Abstract
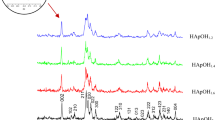
Magnesium substituted hydroxyfluorapatites with the general formula Ca10-xMgx (PO4)6F(OH) with (0 ≤ x ≤ 2.5) were synthesized by the hydrothermal method. The apatite phases were sintered between 1050 and 1150 °C. The substitution of Ca2+ for Mg2+had a strong influence on the densification behavior and mechanical properties of the materials. The density increased simultaneously with the increase of Mg2+content up to x = 1 and then decreased beyond this value. The X-ray diffraction study indicated that the Mg introduced into the solutions was incorporated into the hydroxyfluorapatite. Mechanical properties: Vickers hardness Hv, Young’s modulus E, and shear modulus G were investigated in correlation with the modification of micro-structural characteristics of the sintered materials. According to the obtained properties, these materials possessed sufficient characteristics to be a promising candidate for bone replacement applications.











Similar content being viewed by others
Abbreviations
- HA:
-
Hydroxyapatite
- HFA:
-
Hydroxyfluorapatite
- MHA:
-
Magnesium doped hydroxyapatite
- MHFA:
-
Magnesium doped hydroxyfluorapatite
References
Marra, K.G., Szem, J.W., Kumta, P.N., DiMilla, P.A., Weiss, L.E.: In vitro analysis of biodegradable polymer blend/hydroxyapatite composites for bone tissue engineering. Biomed. Mater. Res. 47, 324–335 (1999)
Kolmas, J., Krukowski, S., Laskus, A., Jurkitewicz, M.: Synthetic hydroxyapatite in pharmaceutical applications. Ceram. Int. 42, 2472–2487 (2016)
Szcześ, A., Hołysz, L., Chibowski, E.: Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interf. Sci. 249, 321–330 (2017)
Pathi, S.P., Lin, D.D., Dorvee, J.R., Estroff, L.A., Fischbach, C.: Hydroxyapatite nanoparticle-containing scaffolds for the study of breast cancer bone metastasis. Biomater. 32, 5112–5122 (2011)
Kim, S.S., Park, M.S., Jeon, O., Choi, C.Y., Kim, B.S.: Poly (lactide-coglycolide)/ hydroxyapatite composite scaffolds for bone tissue engineering. Biomater. 27, 1399–1409 (2006)
Miyaji, F., Kono, Y., Suyama, Y.: Formation and structure of zinc-substituted calcium hydroxyapatite. Mater. Res. Bull. 40, 209–220 (2005)
Ergun, C., Webster, T.J., Bizios, R., Doremus, R.H.: Hydroxylapatite with substituted magnesium, zinc, cadmium, and yttrium. I. Structure and microstructure. Biomed. Mater. Res. 59, 305–311 (2001)
Roy, M., Fielding, G.A., Bandyopadhyay, A., Bose, S.: Effects of zinc and strontium substitution in tricalcium phosphate on osteoclast differentiation and resorption. Biomater. Sci. 1, 74–82 (2013)
Sundfeldt, M., Widmark, M., Wenerberg, A., Karrholm, J., Johansson, C.B., Carlsson, L.V.: Does sodium fluoride in bone cement affect implant fixation? Part I: Bone tissue response, implant fixation and histology in nine rabbits. Mater. Sci: Mater. Med. 13, 1037–1043 (2002)
Qu, H., Wei, M.: The effect of fluoride contents in fluoridated hydroxyapatite on osteoblast behavior. Acta Biomater. 2, 113–119 (2006)
Wang, Y., Zhang, S., Zeng, X., Cheng, K., Qian, M., Weng, W.: In vitro behavior of fluoridated hydroxyapatite coatings in organic-containing simulated body fluid. Mater. Sci. Eng. C. 27, 244–250 (2007)
Bhadang, K.A., Holding, C.A., Thissen, H., Mc-Lean, K.M., Forsythe, J.S., Haynes, D.R.: Biological responses of human osteoblasts and osteoclasts to flame-sprayed coatings of hydroxyapatite and fluorapatite blends. Acta Biomater. 6, 1575–1583 (2010)
Yoon, B.H., Kim, H.W., Lee, S.H., Bae, C.J., Koh, Y.H., Kong, Y.M.: Stability and cellular responses to fluorapatite–collagen composites. Biomater. 26, 2957–2963 (2005)
Zhang, W.G., Wang, L.Z., Liu, Z.: The influence of fluoride on the development of the osteoblast phenotype in rat calvarial osteoblasts: an in vitro study. Shan. K. Qi.Y. X. 7, 88–93 (1998)
Kim, H.W., Lee, E.J., Kim, H.E., Salih, V., Knowles, J.C.: Effect of fluoridation of hydroxyapatite in hydroxyapatite–polycaprolactone composites on osteoblast activity. Biomater. 26, 4395–4404 (2005)
Inoue, M., Nagatsuka, H., Tsujigiwa, H., Inoue, M., LeGeros, R.Z., Yamamoto, T.: In vivo effect of fluoride-substituted apatite on rat bone. Dent. Mater. 24, 398–402 (2005)
Agathopoulos, S., Tulyaganov, D.U., Marques, P.A.A.P., Ferro, M.C., Fernandes, M.H.V., Correia, R.N.: The fluorapatite–anorthite system in biomedicine. Biomater. 24, 1317–1331 (2003)
Tredwin, C.J., Young, A.M., Abou Neel, E.A., Georgiou, G., Knowles, J.C.: Hydroxyapatite, fluor-hydroxyapatite and fluorapatite produced via the sol–gel method: dissolution behaviour and biological properties after crystallization. Mater. Sci.: Mater. Med. 25, 47–53 (2014)
LeGeros, R.Z.: Calcium phosphates in oral biology and medicine. New York University College of Dentistry, New York (1991)
Abdallaha, M.N., Eimar, H., Bassett, D.C., Schnabel, M., Ciobanu, O., Nelea, V., McKee, M.D., Cerruti, M., Tamimi, F.: Diagenesis-inspired reaction of magnesium ions with surface enamel mineral modifies properties of human teeth. Acta Biomater. 37, 174–183 (2016)
Saris, N.E., Mervaala, E., Karppanen, H., Khawaja, J.A., Lewenstam, A.: Magnesium. An update on physiological, clinical and analytical aspects. Clin. Chim. Acta. 294, 1–26 (2000)
Kraus, T., Fischerauer, S.F., Hänzi, A.C., Uggowitzer, P.J., Löffler, J.F., Weinberg, A.M.: Magnesium alloys for temporary implants in osteosynthesis: in vivo studies of their degradation and interaction with bone. Acta Biomater. 8, 1230–1238 (2012)
Staiger, M.P., Pietak, A.M., Huadmai, J., Dias, G.: Magnesium and its alloys as orthopedic biomaterials: a review. Biomater. 27, 1728–1734 (2006)
Hidouri, M., Bouzouita, K., Kooli, F., Khattech, I.: Thermal behaviour of magnesium-containing fluorapatite. Mater. Chem. Phys. 80, 496–505 (2003)
Grigolato, R., Pizzi, N., Brotto, M.C., Corrocher, G., Desando, G., Grigolo, B.: Magnesium-enriched hydroxyapatite as bone filler in an ameloblastoma mandibular defect. Int. Clin. Exp. Med. 8, 281–288 (2015)
Landi, E., Logroscino, G.E., Proietti, L., Tampieri, A., Sandri, M., Sprio, S.: Biomimetic Mg-substituted hydroxyapatite: from synthesis to in vivo behavior. Mater. Sci: Mater. Med. 19, 239–247 (2008)
Ben Abdelkader, S., Khattech, I., Rey, C., Jemal, M.: Synthese, caracterisation et thermochimie d’apatites calco-magnesiennes hydroxylées et fluorées. Therm. Acta. 376, 25–36 (2001)
Suchanek, W.L., Byrappa, K., Shuk, P., Riman, R.E., Janas, V.F., TenHuisen, K.S.: Preparation of magnesium-substituted hydroxyapatite powders by the mechanochemical–hydrothermal method. Biomater. 25, 4647–4657 (2004)
Shannon, R.D.: Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. 32, 751–767 (1976)
Gayathri, B., Muthukumarasamy, N., Velauthapillai, D., Santhosh, S.B., Asokan, V.: Magnesium incorporated hydroxyapatite nanoparticles: preparation, characterization, antibacterial and larvicidal activity. Arab. Chem. 11, 645–654 (2018)
Big, A., Faliai, G., Foresti, E., Gazzano, M., Ripamon, A., Roveri, N.: Magnesium influence on hydroxyapatite crystallization. Inor. Biochem. 49, 69–78 (1993)
Bertoni, E., Bigi, A., Cojazzi, G., Gandol, M., Panzavolta, S., Roveri, N.: Nanocrystals of magnesium and fluoride substituted hydroxyapatite. Inorg. Biochem. 72, 29–35 (1998)
Mayer, I., Scblam, R., Featberstone, J.D.B.: Magnesium-containing carbonate apatites. Inorg. Biochem. 66, 1–6 (1997)
Fowler, B.O.: Infrared Studies of Apatites. I. Vibrational assignments for calcium, strontium, and barium hydroxyapatites utilizing isotopic substitution. Inorg. Chem. 13, 194–207 (1974)
Harrison, J., Melville, A.J., Forsythe, J.S., Muddle, B.C., Trounson, A.O., Gross, K.A., Mollard, R.: Sintered hydroxyfluorapatites—IV: the effect of fluoride substitutions upon colonisation of hydroxyapatites by mouse embryonic stem cells. Biomater. 25, 4977–4986 (2004)
Rodriguez-Lorenzo, L.M., Hart, J.N., Gross, K.A.: Influence of fluorine in the synthesis of apatites. Synthesis of solid solutions of hydroxy-fluorapatite. Biomater. 24, 3777–13785 (2003)
Jha, L.J., Best, S.M., Knowles, J.C., Rehman, I., Santos, J.D., Bonfield, W.: Preparation and characteri zation of fluoride-substituted apatites. Mater. Sci: Mater. Med. 8, 185–191 (1997)
Rodriguez-Lorenzo, L.M., Hart, J.N., Gross, K.A.: Structural and chemical analysis of well-crystallized hydroxyfluorapatites. Phys. Chem. B. 107, 8316–8320 (2003)
Fathi, M.H., Zahrani, E.M.: Mechanical alloying synthesis and bioactivity evaluation of nanocrystalline fluoridated hydroxyapatite. Cryst. Gro. 311, 1392–1403 (2009)
Nasr, S., Ben Salem, E., Bouzouita, K.: Effect of fluorine on the thermal stability of the magnesium-substituted hydroxyapatite. Ann. Chim. Sci. Mater. 36, 159–176 (2011)
Iqbal, K.N., Ijaz, K., Zahid, M., Khan, A.S., Abdul Kadir, M.R., Hussain, R., A.-ur-Rehman, Jawwad, A.D., I-ur-Rehman, Chaudhry Aqif, A.: Microwave assisted synthesis and characterization of magnesium substituted calcium phosphate bioceramics. Mater. Sci. Eng. C. 56, 286–293 (2015)
Fadeev, I.V., Shvorneva, L.I., Barinov, S.M., Orlovskii, V.P.: Synthesis and structure of magnesium-substituted hydroxyapatite. Inorg. Mater. 39, 947–950 (2003)
Baravelli, S., Bigi, A., Ripamonti, A., Roveri, N.: Thermal behavior of bone and synthetic hydroxyapatites submitted to magnesium interaction in aqueous medium. Inorg. Biochem. 20, 1–12 (1984)
Qi, G., Zhang, S., Khor, K.A., Liu, C., Zeng, X., Weng, W., Qian, M.: In vitro effect of magnesium inclusion in sol–gel derived, apatite. Thin Solid Films. 516, 5176–5180 (2008)
Rice, R.W.: Microstructure dependence of mechanical behavior of ceramics. Treat. on Mater. Sci. Tech. 11, 199–381 (1977)
Furukawat, M., Horita, Z., Nemoto, M., Valiev, R.Z., Langdon, T.G.: Microhardness measurements and the Hall-Petch relationship in an al mg alloy with submicrometer grain size. Acta Mater. 44, 4619–4629 (1996)
Thanigai Arul, K., Kolanthai, E., Manikandan, E., Bhalerao, G.M.: Green synthesis of magnesium ion incorporated nanocrystalline hydroxyapatite and their mechanical, dielectric and photoluminescence properties. Mater. Res. Bull. 67, 55–62 (2015)
Vaßen, R., Stover, D.: Processing and properties of nanophase non-oxide ceramics. Mater. Sci. Eng. 301, 59–68 (2001)
Adzila, S., Ramesh, S., Sopyan, I.: Properties of magnesium doped nanocrystalline hydroxyapatite synthesize by mechanochemical method. ARPN. Eng. App. Sci. 11, 14097–14100 (2016)
Ramesh, S., Jeffrey, C.K.L., Tan, C.Y., Wong, Y.H., Ganesan, P., Kutty, M.G., Chandran, H., Devaraj, P.: Sintering behavior and properties of magnesium orthosilicate-hydroxyapatite ceramic. Ceram. Int. 42, 15756–15761 (2016)
Yetmez, M., Erkmen, Z.E., Kalkandelen, C., Ficai, A., Oktar, F.N.: Sintering effects of mullite-doping on mechanical properties of bovine hydroxyapatite. Mater. Sci. Eng. C. 77, 470–475 (2017)
Bouslama, N., Chevalier, Y., Bouaziz, J., Ben Ayed, F.: Influence of the sintering temperature on Young’s modulus and the shear modulus of tricalcium phosphate fluorapatite composites evaluated by ultrasound techniques. Mater. Chem. Phys. 141, 289–297 (2013)
Franz, E.D., Telle, R.: Reaction hot pressing of fluorapatite for dental implants. Hight. Tech.Ceram. 1, 31–41 (1987)
Elliot, J.C.: Structure and Chemistry of the apatite and other calcium orthophosphates. Amesterdam. (1994)
Akao, M., Aoki, H., Kato, K.: Mechanical properties of sintered hydroxyapatite for prosthetic applications. Mater. Sci. 16, 809–812 (1981)
Vashishth, D., Tanner, K.E., Bonfield, W.: Fatigue of cortical bone under combined axial-torsional loading. Ortho. Res. 13, 414–420 (2001)
Ohman, C., Zwierzak, I., Baleani, M., Viceconti, M.: Human bone hardness seems to depend on tissue type but not on anatomical site in the long bones of an old subject. Eng. Med. 227, 200–206 (2012)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ammar, H., Nasr, S., Ageorges, H. et al. Sintering and mechanical properties of magnesium containing hydroxyfluorapatite. J Aust Ceram Soc 56, 931–942 (2020). https://doi.org/10.1007/s41779-019-00422-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41779-019-00422-7