Abstract
To characterize particle properties, a proper conditioning of aerosol samples is required for most aerosol measurement systems. Ultrafine particles are often generated in high concentrations, resulting in the need for dilution prior to measurement. Reasons for this sample dilution include the prevention of condensation of volatile compounds onto the particle surface, the suppression of chemical reactions and simply to bring the particle concentration below the maximum tolerable value for the measuring instruments. A number of different approaches are taken to dilute samples. In this article, a new type of dilution system designed for submicron aerosols with high concentrations is presented and its performance compared with design expectations. The new dilution instrument is based on a capillary/filter technique. Small capillary transports a tiny proportion of the sample and the greater part will be filtered and used as diluting gas. The shuffling between the two parts takes place inside a filter. Therefore, the dilution ratio is determined by the diameter of the capillary and the flow rate. With this principle, the dilution process is realized in a single step and on a fast time scale. Calibration measurements showed a linear relation between the dilution ratio and the control parameters within ± 2%. The dilution ratio was found to be independent of particle diameter in the size range between 10 and 400 nm. The dilution ratio scan for the proposed system can be varied continuously from moderate to very high dilutions (from 1:10 to 1:103); this range could be extended with minor modifications. The proposed instrument was built on a small and portable scale. The described dilution system opens a wide range of applications for particle sampling.
Similar content being viewed by others
1 Introduction
One of the common uses of aerosol counters is to evaluate the concentration of various particle sizes. Characterization of the sizes of ultrafine particles (diameter smaller than or equal to 100 nm) is commonly done using an electrical classifier equipped with a differential mobility analyzer and a condensation particle counter (Knutson and Whitby 1975). Condensation particle counter can detect aerosol concentration of 1000 particles cm−3 or greater. However, aerosol number concentrations in dusty applications greatly exceed the working range of most current condensation particle counters (CPC) making it necessary to dilute the aerosol prior to sampling. Reducing the concentration of particles in an aerosol is known as dilution. Dilution is a mechanical or physical removal of aerosol particles (or increasing the clean air volume). This is achieved when the aerosol passes through a filter medium, which reduces the number of particles in the aerosol. A variety of clean media are available for use, such as filter paper, activated carbon, diatomaceous earth, and cartridges. Since different media have varying efficiencies, at varying size ranges, the user must select the suitable media for the required particle removal (Rushton et al. 2008).
The ease, speed and cost of dilution make the technique frequently the first used to judge the success of ultrafine particle (UFPs) generation. The use of traditional diluter is still having several inherent drawbacks. Sample dilution might be used to prevent adsorption or condensation of volatile compounds onto the particle surface, to suppress particle coagulation or chemical reactions which change the aerosol composition (Cheng et al. 2002). Conditioning and dilution of dusts are very important for a representative measurement. The design of the sampling and dilution system determines largely what is measured later. It serves to reduce the concentration in the raw dust to a concentration which can be handled by the measurement system (Hueglin et al. 1996). Moreover, the measuring range and life of instruments can be extended by sample dilution. Typically, a good dilution method should not change the particle size distribution.
A number of different approaches are taken to dilute samples. One of them is the use of a particle ejector or simply ejector (Abdul-Khalek et al. 1998) to achieve a rapid dilution. Passing particles through the ejector environment may alter the sizes or the size distribution through a number of processes such as particle breakup, condensation, agglomeration and coagulation. A porous tube diluter also provides control over the dilution process (Ranade et al. 1976; Newton et al. 1980). It has the advantage over the ejector diluter that it does not have problems related to plugging of the flow channels and losses are minor even for submicron particles. The control of the flow is not as straightforward as with the ejector diluter. The cavity diluter captures a fixed volume of aerosol into a cavity before mixing with particle-free air. The venturi operates by drawing a known fraction of particle-free sheath air to mix with an aerosol sample (Yoon et al. 2005). The capillary diluter which uses an aerosol capillary to dilute with filtered air from the original air sample. However, some of these systems suffer from high-diffusion losses for the nanometer-sized particles and thus are not suitable for studies in the nano-size range (Hueglin et al. 1997; Helsper et al. 1998). Often, the objective of sampling and dilution is to obtain the properties of the aerosol as it is at the measurement location. Additionally, a dilution method enables the observation with well-established techniques for measuring particle number concentrations or number size distributions (Hueglin et al. 1997).
The overall aim of this paper is to contribute to existing knowledge regarding methods for diluting and measuring characteristics of ultrafine particles. The dilution system proposed and used in this study is designed to give very fast dilution of the dust with dry air and carried it out in one step. The dilution system studied here differs from the aforementioned systems in two ways: (i) the dilution flow is a user-selectable, set by a capillary orifice, and operated in a closed loop; (ii) The sample inlet is adjustable by a valve to provide balanced flow with the dry air. This system’s performance is evaluated for the transmission efficiency of UFPs aerosols with sizes between 10 and 400 nm. Finally, the dilution effectiveness is determined at different conditions (flow rates, capillary diameter, particle concentration distributions) for submicron particles.
2 Diluter Design
The primary diluter is shown in detail in Fig. 1. Durable polypropylene filter housing (1) contains a connection for the undiluted sample (6). When the aerosol passes through the fibrous air filter (3). The filter traps the UFPs with high efficiency. Four mechanisms act to separate a particle from an air stream and retain it on a filter medium, namely interception, inertial impaction, Brownian diffusion, and electrostatic capture. After that, a particle-free gas is obtained, e.g., air (dilution gas). Filtration is done in a T-type flow configuration (perpendicular to the undiluted sample jet) in mixing chamber. Sealing cover and capillary holder (4) is placed on the left side of filter (3) inside the housing (1). Experimenter can choose filter to meet the requirements of the application. The choice of a capillary tube for extracting the undiluted sample makes it possible to avoid turbulent flow in the sampling part (6). No adhesives, binders, or surfactants are used in the manufacturing process. To speed up mixing, the essentials were the maximization of the contact area of different flow streams (filter length) and the minimization of the diffusing distance inside the mixing chamber (T-type flow and jet colliding). Undiluted sample is extracted through a 0.5-cm-long capillary tube (5) and immediately mixes with the filtered airflow (primary dilution). Swirling flows in mixing chamber are generated by collision between the jet of the undiluted sample passed through the capillary and the filtered air.
3 Experimental Evaluation
The experimental configuration is shown in Fig. 2a, b. The aerosol is generated from a solution of sodium chloride (NaCl) in water using six-jet collision nebulizer (CN 2425 model, BGI). The aerosol flow is provided less than 30 PSI pressure to generate 10–400 nm NaCl particles. After exiting the nebulizer, the aerosol is passed through the drying system using silica gel. Afterward, a set of an electrostatic classifier (Model 3080, TSI) containing a long differential mobility analyzer (DMA) and a condensation particle counter (CPC), (Model 3775, TSI) is used to measure the concentration distribution (in terms of particle size) upstream and downstream of the diluter. The proposed dilution system is based on the capillary technique combined with a high efficiency aerosol filtration system. Small capillary is used to provide a constant-mass aerosol of the undiluted sample stream into a primary dilution (Fig. 1). With this principle, the dilution process is realized in a single step and on a fast time scale (Fig. 2a). Then, this diluted sample flows outside of the diluter (7) through a 10-cm-long transport tube to the venturi throat, where it mixes with the secondary filtered airflow (Fig. 2b). This makes it possible to extract a diluted sample and distribute it to measuring instruments at atmospheric pressure.
One of the common approaches to dilution test is to measure the upstream and downstream of a diluter using particle counter. A typical data interpretation of diluter testing is the dilution ratio (or delta ratio).
Using the proposed dilution system (Fig. 2a), sampling measurement is taken in the upstream and downstream flow of diluter system. Concentration distribution in term of size is shown in Fig. 3. Aerosol generation give a particle sizes ranging from 10 to 400 nm. The concentration distributions give a maximum concentration of 2.25 × 106 #/cm3 within the range of 60 and 70 nm. The stability of upstream concentration is also tested and the concentration distribution remains stable over time, once it reaches equilibrium. Hence, upstream concentrations (after reaching the equilibrium value from initially zero) at different times are measured for all selected flow rates. The deviation factors among the samples throughout all size channels are assessed and its maximum throughout the whole channels did not exceed 4.89%. Using a capillary diameter of 2 mm, the downstream concentrations are 16 times less than the upstream ones (Fig. 3). The maximum downstream concentration of 1.4 × 105 #/cm3 occurs within the range of 60–70 nm. From downstream concentrations, the initial concentrations are reached by multiplying all downstream concentrations for all particles size by the same dilution ratio δ = 16. These results show that the proposed diluter concept almost provides uniform dilution ratios for all particle sizes ranging from 10 to 400 nm, which preserve the initial shape of the concentration distributions.
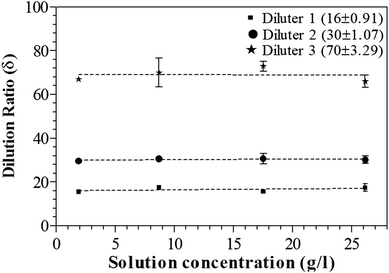
Figure 4 shows the dilution ratio under different solution concentrations as a function of the different diameter capillary. The precision and reproducibility of the dilution method was tested. The dilution ratio value is calculated by dividing the particle concentration distribution of the undiluted and diluted samples. By changing the solutions, we change the particle concentration distribution. The dilution ratios were found to be independent of the solution concentrations, and therefore, of the particle concentration distributions. The impact of capillary diameter on the dilution ratio was also studied. The result shows that the dilution ratio increases with the decrease in diameter of the capillary.
Figure 5 illustrates both the dilution ratio and the particle concentration distributions of the diluted and undiluted samples. The deviation between six consecutive samples throughout all size channels is evaluated. The aim of this test is to check the deviation factor of dilution ratios for all particle sizes. This deviation factor is less than 4.7% for particle sizes within the range of 30 and 300 nm. While it exceeds 5% for particle sizes outside from this range due to very low downstream particle concentrations (Fig. 3). For further study, particle sizes within the range of 30–300 nm were then focused to perform the performance of the diluter system in different conditions. For constant flow rate, the effect of capillary flow rate on dilution ratio is first analyzed (Fig. 6). To increase the dilution ratio, we can add a filter in parallel without making a modification on the proposed dilution system. The flow rate in the capillary is controlled using a valve (Fig. 2b). The dilution ratio is linearly increased and depended to flow rate in the capillary. Then, the both effect of flow rate in the upstream and capillary on dilution ratio is analyzed. These results show that the dilution range can easily be extended by varying the flow rate in the upstream and capillary.
In practice, flow rates are measured and the dilution ratio δ is calculated using Eq. 1. The calibration curves show an excellent linear dependence (R2 > 0.9874) between the flow rate and the dilution ratio (Fig. 6). The error of the calibration curves was within ± 2%. The standard deviation of these data is 1.76%. This discrepancy might be explained by inaccuracies in the fabrication of the capillary, particle diffusion loss, coagulation, the pressure drop across the filter and differences between upstream and downstream pressures. The actual performance of the diluter was also compared with design expectations. At the selected operating condition (5 LPM dilution air flow), the accessible dilution ratios cover the range from performance of the TSI diluter. Because flow measurements were more accurate than the sample measurements, it is likely that actual sample dilution was more accurate than ± 5%. It is concluded that flow dilution is an accurate indicator of sample dilution in this device.
Figure 7 shows that the comparison between a sample diluter (TSI model 3302A) and the proposed dilution system at a total flow rate of 5 LPM. The results showed that the particle size distributions obtained with the proposed dilution system are reliable and compare well with the average measured by TSI device system. An excellent agreement is found for all particle sizes ranging from 30 to 300 nm. Again, the asymmetry for the size distributions of UFPs remains unchanged regardless of the use of the diluter.
Figure 8 shows the particle losses for each size (30–300 nm). The average difference in particle count values is 4.9%. The deviation of the particle concentrations recorded by the CPC as a function of the dilution ratio shows that the dilution efficiency is found to be typically greater than 95% for all size range.
4 Conclusions
A sample dilution system specifically designed to sample UFPs is presented. The design principles are straightforward and may be applied to a number of situations where rapid high dilution ratios are required. The capillary is employed in the first stage. It is capable of rapid dilution on the order of 1:103 and has shown highly repeatable performance. Particle losses and coagulation within the diluter are minimized by design. This dilution system is technically very stable, easy to use and can be built on a low-cost basis. It is found that:
-
The proposed diluter concept provides a uniform dilution ratio for particle sizes ranging from 30 to 300 nm and preserve the initial shape of the concentration distributions.
-
The dilution ratios were found independent of the particle concentration distributions.
-
The dilution ratio increases with the decreasing in diameter of the capillary.
-
The dilution range can easily be extended by varying the flow rate in the upstream and capillary.
-
The particle size distributions obtained with the capillary dilution system are reliable and compare well with the average measured by TSI device system.
That the proposed dilution system can be very helpful in many areas of aerosol measurement and research.
References
Abdul-Khalek IS, Kittelson DB, Graskow BR, Wei Q, Bear F (1998) Diesel exhaust particle size: measurement issues and trends (No. 980525). SAE Technical Paper
Cheng MD, Storey JM, Wainman T, Dam T (2002) Impacts of venturi turbulent mixing on the size distributions of sodium chloride and dioctyl-phthalate aerosols. J Aerosol Sci 33(3):491–502
Helsper JP, Davies JA, Bouwmeester HJ, Krol AF, van Kampen MH (1998) Circadian rhythmicity in emission of volatile compounds by flowers of Rosa hybrida L. cv. Honesty. Planta 207(1):88–95
Hueglin C, Scherrer L, Burtscher H (1996) Description and application of a dilution method for the characterization of particles from emission sources. J Aerosol Sci 27:S311–S312
Hueglin C, Scherrer L, Burtscher H (1997) An accurate, continuously adjustable dilution system (1: 10 to 1: 104) for submicron aerosols. J Aerosol Sci 28(6):1049–1055
Knutson EO, Whitby KT (1975) Aerosol classification by electric mobility: apparatus, theory, and applications. J Aerosol Sci 6(6):443–451
Newton GJ, Carpenter RL, Yeh HC, Peele ER (1980) Respirable aerosols from fluidized bed coal combustion. 1. Sampling methodology for an 18-inch experimental fluidized bed coal combustor. Environ Sci Technol 14(7):849–853
Ranade MB, Werle DK, Wasan DT (1976) Aerosol transport through a porous sampling probe with transpiration air flow. J Colloid Interface Sci 56(1):42–52
Rushton A, Ward AS, Holdich RG (2008) Solid-liquid filtration and separation technology. Wiley, New York
Yoon YJ, Cheevers S, Gerard Jennings S, O’Dowd CD (2005) Performance of a venturi dilution chamber for sampling 3–20 nm particles. J Aerosol Sci 36(4):535–540
Acknowledgements
The authors would like to acknowledge the Institut de Recherche Robert-Sauvé en Santé et en Sécurité du Travail (IRSST) for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Djebara, A., Songmene, V. & Bahloul, A. Performance of a Capillary Dilution System for High-Concentration Sampling of Ultrafine Aerosols. Aerosol Sci Eng 2, 92–97 (2018). https://doi.org/10.1007/s41810-018-0025-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41810-018-0025-5