressure (SBP) (r = − 0371, P < 0.001), diastolic blood pressure (DBP) (r = − 0.285, P = 0.004), triglycerides (TG) (r = − 0.431, P < 0.001), fasting serum insulin (I0) (r = − 0.549, P < 0.001), HOMA-IR (r = − 0.557, P < 0.001), and plasma glucose 2 h after glucose load (G120) (r = − 0.337, P < 0.001) and positively correlated with high-density lipoprotein cholesterol (HDL-C) (r = 0.629, P < 0.001) in patients with PCOS. The optimal cutoff value of SHBG for predicting MetS in patients with PCOS was 21.3 nmol/L, with a sensitivity of 100.0% (95% CI 78.0–100.0%) and specificity of 85.12% (95% CI 77.5–90.9%).
Conclusions
Sixty-five patients had varying degrees of metabolic abnormalities, accounting for 66.3% of the patients with PCOS. SHBG was associated with metabolic indexes, including BMI, SBP, DBP, TG, I0, HOMA-IR, G120, and HDL-C, and can therefore be employed as a useful index for MetS prediction.



Similar content being viewed by others
References
Escobar-Morreale HF (2018) Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol 14:270–284
Skiba MA, Islam RM, Bell RJ, Davis SR (2018) Understanding variation in prevalence estimates of polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod Update 24:694–709
Soares EM, Azevedo GD, Gadelha RG, Lemos TM, Maranhão TM (2008) Prevalence of the metabolic syndrome and its components in Brazilian women with polycystic ovary syndrome. Fertil Steril 89:649–655
Ranasinha S, Joham AE, Norman RJ, Shaw JE, Zoungas S, Boyle J et al (2015) The association between polycystic ovary syndrome (PCOS) and metabolic syndrome: a statistical modelling approach. Clin Endocrinol 83:879–887
Cussons AJ, Watts GF, Burke V, Shaw JE, Zimmet PZ, Stuckey BG (2008) Cardiometabolic risk in polycystic ovary syndrome: a comparison of different approaches to defining the metabolic syndrome. Hum Reprod 23:2352–2358
Wallace IR, McKinley MC, Bell PM, Hunter SJ (2013) Sex hormone binding globulin and insulin resistance. Clin Endocrinol 78:321–329
Lim SS, Norman RJ, Davies MJ, Moran LJ (2013) The effect of obesity on polycystic ovary syndrome: a systematic review and meta-analysis. Obes Rev 14:95–109
(2004) Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363:157–163
Wang K, Yang QF, Chen XL, Liu YW, Shan SS, Zheng HB et al (2018) Metabolic syndrome and its components predict the risk of type 2 diabetes mellitus in the mainland Chinese: a 3-year cohort study. Int J Endocrinol 2018:9376179
Ehrmann DA, Liljenquist DR, Kasza K, Azziz R, Legro RS, Ghazzi MN (2006) Prevalence and predictors of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab 91:48–53
He Y, Lu Y, Zhu Q, Wang Y, Lindheim SR, Qi J et al (2019) Influence of metabolic syndrome on female fertility and in vitro fertilization outcomes in PCOS women. Am J Obstet Gynecol 221:138.e1–138.e12
Zhu JL, Chen Z, Feng WJ, Long SL, Mo ZC (2019) Sex hormone-binding globulin and polycystic ovary syndrome. Clin Chim Acta
Plymate SR, Matej LA, Jones RE, Friedl KE (1988) Inhibition of sex hormone-binding globulin production in the human hepatoma (Hep G2) cell line by insulin and prolactin. J Clin Endocrinol Metab 67:460–464
Allan CA, McLachlan RI (2010) Androgens and obesity. Curr Opin Endocrinol Diabetes Obes 17:224–232
Patlolla S, Vaikkakara S, Sachan A, Venkatanarasu A, Bachimanchi B, Bitla A et al (2018) Heterogenous origins of hyperandrogenism in the polycystic ovary syndrome in relation to body mass index and insulin resistance. Gynecol Endocrinol 34:238–242
Xargay-Torrent S, Carreras-Badosa G, Borrat-Padrosa S, Prats-Puig A, Soriano P, Álvarez-Castaño E et al (2018) Circulating sex hormone binding globulin: an integrating biomarker for an adverse cardio-metabolic profile in obese pregnant women. PLoS One 13:e0205592
Hong Y, Chen D, Li Y, Li C, Liu Y, Xie M (2013) Serum sex hormone binding globulin profile and its association with insulin resistance in Chinese peri-menopausal women. Endokrynol Pol 64:197–201
Duggan C, Tapsoba JD, Stanczyk F, Wang CY, Schubert KF, McTiernan A (2019) Long-term weight loss maintenance, sex steroid hormones, and sex hormone-binding globulin. Menopause 26:417–422
Kim JJ, Chae SJ, Choi YM, Hwang KR, Song SH, Yoon SH et al (2013) Atherogenic changes in low-density lipoprotein particle profiles were not observed in non-obese women with polycystic ovary syndrome. Hum Reprod 28:1354–1360
Pkhaladze L, Barbakadze L, Kvashilava N (2016) Myo-inositol in the treatment of teenagers affected by PCOS. Int J Endocrinol 2016:1473612
Sepilian V, Nagamani M (2005) Effects of rosiglitazone in obese women with polycystic ovary syndrome and severe insulin resistance. J Clin Endocrinol Metab 90:60–65
Penna IA, Canella PR, Reis RM, de Sá MFS, Ferriani RA (2005) Acarbose in obese patients with polycystic ovarian syndrome: a double-blind, randomized, placebo-controlled study. Hum Reprod 20:2396–2401
Cibula D, Fanta M, Vrbikova J, Stanicka S, Dvorakova K, Hill M et al (2005) The effect of combination therapy with metformin and combined oral contraceptives (COC) versus COC alone on insulin sensitivity, hyperandrogenaemia, SHBG and lipids in PCOS patients. Hum Reprod 20:180–184
Alinezhad A, Jafari F (2019) The relationship between components of metabolic syndrome and plasma level of sex hormone-binding globulin. Eur J Transl Myol 29:8196
Zhang T, Du T, Li W, Yang S, Liang W (2018) Sex hormone-binding globulin levels during the first trimester may predict gestational diabetes mellitus development. Biomark Med 12:239–244
Kumarendran B, O'Reilly MW, Manolopoulos KN, Toulis KA, Gokhale KM, Sitch AJ et al (2018) Polycystic ovary syndrome, androgen excess, and the risk of nonalcoholic fatty liver disease in women: a longitudinal study based on a United Kingdom primary care database. PLoS Med 15:e1002542
Jayagopal V, Kilpatrick ES, Jennings PE, Hepburn DA, Atkin SL (2003) The biological variation of testosterone and sex hormone-binding globulin (SHBG) in polycystic ovarian syndrome: implications for SHBG as a surrogate marker of insulin resistance. J Clin Endocrinol Metab 88:1528–1533
Dahan MH, Goldstein J (2006) Serum sex hormone-binding globulin levels show too much variability to be used effectively as a screening marker for insulin resistance in women with polycystic ovary syndrome. Fertil Steril 86:934–941
(2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 19:41–47
Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R et al (2013) Diagnosis and treatment of polycystic ovary syndrome: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 98:4565–4592
Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W et al (2009) The androgen excess and PCOS society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril 91:456–488
Funding
This study was supported by grants from the National Natural Science Foundation of China (81200619).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Ethical approval was granted by the Ethics Committee of the First Affiliated Hospital of Shantou University Medical College.
Informed consent
Informed consent was obtained from participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fu, C., Minjie, C., Weichun, Z. et al. Efficacy of sex hormone-binding globulin on predicting metabolic syndrome in newly diagnosed and untreated patients with polycystic ovary syndrome. Hormones 19, 439–445 (2020). https://doi.org/10.1007/s42000-020-00219-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-020-00219-5