Abstract
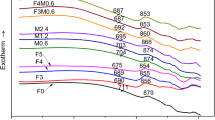
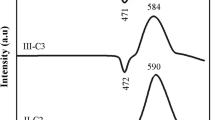
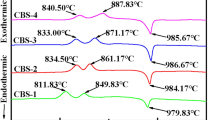
Diopside (CaMgSi2O6) glass-ceramic, a typical derivative of the pyroxene system, has attracted considerable attentions in the fields of anti-erosion, anti-abrasion, light-emitting diode (LED) packaging, or electric circuit substrate materials because of its high mechanical strength, good physical and chemical stability, as well as remarkable low dielectric coefficient and loss. However, the effect of niobium pentoxide (Nb2O5) on the microstructure and property of this technically important glass-ceramic is still not fully understood. Therefore, diopside glass-ceramics with 0 wt%, 0.1 wt%, 0.025 wt%, 0.5 wt%, 1 wt%, 2 wt%, and 4 wt% of Nb2O5 were synthesized respectively by the traditional melting method using Bayan Obo mine tailing and fly ash as the main starting materials. The obtained materials were studied by differential scanning calorimetry, X-ray diffractometry, Raman spectroscopy, scanning electron microscopy, energy-dispersive X-ray spectroscopy, mechanical property, and dielectric property measurements to reveal the effect of Nb2O5 on its microstructure and property. The result shows the addition of Nb2O5 has a clear effect on increasing the formation of diopside crystals and the crystallization of calcium niobate (Ca2Nb2O7) secondary phase at the intergranular areas between diopside crystals. And this latter microstructural change relates closely with the decrease in the average size of diopside crystals and their morphology changing from the ones resembling chrysanthemum flowers to clusters of roughly round islands. Addition of Nb2O5 up to 2 wt% exhibits a near monotonous effect on improving the bending strength. The maximum value of it can reach as high as 236 MPa. The dielectric constant rises from 7 to 9.7 with the increase of Nb2O5 addition from 0 to 4 wt%, while the dielectric loss fluctuates within the range of 0.015 to 0.035. The comparison of the results of this study with those of peers showed the crystallization of diopside crystals was more likely enhanced through the mechanism related to the Nb2O5-induced phase separation in the base glass rather than the normally reported heterogeneous nuclei mechanism reported for Ferric oxide (Fe2O3), chromium trioxide (Cr2O3), and titania (TiO2).






Similar content being viewed by others
References
Holand W., Beall G.H., Glass ceramic technology (Second Edition ed.), John Wiley & Sons,2012
Ouyang, S., Zhang, Y., Chen, Y., Zhao, Z., Wen, M., Li, B., Shi, Y., Zhang, M., Liu, S.: Preparation of glass-ceramics using chromium-containing stainless steel slag: crystal structure and solidification of heavy metal chromium. Sci Rep-UK. 9, 1–9 (2019)
Gao, H.T., Liu, X.H., Chen, J.Q., Qi, J.L., Wang, Y.B., Ai, Z.R.: Preparation of glass-ceramics with low density and high strength using blast furnace slag, glass fiber and water glass. Ceram Int. 44, 6044–6053 (2018). https://doi.org/10.1016/j.ceramint.2017.12.228
Chou, C., Chang, C., Chen, G., Liao, W., Feng, K., Tsao, C.: Control of silver diffusion in low-temperature co-fired diopside glass-ceramic microwave dielectrics. Materials. 11, 55–63 (2018)
Zitani, M.K., Ebadzadeh, T., Banijamali, S., Riahifar, R., Ruessel, C., Abkenar, S.K., Ren, H.: High quality factor microwave dielectric diopside glass-ceramics for the low temperature co-fired ceramic (LTCC) applications. J Non-Cryst Solids. 487, 65–71 (2018). https://doi.org/10.1016/j.jnoncrysol.2018.02.025
Mysen B., Richet P., Silicate glass and melts-properties and structure, Elsevier,2010
He, D., Gao, C., Pan, J., Xu, A.: Preparation of glass-ceramics with diopside as the main crystalline phase from low and medium titanium-bearing blast furnace slag. Ceram Int. 44, 1384–1393 (2018). https://doi.org/10.1016/j.ceramint.2017.09.019
Öveçolu, M.L., Kuban, B., Özer, H.: Characterization and crystallization kinetics of a diopside-based glass-ceramic developed from glass industry raw materials. J Eur Ceram Soc. 17, 957–962 (1997)
Deng, L., Zhang, X., Li, B., Jia, X., Zhang, M., Ouyang, S.: Influence of crucible material on the microstructure and properties of iron rich glass-ceramics. Journal of Wuhan University of Technology-Mater Sci Ed. 33, 49–55 (2018). https://doi.org/10.1007/s11595-018-1784-1
Deng, L., Zhang, X., Zhang, M., Jia, X.: Effect of CaF2 on viscosity, structure and properties of CaO-Al2O3-MgO-SiO2 slag glass ceramics. J Non-Cryst Solids. 500, 310–316 (2018). https://doi.org/10.1016/j.jnoncrysol.2018.08.018
Chen, J., Yan, B., Li, H., Li, P., Guo, H.: Vitrification of blast furnace slag and fluorite tailings for giving diopside-fluorapatite glass-ceramics. Mater Lett. 218, 309–312 (2018). https://doi.org/10.1016/j.matlet.2018.02.020
Li, B., Deng, L., Zhang, H., Jia, X.: Structure and performance of glass-ceramics obtained by Bayan Obo tailing and fly ash. J Non-Cryst Solids. 380, 103–108 (2013)
Voncken J.H.L., The rare earth elements-an introduction, Springer, Switzerland,2016
JR H.D.K., Russak M.A.: Formation of nepheline glass-ceramics using Nb2O5 as a nucleation catalyst. J Am Ceram Soc 382-385, (1974)
Fukumi, K., Sakka, S.: Coordination state of Nb5+ ions in silicate and gallate glasses as studied by Raman spectroscopy. J Mater Sci. 23, 2819–2823 (1988). https://doi.org/10.1007/BF00547456
Lopes, J.H., Magalhães, A., Mazali, I.O., Bertran, C.A.: Effect of niobium oxide on the structure and properties of melt-derived bioactive glasses. J Am Ceram Soc. 97, 3843–3852 (2014). https://doi.org/10.1111/jace.13222
Nowak, N., Cardinal, T., Adamietz, F., Dussauze, M., Rodriguez, V.: Influence of niobium and titanium introduction on optical and physical properties of silicate glasses. Mater Res Bull. 48, 1376–1380 (2013). https://doi.org/10.1016/j.materresbull.2012.12.004
Santos, L.F., Wondraczek, L., Deubener, J., Almeida, R.M.: Vibrational spectroscopy study of niobium germanosilicate glasses. J Non-Cryst Solids. 353, 1875–1881 (2007). https://doi.org/10.1016/j.jnoncrysol.2007.02.018
Pernice, P.: Crystallization of the K2O·Nb2O5·2SiO2 glass: evidences for existence of bulk nanocrystalline structure. J Non-Cryst Solids. 275, 216–224 (2000). https://doi.org/10.1016/S0022-3093(00)00258-1
Aronne, A., Sigaev, V.N., Pernice, P., Fanelli, E., Usmanova, L.Z.: Non-isothermal crystallization and nanostructuring in potassium niobium silicate glasses. J Non-Cryst Solids. 337, 121–129 (2004). https://doi.org/10.1016/j.jnoncrysol.2004.03.120
Sarkisov P.D., Paleari A., Sigaev V.N., Lotarev S.V., Golubev N.V., Stefanovich S.Y., Champagnon B., Vouagner D., and Fargin M.C.: Structure of low-silica glasses in the K2O-Nb2O5-SiO2 system. Theor. Found. Chem. En.+. 47, 1–9 (2013), DOI: https://doi.org/10.1063/1.5081830
Denry, I.L., Holloway, J.A., Gupta, P.K.: Effect of crystallization heat treatment on the microstructure of niobium-doped fluorapatite glass-ceramics. J Biomed Mater Res B Appl Biomater. 100B, 1198–1205 (2012). https://doi.org/10.1002/jbm.b.32684
Denry, I.L., Holloway, J.A., Nakkula, R.J., Walters, J.D.: Effect of niobium content on the microstructure and thermal properties of fluorapatite glass-ceramics. J Biomed Mater Res B Appl Biomater. 75B, 18–24 (2005). https://doi.org/10.1002/jbm.b.30295
Kansal, I., Goel, A., Tulyaganov, D.U., Ferreira, J.: Effect of some rare-earth oxides on structure, devitrification and properties of diopside based glasses. Ceram Int. 35, 3221–3227 (2009). https://doi.org/10.1016/j.ceramint.2009.05.023
Karamanov, A., Pisciella, P., Pelino, M.: The effect of Cr2O3 as a nucleating agent in iron-rich glass-ceramics. J Eur Ceram Soc. 19, 2641–2645 (1999)
Rezvani, M., Eftekhari-Yekta, B., Solati-Hashjin, M., Marghussian, V.K.: Effect of Cr2O3, Fe2O3 and TiO2 nucleants on the crystallization behaviour of SiO2-Al2O3-CaO-MgO(R2O) glass-ceramics. Ceram Int. 31, 75–80 (2005). https://doi.org/10.1016/j.ceramint.2004.03.037
Prencipe M., Mantovani L., Tribaudino M., Bersani D., Lottici and Pier P.: The Raman spectrum of diopside: a comparison between ab initio calculated and experimentally measured frequencies. Eur. J. Mineral. 24, 457–464 (2012), DOI: https://doi.org/10.1371/journal.pone.0226043
Aronne A., Sigaev V.N., Champagnon B., Fanelli E., and Valeria Califano C L.Z.U.B.: The origin of nanostructuring in potassium niobiosilicate glasses by Raman and FTIR spectroscopy. J. Non-Cryst. Solids. 351, 3610–3618 (2005), DOI: https://doi.org/10.1111/nph.16084
Volf, M.B.: Chemical approach to glass. Elsevier, Amsterdam-Oxford-New York-Tokyo (1984)
Marghussian, V.K., Balazadegan, O., Eftekhari-yekta, B.: Crystallization behaviour, microstructure and mechanical properties of cordierite–mullite glass ceramics. J Alloy Compd. 484, 902–906 (2009)
Frederikse H.P.R. (1985) Permittivity (dielectric constant) of inorganic solids., CRC Handbook of Chemistry & Physics. CRC Press, pp. 44-46
Shannon R.D., Dickinson J.E., Rossman G.R.: Dielectric constants of crystalline and amorphous spodumene, anorthite and diopside and the oxide additivity rule. Phys Chem Miner 19, 148–156 (1992)
Goel, A., Tulyaganov, D.U., Kharton, V.V., Yaremchenko, A.A., Ferreira, J.M.F.: The effect of Cr2O3 addition on crystallization and properties of La2O3-containing diopside glass-ceramics. Acta Mater. 56, 3065–3076 (2008). https://doi.org/10.1016/j.actamat.2008.02.036
Shi, Y., Li, B.W., Zhao, M., Zhang, M.X.: Growth of diopside crystals in CMAS glass-ceramics using Cr2O3 as a nucleating agent. J Am Ceram Soc. 101, 3968–3978 (2018). https://doi.org/10.1111/jace.15700
Park, J., Ozturk, A.: Effect of TiO2 addition on the crystallization and tribological properties of MgO-CaO-SiO2-P2O5-F glasses. Thermochim Acta. 470, 60–66 (2008). https://doi.org/10.1016/j.tca.2008.01.018
Li, B., Du, Y., Zhang, H., Jia, X., Zhao, M., Chen, H.: Effects of Iron oxide on the crystallization kinetics of Baiyunebo tailing glass-ceramics. T Indian Ceram Soc. 72, 119–123 (2013)
Karamanov, A., Pisciella, P., Pelino, M.: The effect of Cr2O3 as a nucleating agent in iron-rich glass-ceramics. J Eur Ceram Soc. 19, 2641–2645 (1999)
Li, B., Du, Y., Zhang, X., Zhao, M., Chen, H.: Crystallization characteristics and properties of high-performance glass-ceramics derived from Baiyunebo east mine tailing. Environ Prog Sustain. 34, 420–426 (2015)
Doremus R.H., Glass Science (2nd Edition ed.), John wiley & Sons Inc., 605 Third Avenue, New York,1994
Doremus, R.H.: Structure of inorganic glasses. Annu Rev Mar Sci. 2, 93–120 (1972)
Mernagh T.P., Hoatson D.M.: Raman spectroscopic study of pyroxene structures from the Munni Munni layered intrusion, Western Australia. J Raman Spectrosc 28, 647–658 (1997)
Huanxin, G., Zhongcai, W., Shizhuo, W.: Properties and structure of niobosilicate glasses. J Non-Cryst Solids. 112, 332–335 (1989). https://doi.org/10.1016/0022-3093(89)90548-6
Choi, B.K., Sun, G.N., Kim, E.S.: Microwave dielectric properties of diopside glass-ceramics. Ceram Int. 39, S677–S680 (2013). https://doi.org/10.1016/j.ceramint.2012.10.160
Funding
This study is funded by the projects (0901051701 and 0406091701) supported by “Inner Mongolia Science & Technology Plan”. Sponsorship from the “Inner Mongolia Autonomous Region Science and Technology Major project: Fundamental and key technology research for the integrated exploitation of Bayan Obo Mine with high added value” and “Inner Mongolia autonomous region scientific innovation team of integrated exploitation of Bayan Obo mine multi-metal resource” are also fully acknowledged by all authors of current study. This study is also supported by “The IMUST Innovation Fund (No. 2017QDL-S03).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, M., Gao, J., Shi, Y. et al. Effect of niobium pentoxide (Nb2O5) on the microstructure and properties of the diopside glass-ceramics produced from Bayan Obo mine tailing. J Aust Ceram Soc 56, 1079–1087 (2020). https://doi.org/10.1007/s41779-020-00454-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41779-020-00454-4